Therapeutic gene modulation
Therapeutic gene modulation refers to the practice of altering the expression of a gene at one of various stages, with a view to alleviate some form of ailment. It differs from gene therapy in that gene modulation seeks to alter the expression of an endogenous gene (perhaps through the introduction of a gene encoding a novel modulatory protein) whereas gene therapy concerns the introduction of a gene whose product aids the recipient directly.
Modulation of gene expression can be mediated at the level of
Transcriptional gene modulation
An approach to therapeutic modulation utilizes agents that modulate endogenous transcription by specifically targeting those genes at the gDNA level. The advantage to this approach over modulation at the mRNA or protein level is that every cell contains only a single gDNA copy. Thus the target copy number is significantly lower allowing the drugs to theoretically be administered at much lower doses.[citation needed]
This approach also offers several advantages over traditional
There are three major categories of agents that act as transcriptional gene modulators: triplex-forming oligonucleotides (TFOs), synthetic polyamides (SPAs), and DNA binding proteins.[2]
Triplex-forming oligonucleotides
What are they
Triplex-forming oligonucleotides (TFO) are one potential method to achieve therapeutic gene modulation. TFOs are approximately 10-40 base pairs long and can bind in the major groove in duplex DNA which creates a third strand or a triple helix.[2][3] The binding occurs at polypurine or polypyrimidine regions via Hoogsteen hydrogen bonds to the purine (A / G) bases on the double stranded DNA that is already in the form of the Watson-Crick helix.[4]
How they work
TFOs can be either polypurine or polypyrimidine molecules and bind to one of the two strands in the double helix in either parallel or antiparallel orientation to target polypurine or polypyrimidine regions. Since the DNA-recognition codes are different for the parallel and the anti-parallel fashion of TFO binding, TFOs composed of pyrimidines (C / T) bind to the purine-rich strand of the target double helix via Hoogsteen hydrogen bonds in a parallel fashion.[3] TFOs composed of purines (A / G), or mixed purine and pyrimidine bind to the same purine-rich strand via reverse Hoogsteen bonds in an anti-parallel fashion. TFO's can recognize purine-rich target strands for duplex DNA.[2]

Complications and limitations
In order for TFO motifs to bind in a parallel fashion and create hydrogen bonds, the nitrogen atom at position 3 on the cytosine residue needs to be protonated, but at physiological pH levels it is not, which could prevent parallel binding.[2]
Another limitation is that TFOs can only bind to purine-rich target strands and this would limit the choice of
Other limitations include concerns regarding binding affinity and specificity, in vivo stability, and uptake into cells. Researchers are attempting to overcome these limitations by improving TFO characteristics through chemical modifications, such as modifying the TFO backbone to reduce electrostatic repulsions between the TFO and the DNA duplex. Also due to their high molecular weight, uptake into cells is limited and some strategies to overcome this include DNA condensing agents, coupling of the TFO to hydrophobic residues like cholesterol, or cell permeabilization agents.[2]
What can they do
Scientists are still refining the technology to turn TFOs into a therapeutic product and much of this revolves around their potential applications in antigene therapy. In particular they have been used as inducers of site-specific mutations, reagents that selectively and specifically cleave target DNA, and as modulators of gene expression.[6] One such gene sequence modification method is through the targeting DNA with TFOs to active a target gene. If a target sequence is located between two inactive copies of a gene, DNA ligands, such as TFOs, can bind to the target site and would be recognized as DNA lesions. To fix these lesions, DNA repair complexes are assembled on the targeted sequence, the DNA is repaired. Damage of the intramolecular recombination substrate can then be repaired and detected if resection goes far enough to produce compatible ends on both sides of the cleavage site and then 3' overhangs are ligated leading to the formation of a single active copy of the gene and the loss of all the sequences between the two copies of the gene.[4]
In model systems TFOs can inhibit gene expression at the DNA level as well as induce targeted mutagenesis in the model.[6] TFO-induced inhibition of transcription elongation on endogenous targets have been tested on cell cultures with success.[7] However, despite much in vitro success, there has been limited achievement in cellular applications potentially due to target accessibility.
TFOs have the potential to silence silence gene by targeting transcription initiation or elongation, arresting at the triplex binding sites, or introducing permanent changes in a target sequence via stimulating a cell's inherent repair pathways. These applications can be relevant in creating cancer therapies that inhibit gene expression at the DNA level. Since aberrant gene expression is a hallmark of cancer, modulating these endogenous genes' expression levels could potentially act as a therapy for multiple cancer types.
Synthetic polyamides
Synthetic polyamides are a set of small molecules that form specific hydrogen bonds to the minor groove of DNA. They can exert an effect either directly, by binding a regulatory region or transcribed region of a gene to modify transcription, or indirectly, by designed conjugation with another agent that makes alterations around the DNA target site.
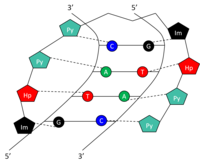
Structure
Specific bases in the minor groove of DNA can be recognized and bound by small synthetic polyamides (SPAs). DNA-binding SPAs have been engineered to contain three polyamide amino acid components: hydroxypyrrole (Hp), imidazole (Im), and pyrrole (Py).[10] Chains of these amino acids loop back on themselves in a hairpin structure. The amino acids on either side of the hairpin form a pair which can specifically recognize both sides of a Watson-Crick base pair. This occurs through hydrogen bonding within the minor groove of DNA. The amide pairs Py/Im, Py/Hp, Hp/Py, and Im/Py recognize the Watson-Crick base pairs C-G, A-T, T-A, and G-C, respectively (Table 1). See figure for a graphical representation of 5'-GTAC-3' recognition by a SPA. SPAs have low toxicity, but have not yet been used in human gene modulation.
| Amide Pair | Nucleotide Pair |
|---|---|
| Py/Im | C-G |
| Py/Hp | A-T |
| Hp/Py | T-A |
| Im/Py | G-C |
Limitations and workarounds
The major structural drawback to unmodified SPAs as gene modulators is that their recognition sequence cannot be extended beyond 5 Watson-Crick base pairings. The natural curvature of the DNA minor groove is too tight a turn for the hairpin structure to match. There are several groups with proposed workarounds to this problem. This approach has increased the recognition length to up to eleven Watson-Crick base pairs.


Direct modulation
SPAs may inhibit transcription through binding within a transcribed region of a target gene. This inhibition occurs through blocking of elongation by an RNA polymerase.
SPAs may also modulate transcription by targeting a transcription regulator binding site. If the regulator is an activator of transcription, this will decrease transcriptional levels. As an example, SPA targeting to the binding site for the activating transcription factor TFIIIA has been demonstrated to inhibit transcription of the downstream 5S RNA.[17] In contrast, if the regulator is a repressor, this will increase transcriptional levels. As an example, SPA targeting to the host factor LSF, which represses expression of the human immunodeficiency virus (HIV) type 1 long terminal repeat (LTR), blocks binding of LSF and consequently de-represses expression of LTR[18] .

Conjugate modulation
SPAs have not been shown to directly modify DNA or have activity other than direct blocking of other factors or processes. However, modifying agents can be bound to the tail ends of the hairpin structure. The specific binding of the SPA to DNA allows for site-specific targeting of the conjugated modifying agent.
SPAs have been paired with the DNA-alkylating moieties cyclopropylpyrroloindole[19] and chlorambucil[20] that were able to damage and crosslink SV40 DNA. This effect inhibited cell cycling and growth. Chlorambucil, a chemotherapeutic agent, was more effective when conjugated to an SPA than without.
In 2012, SPAs were conjugated to SAHA, a potent histone deacetylase (HDAC) inhibitor.[21] SPAs with conjugated SAHA were targeted to Oct-3/4 and Nanog which induced epigenetic remodeling and consequently increased expression of multiple pluripotency related genes in mouse embryonic fibroblasts.
Designer zinc-finger proteins
What they are/structure
Designer zinc-finger proteins are engineered

How they work
Zinc-finger motifs bind into the


Effects and impacts on gene modulation
Designer zinc-finger proteins can modulate
Effector domains bound to the zinc-finger can also have comparable effects. It is the function of these effector domains which are arguably the most important with respect to the use of designer zinc-finger proteins for therapeutic gene modulation. If a
In the first case to successfully demonstrate the use of designer zinc-finger proteins to modulate gene expression
Post-transcriptional gene modulation
The major approach to post-transcriptional gene modulation is via RNA interference (RNAi). The primary problem with using RNAi in gene modulation is drug delivery to target cells.[33][34] RNAi gene modulation has been successfully applied to mice toward the treatment of a mouse model for inflammatory bowel disease.[35] This treatment utilized liposome-based beta-7 integrin-targeted, stabilized nanoparticles entrapping short interfering RNAs (siRNAs). There are several other forms of RNAi delivery, including: polyplex delivery, ligand-siRNA conjugates, naked delivery, inorganic particle deliver using gold nanoparticles, and site specific local delivery.[36]
Clinical significance
Designer zinc-finger proteins, on the other hand, have undergone some trials in the
See also
References
- PMID 10799600.
- ^ PMID 14576293.
- ^ PMID 21417933.
- ^ PMID 18460344.
- PMID 23658228.
- ^ PMID 12467644.
- PMID 10760257.
- ^ PMID 11281837.
- PMID 11553460.
- ^ S2CID 205023593.
- PMID 12831879.
- PMID 2838035.
- S2CID 4335955.
- PMID 10940255.
- PMID 12150881.
- PMID 12059208.
- S2CID 4358491.
- PMID 12414976.
- PMID 12582240.
- PMID 12196541.
- PMID 22848790.
- ^ PMID 16298089.
- ^ ISBN 978-0-8053-9592-1.
- PMID 8460130.
- PMID 11433278.
- ^ S2CID 12701336.
- PMID 14602907.
- PMID 8577732.
- S2CID 4390010.
- PMID 10660690.
- PMID 22782891.
- PMID 14695215.
- PMID 16481219.
- PMID 16834561.
- PMID 18239128.
- PMID 22186795.
- ^ "Identifier NCT00080392.Modulation of Vascular Endothelial Growth Factor (VEGF) Using an Engineered Zinc-Finger Transcription Factor to Treat Lower Limb Intermittent Claudication". ClinicalTrials.gov. U.S. National Institutes of Health. 30 December 2011. Retrieved 25 July 2013.
- PMID 22378343.
