Fluorite
| Fluorite | ||
|---|---|---|
Specific gravity 3.175–3.184; to 3.56 if high in rare-earth elements | | |
| Optical properties | Isotropic; weak anomalous anisotropism; moderate relief | |
| Refractive index | 1.433–1.448 | |
| Fusibility | 3 | |
| Solubility | slightly water soluble and in hot hydrochloric acid | |
| Other characteristics | May be fluorescent, phosphorescent, thermoluminescent, and/or triboluminescent | |
| References | [2][3][4][5] | |
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, so fluorite is useful in making apochromatic lenses, and particularly valuable in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density.
History and etymology
The word fluorite is derived from the
In 1852, fluorite gave its name to the phenomenon of fluorescence, which is prominent in fluorites from certain locations, due to certain impurities in the crystal. Fluorite also gave the name to its constitutive element fluorine.[3] Currently, the word "fluorspar" is most commonly used for fluorite as an industrial and chemical commodity, while "fluorite" is used mineralogically and in most other senses.
In archeology, gemmology, classical studies, and Egyptology, the Latin terms murrina and myrrhina refer to fluorite.
Structure
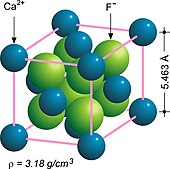
Fluorite
Occurrence and mining

Fluorite forms as a late-crystallizing mineral in
It is a common mineral mainly distributed in South Africa, China, Mexico, Mongolia, the United Kingdom, the United States, Canada, Tanzania, Rwanda and Argentina.
The world reserves of fluorite are estimated at 230 million tonnes (Mt) with the largest deposits being in South Africa (about 41 Mt), Mexico (32 Mt) and China (24 Mt). China is leading the world production with about 3 Mt annually (in 2010), followed by Mexico (1.0 Mt), Mongolia (0.45 Mt), Russia (0.22 Mt), South Africa (0.13 Mt), Spain (0.12 Mt) and Namibia (0.11 Mt).[16][needs update]
One of the largest deposits of fluorspar in North America is located on the Burin Peninsula, Newfoundland, Canada. The first official recognition of fluorspar in the area was recorded by geologist J.B. Jukes in 1843. He noted an occurrence of "galena" or lead ore and fluoride of lime on the west side of St. Lawrence harbour. It is recorded that interest in the commercial mining of fluorspar began in 1928 with the first ore being extracted in 1933. Eventually, at Iron Springs Mine, the shafts reached depths of 970 feet (300 m). In the St. Lawrence area, the veins are persistent for great lengths and several of them have wide lenses. The area with veins of known workable size comprises about 60 square miles (160 km2).[17][18][19]
In 2018, Canada Fluorspar Inc. commenced mine production again[20] in St. Lawrence; in spring 2019, the company was planned to develop a new shipping port on the west side of Burin Peninsula as a more affordable means of moving their product to markets,[21] and they successfully sent the first shipload of ore from the new port on July 31, 2021. This marks the first time in 30 years that ore has been shipped directly out of St. Lawrence.[22]
Cubic crystals up to 20 cm across have been found at Dalnegorsk, Russia.[23] The largest documented single crystal of fluorite was a cube 2.12 meters in size and weighing approximately 16 tonnes.[24]

In Asturias (Spain) there are several fluorite deposits known internationally for the quality of the specimens they have yielded. In the area of Berbes, Ribadesella, fluorite appears as cubic crystals, sometimes with dodecahedron modifications, which can reach a size of up to 10 cm of edge, with internal colour zoning, almost always violet in colour. It is associated with quartz and leafy aggregates of baryte. In the Emilio mine, in Loroñe, Colunga, the fluorite crystals, cubes with small modifications of other figures, are colourless and transparent. They can reach 10 cm of edge. In the Moscona mine, in Villabona, the fluorite crystals, cubic without modifications of other shapes, are yellow, up to 3 cm of edge. They are associated with large crystals of calcite and barite.[25]
"Blue John"
One of the most famous of the older-known localities of fluorite is Castleton in Derbyshire, England, where, under the name of "Derbyshire Blue John", purple-blue fluorite was extracted from several mines or caves. During the 19th century, this attractive fluorite was mined for its ornamental value. The mineral Blue John is now scarce, and only a few hundred kilograms are mined each year for ornamental and lapidary use. Mining still takes place in Blue John Cavern and Treak Cliff Cavern.[26]
Recently discovered deposits in China have produced fluorite with coloring and banding similar to the classic Blue John stone.[27]
Fluorescence

Many samples of fluorite exhibit
One fluorescent variety of fluorite is chlorophane, which is reddish or purple in color and fluoresces brightly in emerald green when heated (thermoluminescence), or when illuminated with ultraviolet light.
The color of visible light emitted when a sample of fluorite is fluorescing depends on where the original specimen was collected; different impurities having been included in the crystal lattice in different places. Neither does all fluorite fluoresce equally brightly, even from the same locality. Therefore, ultraviolet light is not a reliable tool for the identification of specimens, nor for quantifying the mineral in mixtures. For example, among British fluorites, those from Northumberland, County Durham, and eastern Cumbria are the most consistently fluorescent, whereas fluorite from Yorkshire, Derbyshire, and Cornwall, if they fluoresce at all, are generally only feebly fluorescent.
Fluorite also exhibits the property of thermoluminescence.[32]
Color
Fluorite is allochromatic, meaning that it can be tinted with elemental impurities. Fluorite comes in a wide range of colors and has consequently been dubbed "the most colorful mineral in the world". Every color of the rainbow in various shades is represented by fluorite samples, along with white, black, and clear crystals. The most common colors are purple, blue, green, yellow, or colorless. Less common are pink, red, white, brown, and black. Color zoning or banding is commonly present. The color of the fluorite is determined by factors including impurities, exposure to radiation, and the absence of voids of the color centers.
-
Pastel green fluorite crystal on galena
-
A golden yellow with hints of purple fluorite
-
Freestanding purple fluorite cluster between two quartzes
-
Light to dark burgundy color fluorite
-
Transparent teal color fluorite with purple highlights
-
Grass-green fluorite octahedrons clustered on a quartz-rich matrix
Uses
Source of fluorine and fluoride
Fluorite is a major source of hydrogen fluoride, a commodity chemical used to produce a wide range of materials. Hydrogen fluoride is liberated from the mineral by the action of concentrated sulfuric acid:
The resulting HF is converted into fluorine, fluorocarbons, and diverse fluoride materials. As of the late 1990s, five billion kilograms were mined annually.[33]
There are three principal types of industrial use for natural fluorite, commonly referred to as "fluorspar" in these industries, corresponding to different grades of purity. Metallurgical grade fluorite (60–85% CaF2), the lowest of the three grades, has traditionally been used as a flux to lower the melting point of raw materials in steel production to aid the removal of impurities, and later in the production of aluminium. Ceramic grade fluorite (85–95% CaF2) is used in the manufacture of opalescent glass, enamels, and cooking utensils. The highest grade, "acid grade fluorite" (97% or more CaF2), accounts for about 95% of fluorite consumption in the US where it is used to make hydrogen fluoride and hydrofluoric acid by reacting the fluorite with sulfuric acid.[34]
Internationally, acid-grade fluorite is also used in the production of AlF3 and
Niche uses

Lapidary uses
Natural fluorite mineral has ornamental and lapidary uses. Fluorite may be drilled into beads and used in jewelry, although due to its relative softness it is not widely used as a semiprecious stone. It is also used for ornamental carvings, with expert carvings taking advantage of the stone's zonation.
Optics
In the laboratory, calcium fluoride is commonly used as a window material for both
Fluorite should not be confused with fluoro-crown (or fluorine crown) glass, a type of low-dispersion glass that has special optical properties approaching fluorite. True fluorite is not a glass but a crystalline material. Lenses or optical groups made using this low dispersion glass as one or more elements exhibit less chromatic aberration than those utilizing conventional, less expensive crown glass and flint glass elements to make an achromatic lens. Optical groups employ a combination of different types of glass; each type of glass refracts light in a different way. By using combinations of different types of glass, lens manufacturers are able to cancel out or significantly reduce unwanted characteristics; chromatic aberration being the most important. The best of such lens designs are often called apochromatic (see above). Fluoro-crown glass (such as Schott FK51) usually in combination with an appropriate "flint" glass (such as Schott KzFSN 2) can give very high performance in telescope objective lenses, as well as microscope objectives, and camera telephoto lenses. Fluorite elements are similarly paired with complementary "flint" elements (such as Schott LaK 10).[36] The refractive qualities of fluorite and of certain flint elements provide a lower and more uniform dispersion across the spectrum of visible light, thereby keeping colors focused more closely together. Lenses made with fluorite are superior to fluoro-crown based lenses, at least for doublet telescope objectives; but are more difficult to produce and more costly.[37]
The use of fluorite for prisms and lenses was studied and promoted by Victor Schumann near the end of the 19th century.[38] Naturally occurring fluorite crystals without optical defects were only large enough to produce microscope objectives.
With the advent of synthetically grown fluorite crystals in the 1950s - 60s, it could be used instead of glass in some high-performance optical telescope and camera lens elements. In telescopes, fluorite elements allow high-resolution images of astronomical objects at high magnifications. Canon Inc. produces synthetic fluorite crystals that are used in their better telephoto lenses. The use of fluorite for telescope lenses has declined since the 1990s, as newer designs using fluoro-crown glass, including triplets, have offered comparable performance at lower prices. Fluorite and various combinations of fluoride compounds can be made into synthetic crystals which have applications in lasers and special optics for UV and infrared.[39]
Exposure tools for the
Source of fluorine gas in nature
In 2012, the first source of naturally occurring fluorine gas was found in fluorite mines in Bavaria, Germany. It was previously thought that
Gallery
-
Fluorite crystals on display at the Cullen Hall of Gems and Minerals, Houston Museum of Natural Science
-
Fluorite and sphalerite, from Elmwood mine, Smith county, Tennessee, US
-
Translucent ball of botryoidal fluorite perched on a calcite crystal
-
Fluorite with baryte, from Berbes Mine, Berbes Mining area, Ribadesella, Asturias, Spain
-
Fluorite from Diana Maria mine, Weardale, England, UK
-
Fluorite from El Hammam Mine, Meknès Prefecture, Meknès-Tafilalet Region, Morocco
-
Toad carved in fluorite. Length 8 cm (3 in).
See also
- List of countries by fluorite production
- List of minerals
- Magnesium fluoride – also used in UV optics
References
![]() This article incorporates public domain material from Fluorspar (PDF). United States Geological Survey.
This article incorporates public domain material from Fluorspar (PDF). United States Geological Survey.
- S2CID 235729616.
- ISBN 0962209724. Archived(PDF) from the original on 2006-09-06. Retrieved December 5, 2011.
- ^ a b Fluorite. Mindat.org
- ^ Fluorite. Webmineral.com
- ^ ISBN 0-471-80580-7
- . Retrieved 19 January 2022.
- ^ "Discovery of fluorine". Fluoride History.
- ISBN 978-0-444-52239-9.
- Online Etymology Dictionary.
- Online Etymology Dictionary.
- ^ James Harrell 2012. UCLA Encyclopedia of Egyptology, Gemstones.
- ISSN 0974-6889.
- ISBN 978-0-08-037941-8.
- ISBN 9780195106916.
- ^ OCLC 858884283.
- ^ Fluorspar. USGS.gov (2011)
- ^ Reactivation of the St. Lawrence fluorspar mine at St. Lawrence, NL. Burin Minerals Ltd. (April 9, 2009).
- .
- .
- ^ "St. Lawrence fluorspar mine gets $5M from feds, hundreds of jobs touted". Cbc.ca. Retrieved 14 December 2021.
- ^ Farrell, Colin. "CFI seeking new location for shipping port in St. Lawrence, NL | SaltWire". Saltwire.com. Retrieved 14 December 2021.
- ^ Sheppard, Noah. "First Shipment of Fluorspar in Over 30 Years Exported From St. Lawrence". Vocm.com. Retrieved 14 December 2021.
- ISBN 0785815201.
- ^ Rickwood, P. C. (1981). "The largest crystals" (PDF). American Mineralogist. 66: 885–907. Archived (PDF) from the original on 2009-06-20.
- ^ Calvo Sevillano, Guiomar; Calvo Rebollar, Miguel (2006). "Fluorite from Spain. Every color under the Sun". Fluorite. The Collector's Choice. Connecticut, USA: Lithographie LLC. Connecticut, USA. pp. 38–42.
- ISBN 0174482760.
- .
- ^ .
- S2CID 186207789.
- S2CID 4104586.
- S2CID 227176307.
- ISBN 0-521-36811-1.
- ISBN 3527306730.
- ^ a b Miller, M. Michael. Fluorspar, USGS 2009 Minerals Yearbook
- ^ "The Crawford Cup". British Museum. Retrieved 20 December 2014.
- ^ "Interactive Abbe Diagram". SCHOTT AG. 2019. Retrieved February 20, 2018.
- ^ Rutten, Harrie; van Venrooij, Martin (1988). Telescope Optics Evaluation and Design. Willmann-Bell, Inc.
- doi:10.1086/142050.
- ISBN 0-470-85142-2.
- ISBN 0-521-77096-3.
- ISBN 0-240-51323-1.
- ^ First direct evidence that elemental fluorine occurs in nature. Labspaces.net (2012-07-06). Retrieved on 2013-08-05.
- ^ Withers, Neil (1 July 2012) Fluorine finally found in nature |Chemistry World. Rsc.org.













