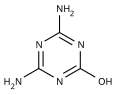
Ammeline
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
4,6-Diamino-1,3,5-triazin-2-ol | |||
| Other names
2,4-Diamino-6-hydroxy-1,3,5-triazine
4,6-Diamino-2-hydroxy-1,3,5-triazine 4,6-diamino-1,3,5-triazin-2(1H)-one | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.010.415 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H5N5O | |||
| Molar mass | 127.107 g·mol−1 | ||
| Appearance | White powder | ||
| Melting point | N/A (decomposes before melting) | ||
| Trace | |||
| Solubility | Soluble in aqueous alkalies and mineral acids, but not acetic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Ammeline (4,6-diamino-2-hydroxy-1,3,5-triazine) is a triazine derivative. It is the hydrolysis product of melamine.[1]
Synthesis
Ammeline can be synthesized by the
dicyandiamide and 1 mole of biuret
.
- 2 C2H4N4 + C2H5N3O2 → 2C3H5N5O + NH3
Chemical properties
Ammeline is weakly acidic with
chromate, and oxalate salts. Ammeline reacts with boiling dilute hydrochloric acid to form melem and ammonia
.
Ammeline is the first step in melamine hydrolysis. Further hydrolysis (e.g. boiling ammeline with dilute alkali) yields ammelide.
References
- .