Leidenfrost effect


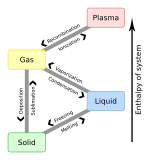
The Leidenfrost effect is a physical phenomenon in which a liquid, close to a solid surface of another body that is significantly hotter than the liquid's boiling point, produces an insulating vapor layer that keeps the liquid from boiling rapidly. Because of this repulsive force, a droplet hovers over the surface, rather than making physical contact with it. The effect is named after the German doctor Johann Gottlob Leidenfrost, who described it in A Tract About Some Qualities of Common Water.
This is most commonly seen when cooking, when drops of water are sprinkled onto a hot pan. If the pan's temperature is at or above the Leidenfrost point, which is approximately 193 °C (379 °F) for water, the water skitters across the pan and takes longer to evaporate than it would take if the water droplets had been sprinkled onto a cooler pan.
Details
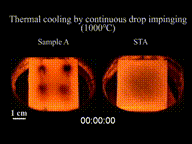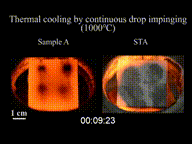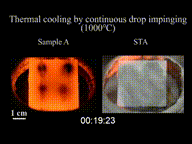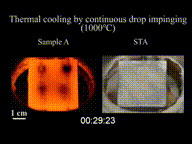
The effect can be seen as drops of water are sprinkled onto a pan at various times as it heats up. Initially, as the temperature of the pan is just below 100 °C (212 °F), the water flattens out and slowly evaporates, or if the temperature of the pan is well below 100 °C (212 °F), the water stays liquid. As the temperature of the pan rises above 100 °C (212 °F), the water droplets hiss when touching the pan, and these droplets evaporate quickly. When the temperature exceeds the Leidenfrost point, the Leidenfrost effect appears. On contact with the pan, the water droplets bunch up into small balls of water and skitter around, lasting much longer than when the temperature of the pan was lower. This effect works until a much higher temperature causes any further drops of water to evaporate too quickly to cause this effect.
The effect happens because, at temperatures at or above the Leidenfrost point, the bottom part of the water droplet vaporizes immediately on contact with the hot pan. The resulting gas suspends the rest of the water droplet just above it, preventing any further direct contact between the liquid water and the hot pan. As steam has much poorer

The temperature at which the Leidenfrost effect appears is difficult to predict. Even if the volume of the drop of liquid stays the same, the Leidenfrost point may be quite different, with a complicated dependence on the properties of the surface, as well as any impurities in the liquid. Some research has been conducted into a theoretical model of the system, but it is quite complicated.[1]
The effect was also described by the Victorian steam boiler designer,
The Leidenfrost point may also be taken to be the temperature for which the hovering droplet lasts longest.[3]
It has been demonstrated that it is possible to stabilize the Leidenfrost vapor layer of water by exploiting
Droplets of different liquids with different boiling temperatures will also exhibit a Leidenfrost effect with respect to each other and repel each other.[5]
The Leidenfrost effect has been used for the development of high sensitivity ambient mass spectrometry. Under the influence of the Leidenfrost condition, the levitating droplet does not release molecules, and the molecules are enriched inside the droplet. At the last moment of droplet evaporation, all the enriched molecules release in a short time period and thereby increase the sensitivity.[6]
A heat engine based on the Leidenfrost effect has been prototyped; it has the advantage of extremely low friction.[7]
The effect also applies when the surface is at room temperature but the liquid is
Leidenfrost point

The Leidenfrost point signifies the onset of stable film boiling. It represents the point on the boiling curve where the heat flux is at the minimum and the surface is completely covered by a vapor blanket. Heat transfer from the surface to the liquid occurs by conduction and radiation through the vapour. In 1756, Leidenfrost observed that water droplets supported by the vapor film slowly evaporate as they move about on the hot surface. As the surface temperature is increased, radiation through the vapor film becomes more significant and the heat flux increases with increasing excess temperature.
The minimum heat flux for a large horizontal plate can be derived from Zuber's equation,[3]
where the properties are evaluated at saturation temperature. Zuber's constant, , is approximately 0.09 for most fluids at moderate pressures.
Heat transfer correlations
The heat transfer coefficient may be approximated using Bromley's equation,[3]
where is the outside diameter of the tube. The correlation constant C is 0.62 for horizontal cylinders and vertical plates, and 0.67 for spheres. Vapor properties are evaluated at film temperature.
For stable film boiling on a horizontal surface, Berenson has modified Bromley's equation to yield,[10]
For vertical tubes, Hsu and Westwater have correlated the following equation,[10]
where m is the mass flow rate in at the upper end of the tube.
At excess temperatures above that at the minimum heat flux, the contribution of radiation becomes appreciable, and it becomes dominant at high excess temperatures. The total heat transfer coefficient is thus a combination of the two. Bromley has suggested the following equations for film boiling from the outer surface of horizontal tubes:
If ,
The effective radiation coefficient, can be expressed as,
where is the emissivity of the solid and is the Stefan–Boltzmann constant.
Pressure field in a Leidenfrost droplet
The equation for the pressure field in the vapor region between the droplet and the solid surface can be solved for using the standard momentum and
Leidenfrost temperature and surface tension effects
The Leidenfrost temperature is the property of a given set of solid–liquid pair. The temperature of the solid surface beyond which the liquid undergoes the Leidenfrost phenomenon is termed the Leidenfrost temperature. Calculation of the Leidenfrost temperature involves the calculation of the minimum film boiling temperature of a fluid. Berenson[12] obtained a relation for the minimum film boiling temperature from minimum heat flux arguments. While the equation for the minimum film boiling temperature, which can be found in the reference above, is quite complex, the features of it can be understood from a physical perspective. One critical parameter to consider is the surface tension. The proportional relationship between the minimum film boiling temperature and surface tension is to be expected, since fluids with higher surface tension need higher quantities of heat flux for the onset of nucleate boiling. Since film boiling occurs after nucleate boiling, the minimum temperature for film boiling should have a proportional dependence on the surface tension.
Henry developed a model for Leidenfrost phenomenon which includes transient wetting and microlayer evaporation.[13] Since the Leidenfrost phenomenon is a special case of film boiling, the Leidenfrost temperature is related to the minimum film boiling temperature via a relation which factors in the properties of the solid being used. While the Leidenfrost temperature is not directly related to the surface tension of the fluid, it is indirectly dependent on it through the film boiling temperature. For fluids with similar thermophysical properties, the one with higher surface tension usually has a higher Leidenfrost temperature.
For example, for a saturated water–copper interface, the Leidenfrost temperature is 257 °C (495 °F). The Leidenfrost temperatures for glycerol and common alcohols are significantly smaller because of their lower surface tension values (density and viscosity differences are also contributing factors.)
Reactive Leidenfrost effect

Non-volatile materials were discovered in 2015 to also exhibit a 'reactive Leidenfrost effect', whereby solid particles were observed to float above hot surfaces and skitter around erratically.
High speed photography of the reactive Leidenfrost effect of cellulose on porous surfaces (macroporous
The Leidenfrost effect has also been used as a means to promote chemical change of various organic liquids through their conversion by thermal decomposition into various products. Examples include decomposition of ethanol,[15] diethyl carbonate,[16] and glycerol.[17]
In popular culture
In Jules Verne's 1876 book Michael Strogoff, the protagonist is saved from being blinded with a hot blade by evaporating tears.[18]
In the 2009 season 7 finale of MythBusters, "Mini Myth Mayhem", the team demonstrated that a person can wet their hand and briefly dip it into molten lead without injury, using the Leidenfrost effect as the scientific basis.[19]
See also
References
- .
- ^ William Fairbairn (1851). Two Lectures: The Construction of Boilers, and on Boiler Explosions, with the means of prevention. Archived from the original on 2017-11-23.[page needed]
- ^ ISBN 0-471-45728-0.
- S2CID 4411432.
- S2CID 235694660.
- Leah Crane (24 November 2021). "Watch droplets bounce off each other as they levitate on a hot plate". New Scientist.
- S2CID 39368022.
- PMID 25731669.
- ^ "Who What Why: How dangerous is liquid nitrogen?". BBC News. 8 October 2012.
- S2CID 21732968.
- ^ ISBN 978-0-470-12868-8.
- S2CID 243345698.[page needed]
- .
- OSTI 4694181.
- ^ PMID 26057818.
- Paul J. Dauenhauer (June 10, 2015). "Scientists levitate wood on structured surfaces captured by high speed photography". Phys.org.
- .
- .
- S2CID 191155524.
- .
- ^ "Mini-Myth Mayhem". MythBusters. Season 7. Episode 136. December 28, 2009. Discovery Channel.
External links
- Essay about the effect and demonstrations by Jearl Walker (PDF)
- Site with high-speed video, pictures and explanation of film-boiling by Heiner Linke at the University of Oregon, USA
- "Scientists make water run uphill" by BBC News about using the Leidenfrost effect for cooling of computer chips.
- "Uphill Water" – ABC Catalyst story
- "Leidenfrost Maze" – University of Bath undergraduate students Carmen Cheng and Matthew Guy
- "When Water Flows Uphill" – Science Friday with Univ. of Bath professor Kei Takashina
- Jeffrey, Colin (March 10, 2015). "Engine running on frozen carbon dioxide may power mission to Mars". Gizmag. Retrieved 10 March 2015.
- Carolyn Embach, ResearchGate: English translation of Johan Gottlob Leidenfrost, De aquae communes nonnullis qualitatibus tractatus, Duisburg on Rhine, 1756. (Carolyn S. E. Wares aka Carolyn Embach, translator, 1964)

![{\displaystyle {{\frac {q}{A}}_{min}}=C{{h}_{fg}}{{\rho }_{v}}{{\left[{\frac {\sigma g\left({{\rho }_{L}}-{{\rho }_{v}}\right)}{{\left({{\rho }_{L}}+{{\rho }_{v}}\right)}^{2}}}\right]}^{{}^{1}\!\!\diagup \!\!{}_{4}\;}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a94fba1cc42308142bf2448820f0cd14a1fdb86d)

![{\displaystyle h=C{{\left[{\frac {k_{v}^{3}{{\rho }_{v}}g\left({{\rho }_{L}}-{{\rho }_{v}}\right)\left({{h}_{fg}}+0.4{{c}_{pv}}\left({{T}_{s}}-{{T}_{sat}}\right)\right)}{{{D}_{o}}{{\mu }_{v}}\left({{T}_{s}}-{{T}_{sat}}\right)}}\right]}^{{}^{1}\!\!\diagup \!\!{}_{4}\;}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3f3d23ccc06ec9190f361d504cd6b982deeea09c)

![{\displaystyle h=0.425{{\left[{\frac {k_{vf}^{3}{{\rho }_{vf}}g\left({{\rho }_{L}}-{{\rho }_{v}}\right)\left({{h}_{fg}}+0.4{{c}_{pv}}\left({{T}_{s}}-{{T}_{sat}}\right)\right)}{{{\mu }_{vf}}\left({{T}_{s}}-{{T}_{sat}}\right){\sqrt {\sigma /g\left({{\rho }_{L}}-{{\rho }_{v}}\right)}}}}\right]}^{{}^{1}\!\!\diagup \!\!{}_{4}\;}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f0c9bf294fc90d5b0b54986a8bd1b4684966917b)
![{\displaystyle h{{\left[{\frac {\mu _{v}^{2}}{g{{\rho }_{v}}\left({{\rho }_{L}}-{{\rho }_{v}}\right)k_{v}^{3}}}\right]}^{{}^{1}\!\!\diagup \!\!{}_{3}\;}}=0.0020{{\left[{\frac {4m}{\pi {{D}_{v}}{{\mu }_{v}}}}\right]}^{0.6}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/315bcfe771ba9265972a7431972f7de1ab2fd75b)