Hydrocyanation
In
Hydrocyanation of unactivated alkenes
Industrially, hydrocyanation is commonly performed on
- RCH=CH2 + HCN → RCH2−CH2−CN
Stoichiometry and mechanism
The reaction involves the addition of H+ and cyanide (−CN) to the substrate. Usually the substrate is an alkene and the product is a nitrile.
The reaction proceeds via the oxidative addition of HCN to a low-valent metal complex to give a hydrido cyanide complex. Subsequent binding of the alkene gives the intermediate M(H)(CN)Ln(alkene), which then undergoes migratory insertion to give an alkylmetal cyanide. The cycle is completed by the reductive elimination of the nitrile.
In the case of nickel-based systems, catalyst deactivation involves formation of dicyanonickel(II) species, which are unreactive toward alkenes. The dicyanide arises via two pathways (L = phosphite):[1]
- Ni(H)(CN)L2 + HCN → Ni(CN)2L2 + H2
- Ni(R)(CN)L2 + HCN → Ni(CN)2L2 + RH
Asymmetric hydrocyanation
Most alkenes are prochiral, meaning in this context that their hydrocyanation generates chiral nitriles. Conventional hydrocyanation catalysts, e.g. Ni(P(OR)3)4, catalyse the formation of racemic mixtures. When however the supporting ligands are chiral, the hydrocyanation can be highly
Applications
The most important industrial application is the nickel-catalyzed synthesis of
This process consists of three steps: hydrocyanation of butadiene to a mixture of 2-methyl-butene-3-nitrile (2M3BM) and pentene-3-nitrile (3PN), an
Asymmetric hydrocyanation
Hydrocyanation is important due to the versatility of
History
Hydrocyanation was first reported by Arthur and Pratt in 1954, when they homogeneously catalyzed the hydrocyanation of linear alkenes.[5] The industrial process for catalytic hydrocyanation of butadiene to adiponitrile was invented by William C. Drinkard.
Transhydrocyanation
In transhydrocyanation, an equivalent of HCN is transferred from a cyanohydrin, e.g. acetone cyanohydrin, to another HCN acceptor. The transfer is an equilibrium process, initiated by base. The reaction can be driven by trapping reactions or by the use of a superior HCN acceptor, such as an aldehyde.[6]
With unsaturated carbonyl compounds
α,β-unsaturated carbonyl compounds undergo hydrocyanation in the absence of metal catalysts. One manifestation is a special case of the
(1)
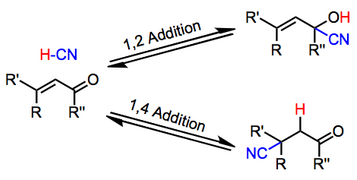
Generally acidic conditions favor 1,2-adducts, while basic conditions favor 1,4-adducts. Additions of alkali metal cyanides, for instance, lead exclusively to 1,4-addition.[8] In contrast to alkali metal cyanides and cyanoaluminates, Lewis acidic cyanides, such as TMSCN, favor 1,2-addition. Acetylenic substrates undergo the reaction; however the scope of this reaction is limited and yields are often low.[9]
(5)

1,4-Addition to imines has been observed in a few cases, although imines are often base labile.[10]
(6)

Esters,[11] nitriles[12] and other carbonyl derivatives also undergo conjugative hydrocyanation.
When alkali metal cyanides are used, at least partial neutralization of the reaction medium is usually necessary. Neutralization can be accomplished through an acidic group on the substate itself (internal neutralization).[13] or through the addition of an external acid (external neutralization). Acetic acid is commonly used for this purpose, in a procedure pioneered by Lapworth.[14]
(7)

Conjugative hydrocyanation was used to prepare the steroidal D ring.[15] Diastereoselectivity is generally high in these addition reactions, and the resulting β-cyano carbonyl compounds can be converted to a number of steroidal products.
References
- ^ ISBN 1-4020-2000-7
- .
- S2CID 96253038.
- PMID 17902667.
- ISSN 0002-7863.
- ISBN 978-0471936237.
- ISBN 0471264180.
- PMID 18914000.
- ^ Kurtz, P. Ann. Chem. 1951, 572, 23.
- ^ Nagata, W. ; Yoshioka, M. ; Okumura, T. ; Murakami, M. J. Chem. Soc., C, 1970, 2355.
- ^ Allen, H. ; Johnson, B. Org. Synth. 1963, Coll. Vol. IV, 804.
- ^ Kurtz, P. Ann. Chem. 1951, 572, 23.
- ^ Crabbé, P.; Pérez, M.; Vera, G. Can. J. Chem. 1963, 41, 156.
- ^ Lapworth, A. ; Wechsler, E. J. Chem. Soc. 1910, 97, 38.
- ^ Nagata, W. ; Terasawa, T. ; Hirai, S. ; Takeda, K. Tetrahedron Lett., 1960, 17, 27.

