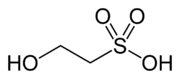
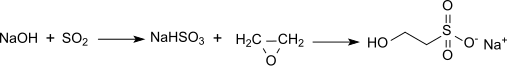Isethionic acid
| |
 Isethionic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Hydroxyethane-1-sulfonic acid | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.003.169 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H6O4S | |
| Molar mass | 126.13 g/mol |
| Density | 1.63 g/cm3 |
| Acidity (pKa) | 1.39 (predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isethionic acid is an
Synthesis
The original synthesis of the compound, involving the reaction of sulfur trioxide with ethanol, has largely been replaced by more advanced methods. An alternative production method involves the hydrolysis of carbyl sulfate, which is derived from the sulfonation of ethylene.
However the most common route is the reaction of ethylene oxide with aqueous sodium bisulfite, which produces the sodium salt (sodium isethionate):
Reactions
Isethionic acid is used as a starting material in the industrial production of taurine.
Dehydration of isethionic acid gives vinylsulfonic acid.[2]
Derivatives
Isethionic acid is also used as a
Biological importance
Studies made on dog heart slices suggested that heart tissue may be capable of converting taurine to isethionic acid, further experiments demonstrated that this tissue may synthesize taurine from cystine.[6]