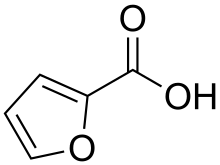
2-Furoic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
Furan-2-carboxylic acid[1] | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 110149 | |
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.001.639 |
| 3056 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H4O3 | |
| Molar mass | 112.084 g·mol−1 |
| Appearance | White/ Off-White (Beige) Crystalline Powder |
| Density | 0.55 g/cm3 |
| Melting point | 128 to 132 °C (262 to 270 °F; 401 to 405 K) |
| Boiling point | 230 to 232 °C (446 to 450 °F; 503 to 505 K) |
| Easily soluble in cold and hot water, 27.1 g/L | |
| Acidity (pKa) | 3.12 at 25 °C |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritating to eyes, respiratory system and skin. |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds
|
2-Thiophenecarboxylic acid, 3-Furoic acid, Furfuryl alcohol, 2,5-Furandicarboxylic acid, Furfurylamine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Furoic acid is an organic compound, consisting of a furan ring and a carboxylic acid side-group. Along with other furans, its name is derived from the Latin word furfur, meaning bran, from which these compounds were first produced.[2] The salts and esters of furoic acids are known as furoates. 2-Furoic acid is most widely encountered in food products as a preservative and a flavouring agent, where it imparts a sweet, earthy flavour.[3]
History
The compound was first described by Carl Wilhelm Scheele in 1780, who obtained it by the dry distillation of mucic acid. For this reason it was initially known as pyromucic acid. This was the first known synthesis of a furan compound, the second being furfural in 1821.[4][5] Despite this, it was furfural which came to set naming conventions for later furans.
Preparation and synthesis
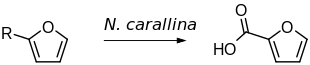
2-Furoic acid can be synthesized by the oxidation of either furfuryl alcohol or furfural. This can be achieved either chemically or biocatalytically.
The current industrial route involves the
Applications and occurrences

In terms of commercial uses, 2-furoic acid is often used in the production of furoate esters, some of which are drugs and pesticides.[10]
In foods
It is a flavoring ingredient and achieved a
2-Furoic acid helps sterilize and
Optic properties
2-Furoic acid crystals are highly transparent in the 200–2000 nm wavelength region, are stable up to 130 °C, and generally have low absorption in the
Microbial metabolism
2-Furoic acid can be the sole source of carbon and energy for the organism Pseudomonas putida. The organism aerobically degrades the compound. [15] [16]
Hazards
The LD50 is 100 mg/kg (oral, rats).[17]
References
- ISBN 978-0-85404-182-4.
- ISBN 0-444-52239-5.
- ^ ISBN 0-8493-9414-7.
- . From p. 141: "Ich verbinde mit diese Bitte noch die Bemerkung, … Bittermandelöl riechende Materie enthält, … " (I join to this request also the observation that the formic acid which is formed by the simultaneous reaction of sulfuric acid and manganese peroxide with sugar and which contains a volatile material that appears oily in an isolated condition and that smells like a mixture of cassia and bitter almond oil … )
- JSTOR 111080.
- See also: Stenhouse, John (1850). "On the oils produced by the action of sulphuric acid upon various classes of vegetables". Philosophical Transactions of the Royal Society of London. 140: 467–480. S2CID 186214485.
- See also: Stenhouse, John (1850). "On the oils produced by the action of sulphuric acid upon various classes of vegetables". Philosophical Transactions of the Royal Society of London. 140: 467–480.
- S2CID 101343477.
- ^ Pérez, Herminia (2009). "Microbial biocatalytic preparation of 2-furoic acid by oxidation of 2-furfuryl alcohol and 2-furanaldehyde with Nocardia corallina". African Journal of Biotechnology. 8 (10).
- S2CID 19657328.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- S2CID 42398133.
- .
- S2CID 222280614.
- .
- .
- .
- PMID 16347977.
- ISBN 978-3527306732.
Further reading
- ISBN 978-0-08-037941-8.
- "2-Furoic Acid [Material Safety Data Sheet]". Sciencelab.com. October 9, 2005. Archived from the original on October 17, 2012. Retrieved March 15, 2013.

