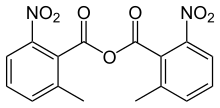
2-Methyl-6-nitrobenzoic anhydride
Appearance
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methyl-6-nitrobenzoic anhydride | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.156.789 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H12N2O7 | |
| Molar mass | 344.279 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Methyl-6-nitrobenzoic anhydride is an organic acid anhydride also known as the Shiina reagent,[1][2] having a structure wherein carboxylic acids undergo intermolecular dehydration condensation. It was developed in 2002 by Prof. Isamu Shiina (Tokyo University of Science, Japan).[3] The compound is often abbreviated MNBA.
Abstract
The reagent is used for synthetic reactions wherein medium- and large-sized lactones are formed from hydroxycarboxylic acids via intramolecular ring closure (Shiina macrolactonization).[4][5] The reaction proceeds at room temperature under basic or neutral conditions. This reagent can be used not only for macrolactonization but also for esterification, amidation, and peptide coupling.
See also
- Condensation reaction
- Fischer-Speier esterification
- Mitsunobu reaction
- Shiina esterification
- Steglich esterification
- Yamaguchi esterification
References
- ^ "Named Reagents". OChemOnline. Archived from the original on 2017-09-04.
- PMID 28741352.
- .
- PMID 15058924.
- .
External links
- 2-Methyl-6-nitrobenzoic Anhydride (MNBA)
- Enantioselective Total Synthesis of Octalactin A Using Asymmetric Aldol Reactions and a Rapid Lactonization To Form a Medium-Sized Ring
- Total Synthesis of Iejimalide B. An Application of the Shiina Macrolactonization
