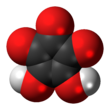
Croconic acid
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
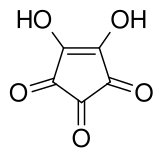
4,5-Dihydroxycyclopent-4-ene-1,2,3-trione | |||
| Other names
Crocic acid
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
ECHA InfoCard
|
100.201.686 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H2O5 | |||
| Molar mass | 142.07 | ||
| Melting point | > 300 °C (572 °F; 573 K) (decomposes) | ||
| Acidity (pKa) | 0.80, 2.24 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Croconic acid (also known as 4,5-dihydroxycyclopentenetrione, crocic acid or pentagonic acid) is a
The compound is acidic and loses the
Croconic acid also forms
History
Croconic acid and potassium croconate dihydrate were discovered by Leopold Gmelin in 1825, who named the compounds from Greek κρόκος meaning "crocus" or "egg yolk".[7] The structure of ammonium croconate was determined by Baenziger et al. in 1964. The structure of K2C5O5·2H2O was determined by Dunitz in 2001.[8]
Structure
In the solid state, croconic acid has a peculiar structure consisting of pleated strips, each "page" of the strip being a planar ring of 4 molecules of C5O5H2 held together by
In the solid alkali metal salts, the croconate anions and the alkali cations form parallel columns.[7] In the mixed salt K3(HC5O5)(C5O5)·2H2O, which formally contains both one croconate dianion C5O2−5 and one hydrogencroconate monoanion (HC5O−5), the hydrogen is shared equally by two adjacent croconate units.[7]
Salts of the croconate anion and its derivatives are of interest in
Infrared and Raman assignments indicate that the equalization of the carbon–carbon bond lengths, thus the electronic delocalization, follows with an increase in counter-ion size for salts.[6] This result leads to a further interpretation that the degree of aromaticity is enhanced for salts as a function of the size of the counter-ion. The same study provided quantum mechanical DFT calculations for the optimized structures and vibrational spectra which were in agreement with experimental findings. The values for calculated theoretical indices of aromaticity also increased with counterion size.
The croconate anion forms

The croconate anion also forms compounds with trivalent cations such as
See also
- Croconate violet
- Croconate blue
- Rhodizonic acid
- Squaric acid
- Deltic acid
- Cyclopentanepentone (leuconic acid)
References
- ^ .
- ^ Miller, W. A. (1868). Elements of Chemistry: Theoretical and Practical (4th ed.). Longmans.[page needed]
- ^ Turner, E. Elements of Chemistry.[page needed]
- ^ .
- .
- ^ .
- ^ PMID 11933108.
- PMID 11353510.
- ^ S2CID 205219520.
- .
- ^ .
- .
- .
- ISSN 1528-7483.
External links
 Media related to Croconic acid at Wikimedia Commons
Media related to Croconic acid at Wikimedia Commons