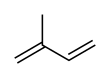
Isoprene
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Isoprene
| |||
| Preferred IUPAC name
2-Methylbuta-1,3-diene | |||
| Other names
2-Methyl-1,3-butadiene
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.001.040 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H8 | |||
| Molar mass | 68.12 g/mol | ||
| Density | 0.681 g/cm3 | ||
| Melting point | −143.95 °C (−227.11 °F; 129.20 K) | ||
| Boiling point | 34.067 °C (93.321 °F; 307.217 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Isoprene, or 2-methyl-1,3-butadiene, is a common volatile organic compound with the formula CH2=C(CH3)−CH=CH2. In its pure form it is a colorless volatile liquid. It is produced by many plants and animals[1] (including humans) and its polymers are the main component of natural rubber. C. G. Williams named the compound in 1860 after obtaining it from the pyrolysis of natural rubber; he correctly deduced the empirical formula C5H8.[2][3]
Natural occurrences
Isoprene is produced and emitted by many species of trees (major producers are
Plants
Isoprene is made through the methyl-erythritol 4-phosphate pathway (MEP pathway, also called the non-mevalonate pathway) in the chloroplasts of plants. One of the two end-products of MEP pathway, dimethylallyl pyrophosphate (DMAPP), is cleaved by the enzyme isoprene synthase to form isoprene and diphosphate. Therefore, inhibitors that block the MEP pathway, such as fosmidomycin, also block isoprene formation. Isoprene emission increases dramatically with temperature and maximizes at around 40 °C. This has led to the hypothesis that isoprene may protect plants against heat stress (thermotolerance hypothesis, see below). Emission of isoprene is also observed in some bacteria and this is thought to come from non-enzymatic degradations from DMAPP. Global emission of isoprene by plants is estimated at 350 million tons per year.[6]
Regulation
Isoprene emission in plants is controlled both by the availability of the substrate (DMAPP) and by enzyme (isoprene synthase) activity. In particular, light, CO2 and O2 dependencies of isoprene emission are controlled by substrate availability, whereas temperature dependency of isoprene emission is regulated both by substrate level and enzyme activity.
Other organisms
Isoprene is the most abundant hydrocarbon measurable in the breath of humans.[7][8][9] The estimated production rate of isoprene in the human body is 0.15 µmol/(kg·h), equivalent to approximately 17 mg/day for a person weighing 70 kg. Isoprene is common in low concentrations in many foods. Many species of soil and marine bacteria, such as Actinomycetota, are capable of degrading isoprene and using it as a fuel source.

Biological roles
Isoprene emission appears to be a mechanism that trees use to combat abiotic stresses.[10] In particular, isoprene has been shown to protect against moderate heat stress (around 40 °C). It may also protect plants against large fluctuations in leaf temperature. Isoprene is incorporated into and helps stabilize cell membranes in response to heat stress.
Isoprene also confers resistance to reactive oxygen species.[11] The amount of isoprene released from isoprene-emitting vegetation depends on leaf mass, leaf area, light (particularly photosynthetic photon flux density, or PPFD) and leaf temperature. Thus, during the night, little isoprene is emitted from tree leaves, whereas daytime emissions are expected to be substantial during hot and sunny days, up to 25 μg/(g dry-leaf-weight)/hour in many oak species.[12]
Isoprenoids
The isoprene skeleton can be found in naturally occurring compounds called
Examples of isoprenoids include
Similarly,

Impact on aerosols
After release, isoprene is converted into various species, such as
While most experts acknowledge that isoprene emission affects aerosol formation, whether isoprene increases or decreases aerosol formation is debated. A second major effect of isoprene on the atmosphere is that in the presence of
As an example the Blue Ridge Mountains are noted for having a bluish color when seen from a distance. Trees put the "blue" in Blue Ridge, from the isoprene released into the atmosphere.[18] This contributes to the characteristic haze on the mountains and their distinctive color.[19] The similarly named, but geologically district, Blue Mountains of New South Wales also derive their color and name from isoprene and isoprenoids released by Eucalyptus trees.[20]
Industrial production
Isoprene is most readily available industrially as a byproduct of the thermal cracking of petroleum naphtha or oil, as a side product in the production of ethylene. About 800,000 metric tons are produced annually. About 95% of isoprene production is used to produce cis-1,4-polyisoprene—a synthetic version of natural rubber.[13]
Natural rubber consists mainly of poly-cis-isoprene with a molecular mass of 100,000 to 1,000,000 g/mol. Typically natural rubber contains a few percent of other materials, such as proteins, fatty acids, resins, and inorganic materials. Some natural rubber sources, called
See also
References
- PMID 8690002.
- S2CID 104233421.
- ISBN 9789401144353.
- .
- PMID 28654185.
- ^ "Isoprene emissions version 2021". emissions.aeronomie.be. Retrieved 2022-09-26.
- PMID 7259787.
- S2CID 10267120.
- PMID 27160439.
- PMID 17921528.
- PMID 19183288.
- .
- ^ ISBN 978-3527306732.
- ^ Organic Carbon Compounds Emitted By Trees Affect Air Quality, ScienceDaily, Aug. 7, 2009
- ^ IUPAC Subcommittee on Gas Kinetic Data Evaluation – Data Sheet Ox_VOC7, 2007
- ^ A source of haze, ScienceNews, August 6, 2009
- ISSN 1680-7316.
- ISBN 978-0-7637-0432-2.
blue mountains chemical terpene.
- ^ "Blue Ridge Parkway, Frequently Asked Questions". National Park Service. 2007. Archived from the original on December 28, 2007. Retrieved December 29, 2007.
- ^ CSIRO. "Beating the eucalypt blues – new ways to model air quality". www.csiro.au. Retrieved 2023-12-10.
Further reading
- Budavari S, O'Neil MJ, Smith A, Heckelaman PE, eds. (1989). The Merck Index (11th ed.). Rahway NJ. USA: Merck & Co Inc. ISBN 978-0-911910-28-5.
- Bekkedahl N, Wood LA, Wojciechowski M (1936). "Some physical properties of isoprene". Journal of Research of the National Bureau of Standards. 17 (6): 883. .
- Poisson N, Kanakidou M, Crutzen PJ (2000). "Impact of Non-Methane Hydrocarbons on Tropospheric Chemistry and the Oxidizing Power of the Global Troposphere: 3-Dimensional Modelling Results". Journal of Atmospheric Chemistry. 36 (2): 157–230. S2CID 94217044.
- Claeys M, Graham B, Vas G, Wang W, Vermeylen R, Pashynska V, et al. (February 2004). "Formation of secondary organic aerosols through photooxidation of isoprene". Science. 303 (5661): 1173–6. S2CID 19268599.
- Pier PA, McDuffie C (1997). "Seasonal isoprene emission rates and model comparisons using whole-tree emissions from white oak". Journal of Geophysical Research: Atmospheres. 102 (D20): 23963–23971. .
- Pöschl U, Von Kuhlmann R, Poisson N, Crutzen PJ (2000). "Development and Intercomparison of Condensed Isoprene Oxidation Mechanisms for Global Atmospheric Modeling". Journal of Atmospheric Chemistry. 37 (1): 29–52. S2CID 93419825.
- Monson RK, Holland EA (2001). "Biospheric Trace Gas Fluxes and Their Control over Tropospheric Chemistry". Annual Review of Ecology and Systematics. 32: 547–576. .