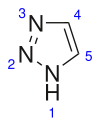
1,2,3-Triazole
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1H-1,2,3-Triazole | |||
| Other names
1,2,3-Triazole
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.128.405 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H3N3 | |||
| Molar mass | 69.0654 | ||
| Appearance | colorless liquid | ||
| Density | 1.192 | ||
| Melting point | 23 to 25 °C (73 to 77 °F; 296 to 298 K) | ||
| Boiling point | 203 °C (397 °F; 476 K) | ||
| very soluble | |||
| Acidity (pKa) | 9.4[1] | ||
| Basicity (pKb) | 1.2[1] | ||
| Hazards | |||
| GHS labelling: | |||

| |||
| Warning | |||
| H315, H319, H335 | |||
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |||
| Related compounds | |||
Related compounds
|
1,2,4-triazole imidazole
| ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,2,3-Triazole is one of a pair of
Synthesis
The unsubstituted ring can be produced by an oxidative coupling of glyoxal, hydrazine and sodium nitrite.[3]
A wide range of methods exist for forming substituted 1,2,3-triazoles.
Properties
The 2H-1,2,3-triazole tautomer is the major form in aqueous solution.[6] It is a surprisingly stable structure compared to other organic compounds with three adjacent nitrogen atoms. However, flash vacuum
Applications
1,2,3-Triazole finds use in research as a bioisostere in medicinal chemistry[7] building block for more complex chemical compounds, including pharmaceutical drugs such as mubritinib and tazobactam.
References
- ^ a b "1,2,3-triazole - an overview". Comprehensive Heterocyclic Chemistry. 1984.
- ISBN 0-582-01421-2.
- .
- .
- PMID 29630830.
- .
- PMID 28676407.