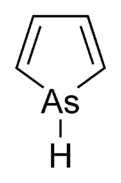
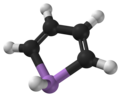
Arsole
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1H-Arsole | |||
| Other names
Arsenole
Arsacyclopentadiene | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H4AsH | |||
| Molar mass | 128.00 g mol−1 | ||
| Related compounds | |||
Related compounds
|
Pyrrole, phosphole, bismole, stibole | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Arsole, also called arsenole
Nomenclature
Arsole belongs to the series of heterocyclic pnictogen compounds. The naming of cyclic
| Ring size | Unsaturated ring |
Saturated ring |
|---|---|---|
| 3 | Arsirene | Arsirane |
| 4 | Arsete | Arsetane |
| 5 | Arsole | Arsolane |
| 6 | Arsinine |
Arsinane |
| 7 | Arsepine | Arsepane |
| 8 | Arsocine | Arsocane |
| 9 | Arsonine | Arsonane |
| 10 | Arsecine | Arsecane |
Because of its similarity to the English slang word "
Properties
| M | d(M-C), Å |
d(M-H), Å | α(C-M-C), ° | E, kJ/mol |
|---|---|---|---|---|
| N | 1.37 | 1.01 | 110 | 0 |
| P | 1.81 | 1.425 | 90.5 | 67 |
| As | 1.94 | 1.53 | 86 | 125 |
| Sb | 2.14 | 1.725 | 80.5 | 160 |
| Bi | 2.24 | 1.82 | 78 | 220 |
Arsole itself has not been isolated experimentally yet, but the molecular geometry and electronic configuration of arsole have been studied theoretically. Calculations also addressed properties of simple arsole derivatives, where hydrogen atoms are substituted by other atoms or small hydrocarbon groups, and there are experimental reports on chemical properties of more complex arsole derivatives. The situation is similar for other C4H4MH metalloles where M = P, As, Sb and Bi.
Planarity
Calculations suggest that whereas pyrrole (C4H4NH) molecule is planar, phosphole (C4H4PH) and heavier metalloles are not, and their
Aromaticity
Chemical properties (arsole derivatives)
Chemical properties of arsole derivatives have been studied experimentally; they are similar to those of
Substituting in this reaction
Reaction of phenylarsenous dichloride with linear diphenyls results in 1,2,5-triphenylarsole (see below), a solid with a melting point of about 170 °C.
See also
- Pyrrole, a nitrogen analog.
- Furan, an oxygen analog.
- Thiophene, a sulfur analog.
- Simple aromatic rings
- Varsol, a petroleum distillate with a boiling range of 150–200 °C.
References
- ^ Mann 1970: "In English this ring system has frequently named arsenole 'for euphony'."
- ^ .
Using quantum chemical methodology, we reinvestigate the aromaticity of the much debated arsole, using the newly developed gauge-including magnetically induced currents (GIMIC) method. GIMIC provides a quantitative measure of the induced ring current strength, showing arsole to be moderately aromatic.
- PMID 19099440.
- ^ "Revision of the Extended Hantzsch-Widman System of Nomenclature for Heteromonocycles Archived 2017-09-08 at the Wayback Machine" at IUPAC, retrieved 29 Sept 2008
- ISBN 978-0-7514-0389-3. Retrieved 15 March 2011.
- ISBN 978-1-85788-372-5. Retrieved 15 March 2011.
- ISBN 978-1-84816-207-5(pbk). See also the Web page "Molecules with Silly or Unusual Names Archived 2009-09-07 at the Wayback Machine" at the School of Chemistry, University of Bristol, (retrieved 29 Sept 2008)
- ^ .
- ^ ISBN 978-3-540-68329-2. Retrieved 21 March 2011.
- S2CID 97528113.
- ^ ISBN 978-0-471-37489-3. Archivedfrom the original on 13 June 2014. Retrieved 21 March 2011.
- .
- ISBN 978-0-85186-753-3. Archivedfrom the original on 13 June 2014. Retrieved 23 March 2011.
- (PDF) from the original on 2011-05-24. Retrieved 2011-03-23.
- .