Hydrofluoric acid
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Fluorane[1]
| |||
| Other names
Fluorhydric acid
Hydronium fluoride | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| HF (aq) | |||
| Appearance | Colorless liquid | ||
| Density | 1.15 g/mL (for 48% soln.) | ||
| Acidity (pKa) | 3.17[2] | ||
| Hazards[3] | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H280, H300+H310+H330, H314 | |||
| P260, P262, P264, P270, P271, P280, P284, P301+P310, P301+P330+P331, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P321, P322, P330, P361, P363, P403+P233, P405, P410+P403, P501 | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrofluoric acid is a
Uses
Production of organofluorine compounds
The principal use of hydrofluoric acid is in
Production of inorganic fluorides
Most high-volume inorganic fluoride compounds are prepared from hydrofluoric acid. Foremost are Na3AlF6,
Etchant, cleaner

It is used in the
A 5% to 9% hydrofluoric acid gel is also commonly used to etch all ceramic dental restorations to improve bonding.
Oil refining
In a standard
Production
Hydrofluoric acid was first prepared in 1771, by Carl Wilhelm Scheele.[9] It is now mainly produced by treatment of the mineral fluorite, CaF2, with concentrated sulfuric acid at approximately 265 °C.
- CaF2 + H2SO4 → 2 HF + CaSO4
The acid is also a by-product of the production of
Because of its high reactivity toward glass, hydrofluoric acid is stored in fluorinated
Properties
In dilute aqueous solution hydrogen fluoride behaves as a weak acid,[10]
- pKa= 3.17
This
- HF + F− ⇌ HF−
2 log K = 0.6
It is assumed that polymerization occurs as the concentration increases. This assumption is supported by the isolation of a salt of a tetrameric anion H
3F−
4[14] and by low-temperature X-ray crystallography.[13] The species that are present in concentrated aqueous solutions of hydrogen fluoride have not all been characterized; in addition to HF−
2 which is known[11] the formation of other polymeric species, H
n−1F−
n, is highly likely.
The Hammett acidity function, H0, for 100% HF was first reported as -10.2,[15] while later compilations show -11, comparable to values near -12 for pure sulfuric acid.[16][17]
Acidity
Unlike other
2O, and F−
anions to form clusters.[19] At high concentrations, HF molecules undergo homoassociation to form polyatomic ions (such as bifluoride, HF−
2) and protons, thus greatly increasing the acidity.[20] This leads to protonation of very strong acids like hydrochloric, sulfuric, or nitric acids when using concentrated hydrofluoric acid solutions.[21] Although hydrofluoric acid is regarded as a weak acid, it is very corrosive, even attacking glass when hydrated.[20]
Dilute solutions are weakly acidic with an
In thermodynamic terms, HF solutions are highly
- H2O + HF ⇌ H3O+⋅F−
With increasing concentration of HF the concentration of the hydrogen difluoride ion also increases.[11] The reaction
- 3 HF ⇌ HF−
2 + H2F+
is an example of homoconjugation.
Health and safety
This article needs more primary sources. (November 2019) |  |
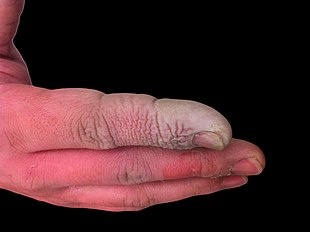
In addition to being a highly
Hydrofluoric burns are treated with a calcium gluconate gel.
In popular culture
In the episodes "Cat's in the Bag..." and "Box Cutter" of the crime drama television series Breaking Bad, Walter White and Jesse Pinkman use hydrofluoric acid to chemically disincorporate bodies of gangsters.[27][28]
See also
- Vapour phase decomposition
- 2019 Philadelphia Energy Solutions refinery explosion
References
- ISBN 9781849733069.
- ISBN 978-1429263092.
- ^ "Hydrofluoric Acid". PubChem. National Institute of Health. Retrieved October 12, 2017.
- ^ ISBN 978-3527306732.
- ^ a b c d "Hydrogen Fluoride/Hydrofluoric Acid: Systemic Agent". Emergency Response Safety and Health Database. NIOSH - CDC. May 12, 2011. Archived from the original on Dec 7, 2015. Retrieved 2015-12-04.
- OCLC 68207297.
- ^ Strachan, John (January 1999). "A deadly rinse: The dangers of hydrofluoric acid". Professional Carwashing & Detailing. 23 (1). Archived from the original on April 25, 2012.
- .
- ISBN 978-0-08-022057-4.
- ^ ISBN 978-0-13-149330-8. Retrieved 22 August 2011.
- ^ .
- ^ PMID 18386892.
- ^ a b Mootz, D. (1981). "Crystallochemical Correlate to the Anomaly of Hydrofluoric Acid". Angew. Chem. Int. Ed. Engl. 20 (123): 791. .
- .
- ^ ISSN 0002-7863.
- ^ OCLC 22861992.
- ^ OCLC 16580057.
- ISBN 978-0-12-352651-9.
- ^ Clark, Jim (2002). "The acidity of the hydrogen halides". Retrieved 4 September 2011.
- ^ a b Chambers, C.; Holliday, A. K. (1975). Modern inorganic chemistry (An intermediate text) (PDF). The Butterworth Group. pp. 328–329. Archived from the original (PDF) on 2013-03-23.
- ISBN 9780070703360.
- ^ C. E. Housecroft and A. G. Sharpe "Inorganic Chemistry" (Pearson Prentice Hall, 2nd ed. 2005), p. 170.
- ^ Cotton & Wilkinson (1988), p. 104
- ^ S2CID 45595073.
- ^ "NIOSH Pocket Guide to Chemical Hazards – Hydrogen fluoride". CDC. Retrieved 2015-11-28.
- ^ "Hydrofluoric Acid Fact Sheet" (PDF). Department of Environmental Safety, Sustainability & Risk. University of Maryland. Retrieved 11 March 2024.
the HF molecule can cause deep tissue damage, including destruction of the bone. ... when fluoride ions bind to calcium and magnesium
- ^ "How much of the science in Breaking Bad is real?". BBC News. 16 August 2013. Archived from the original on 17 August 2023.
- ^ Hare, Jonathan (1 May 2011). "Breaking Bad II – acid bath disposal of bodies". education in chemistry. Royal Society of Chemistry. Archived from the original on 10 June 2023.
External links
- International Chemical Safety Card 0283
- NIOSH Pocket Guide to Chemical Hazards
- CID 14917 from PubChem (HF)
- CID 144681 from PubChem (5HF)
- CID 141165 from PubChem (6HF)
- CID 144682 from PubChem (7HF)
- "Hydrofluoric Acid Burn", The New England Journal of Medicine—Acid burn case study