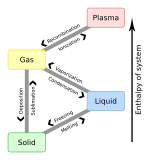
Liquid

| Part of a series on |
| Continuum mechanics |
|---|
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a nearly constant volume independent of pressure. It is one of the four fundamental states of matter (the others being solid, gas, and plasma), and is the only state with a definite volume but no fixed shape.
The density of a liquid is usually close to that of a solid, and much higher than that of a gas. Therefore, liquid and solid are both termed condensed matter. On the other hand, as liquids and gases share the ability to flow, they are both called fluids.
A liquid is made up of tiny vibrating particles of matter, such as atoms, held together by
Although liquid water is abundant on Earth, this state of matter is actually the least common in the known universe, because liquids require a relatively narrow temperature/pressure range to exist. Most known matter in the universe is either gas (as interstellar clouds) or plasma (as stars).
Introduction
Liquid is one of the four primary states of matter, with the others being solid, gas and plasma. A liquid is a fluid. Unlike a solid, the molecules in a liquid have a much greater freedom to move. The forces that bind the molecules together in a solid are only temporary in a liquid, allowing a liquid to flow while a solid remains rigid.
A liquid, like a gas, displays the properties of a fluid. A liquid can flow, assume the shape of a container, and, if placed in a sealed container, will distribute applied pressure evenly to every surface in the container. If liquid is placed in a bag, it can be squeezed into any shape. Unlike a gas, a liquid is nearly incompressible, meaning that it occupies nearly a constant volume over a wide range of pressures; it does not generally expand to fill available space in a container but forms its own surface, and it may not always mix readily with another liquid. These properties make a liquid suitable for applications such as hydraulics.
Liquid particles are bound firmly but not rigidly. They are able to move around one another freely, resulting in a limited degree of particle mobility. As the temperature increases, the increased vibrations of the molecules causes distances between the molecules to increase. When a liquid reaches its boiling point, the cohesive forces that bind the molecules closely together break, and the liquid changes to its gaseous state (unless superheating occurs). If the temperature is decreased, the distances between the molecules become smaller. When the liquid reaches its freezing point the molecules will usually lock into a very specific order, called crystallizing, and the bonds between them become more rigid, changing the liquid into its solid state (unless supercooling occurs).
Examples
Only two
Pure substances that are liquid under normal conditions include water, ethanol and many other organic solvents. Liquid water is of vital importance in chemistry and biology, and it is necessary for all known forms of life.[4][5]
Inorganic liquids include water, magma, inorganic nonaqueous solvents and many acids.
Important everyday liquids include aqueous solutions like household bleach, other mixtures of different substances such as mineral oil and gasoline, emulsions like vinaigrette or mayonnaise, suspensions like blood, and colloids like paint and milk.
Many gases can be
Some materials cannot be classified within the classical three states of matter. For example, liquid crystals (used in liquid-crystal displays) possess both solid-like and liquid-like properties, and belong to their own state of matter distinct from either liquid or solid.[7]
Applications

Lubrication
Liquids are useful as
Solvation
Many liquids are used as solvents, to dissolve other liquids or solids. Solutions are found in a wide variety of applications, including paints, sealants, and adhesives. Naphtha and acetone are used frequently in industry to clean oil, grease, and tar from parts and machinery. Body fluids are water-based solutions.
Surfactants are commonly found in soaps and detergents. Solvents like alcohol are often used as antimicrobials. They are found in cosmetics, inks, and liquid dye lasers. They are used in the food industry, in processes such as the extraction of vegetable oil.[9]
Cooling
Liquids tend to have better
Cooking
Liquids are often used in
Distillation
Since liquids often have different boiling points, mixtures or solutions of liquids or gases can typically be separated by distillation, using heat, cold, vacuum, pressure, or other means. Distillation can be found in everything from the production of alcoholic beverages, to oil refineries, to the cryogenic distillation of gases such as argon, oxygen, nitrogen, neon, or xenon by liquefaction (cooling them below their individual boiling points).[15]
Hydraulics
Liquid is the primary component of
Liquid metals
Liquid metals have several properties that are useful in
Miscellaneous
Liquids are sometimes used in measuring devices. A
The free surface of a rotating liquid forms a circular paraboloid and can therefore be used as a telescope. These are known as liquid-mirror telescopes.[21] They are significantly cheaper than conventional telescopes,[22] but can only point straight upward (zenith telescope). A common choice for the liquid is mercury.
Mechanical properties
Volume

Quantities of liquids are measured in units of volume. These include the SI unit cubic metre (m3) and its divisions, in particular the cubic decimeter, more commonly called the litre (1 dm3 = 1 L = 0.001 m3), and the cubic centimetre, also called millilitre (1 cm3 = 1 mL = 0.001 L = 10−6 m3).[23]
The volume of a quantity of liquid is fixed by its temperature and pressure. Liquids generally expand when heated, and contract when cooled. Water between 0 °C and 4 °C is a notable exception.[24]
On the other hand, liquids have little
However, the negligible compressibility does lead to other phenomena. The banging of pipes, called water hammer, occurs when a valve is suddenly closed, creating a huge pressure-spike at the valve that travels backward through the system at just under the speed of sound. Another phenomenon caused by liquid's incompressibility is cavitation. Because liquids have little elasticity they can literally be pulled apart in areas of high turbulence or dramatic change in direction, such as the trailing edge of a boat propeller or a sharp corner in a pipe. A liquid in an area of low pressure (vacuum) vaporizes and forms bubbles, which then collapse as they enter high pressure areas. This causes liquid to fill the cavities left by the bubbles with tremendous localized force, eroding any adjacent solid surface.[28]
Pressure and buoyancy
In a gravitational field, liquids exert pressure on the sides of a container as well as on anything within the liquid itself. This pressure is transmitted in all directions and increases with depth. If a liquid is at rest in a uniform gravitational field, the pressure at depth is given by[29]
where:
- is the pressure at the surface
- is the density of the liquid, assumed uniform with depth
- is the gravitational acceleration
For a body of water open to the air, would be the atmospheric pressure.
Static liquids in uniform gravitational fields also exhibit the phenomenon of buoyancy, where objects immersed in the liquid experience a net force due to the pressure variation with depth. The magnitude of the force is equal to the weight of the liquid displaced by the object, and the direction of the force depends on the average density of the immersed object. If the density is smaller than that of the liquid, the buoyant force points upward and the object floats, whereas if the density is larger, the buoyant force points downward and the object sinks. This is known as Archimedes' principle.[30]
Surfaces

Unless the volume of a liquid exactly matches the volume of its container, one or more surfaces are observed. The presence of a surface introduces new phenomena which are not present in a bulk liquid. This is because a molecule at a surface possesses bonds with other liquid molecules only on the inner side of the surface, which implies a net force pulling surface molecules inward. Equivalently, this force can be described in terms of energy: there is a fixed amount of energy associated with forming a surface of a given area. This quantity is a material property called the
A practical implication of surface tension is that liquids tend to minimize their surface area, forming spherical drops and bubbles unless other constraints are present. Surface tension is responsible for a range of other phenomena as well, including surface waves, capillary action, wetting, and ripples. In liquids under nanoscale confinement, surface effects can play a dominating role since – compared with a macroscopic sample of liquid – a much greater fraction of molecules are located near a surface.
The surface tension of a liquid directly affects its
The surface tensions of common liquids occupy a relatively narrow range of values when exposed to changing conditions such as temperature, which contrasts strongly with the enormous variation seen in other mechanical properties, such as viscosity.[32]
The
Flow

An important physical property characterizing the flow of liquids is viscosity. Intuitively, viscosity describes the resistance of a liquid to flow.
More technically, viscosity measures the resistance of a liquid to deformation at a given rate, such as when it is being sheared at finite velocity.[33] A specific example is a liquid flowing through a pipe: in this case the liquid undergoes shear deformation since it flows more slowly near the walls of the pipe than near the center. As a result, it exhibits viscous resistance to flow. In order to maintain flow, an external force must be applied, such as a pressure difference between the ends of the pipe.
The viscosity of liquids decreases with increasing temperature.[34]
Precise control of viscosity is important in many applications, particularly the lubrication industry. One way to achieve such control is by blending two or more liquids of differing viscosities in precise ratios.[35] In addition, various additives exist which can modulate the temperature-dependence of the viscosity of lubricating oils. This capability is important since machinery often operate over a range of temperatures (see also viscosity index).[36]
The viscous behavior of a liquid can be either
Sound propagation
The speed of sound in a liquid is given by where is the bulk modulus of the liquid and the density. As an example, water has a bulk modulus of about 2.2 GPa and a density of 1000 kg/m3, which gives c = 1.5 km/s.[38]
Thermodynamics
Phase transitions
At a temperature below the boiling point, any matter in liquid form will evaporate until reaching equilibrium with the reverse process of condensation of its vapor. At this point the vapor will condense at the same rate as the liquid evaporates. Thus, a liquid cannot exist permanently if the evaporated liquid is continually removed.[39] A liquid at or above its boiling point will normally boil, though superheating can prevent this in certain circumstances.
At a temperature below the freezing point, a liquid will tend to crystallize, changing to its solid form. Unlike the transition to gas, there is no equilibrium at this transition under constant pressure,[citation needed] so unless supercooling occurs, the liquid will eventually completely crystallize. However, this is only true under constant pressure, so that (for example) water and ice in a closed, strong container might reach an equilibrium where both phases coexist. For the opposite transition from solid to liquid, see melting.
Liquids in space
The phase diagram explains why liquids do not exist in space or any other vacuum. Since the pressure is essentially zero (except on surfaces or interiors of planets and moons) water and other liquids exposed to space will either immediately boil or freeze depending on the temperature. In regions of space near the Earth, water will freeze if the sun is not shining directly on it and vaporize (sublime) as soon as it is in sunlight. If water exists as ice on the Moon, it can only exist in shadowed holes where the sun never shines and where the surrounding rock does not heat it up too much. At some point near the orbit of Saturn, the light from the Sun is too faint to sublime ice to water vapor. This is evident from the longevity of the ice that composes Saturn's rings.[40]
Solutions
Liquids can form solutions with gases, solids, and other liquids.
Two liquids are said to be
Microscopic description
The microscopic structure of liquids is complex and historically has been the subject of intense research and debate.[43][44][45][46] A few of the key ideas are explained below.
General description
Microscopically, liquids consist of a dense, disordered packing of molecules. This contrasts with the other two common phases of matter, gases and solids. Although gases are disordered, the molecules are well-separated in space and interact primarily through molecule-molecule collisions. Conversely, although the molecules in solids are densely packed, they usually fall into a regular structure, such as a
Short-range ordering
While liquids do not exhibit long-range ordering as in a crystalline lattice, they do possess short-range order, which persists over a few molecular diameters.[47][48]
In all liquids, excluded volume interactions induce short-range order in molecular positions (center-of-mass coordinates). Classical monatomic liquids like argon and krypton are the simplest examples. Such liquids can be modeled as disordered "heaps" of closely packed spheres, and the short-range order corresponds to the fact that nearest and next-nearest neighbors in a packing of spheres tend to be separated by integer multiples of the diameter.[49][50]
In most liquids, molecules are not spheres, and intermolecular forces possess a directionality, i.e., they depend on the relative orientation of molecules. As a result, there is short-ranged orientational order in addition to the positional order mentioned above. Orientational order is especially important in hydrogen-bonded liquids like water.[51][52] The strength and directional nature of hydrogen bonds drives the formation of local "networks" or "clusters" of molecules. Due to the relative importance of thermal fluctuations in liquids (compared with solids), these structures are highly dynamic, continuously deforming, breaking, and reforming.[49][51]
Energy and entropy
The microscopic features of liquids derive from an interplay between attractive intermolecular forces and entropic forces.[53]
The attractive forces tend to pull molecules close together, and along with short-range repulsive interactions, they are the dominant forces behind the regular structure of solids. The entropic forces are not "forces" in the mechanical sense; rather, they describe the tendency of a system to maximize its entropy at fixed energy (see microcanonical ensemble). Roughly speaking, entropic forces drive molecules apart from each other, maximizing the volume they occupy. Entropic forces dominant in gases and explain the tendency of gases to fill their containers. In liquids, by contrast, the intermolecular and entropic forces are comparable, so it is not possible to neglect one in favor of the other. Quantitatively, the binding energy between adjacent molecules is the same order of magnitude as the thermal energy .[54]
No small parameter
The competition between energy and entropy makes liquids difficult to model at the molecular level, as there is no idealized "reference state" that can serve as a starting point for tractable theoretical descriptions. Mathematically, there is no small parameter from which one can develop a systematic
Role of quantum mechanics
Like all known forms of matter, liquids are fundamentally
| Liquid | Temperature (K) | (nm) | |
|---|---|---|---|
| Hydrogen (H2) | 14.1 | 0.33 | 0.97 |
| Neon | 24.5 | 0.078 | 0.26 |
| Krypton | 116 | 0.018 | 0.046 |
| Carbon tetrachloride (CCl4) | 250 | 0.009 | 0.017 |
For the classical limit to apply, a necessary condition is that the thermal
is small compared with the length scale under consideration.[54][58] Here, is the Planck constant and is the molecule's mass. Typical values of are about 0.01-0.1 nanometers (Table 1). Hence, a high-resolution model of liquid structure at the nanoscale may require quantum mechanical considerations. A notable example is hydrogen bonding in associated liquids like water,
For a liquid to behave classically at the macroscopic level, must be small compared with the average distance between molecules.[54] That is,
Representative values of this ratio for a few liquids are given in Table 1. The conclusion is that quantum effects are important for liquids at low temperatures and with small molecular mass.[54][56] For dynamic processes, there is an additional timescale constraint:
where is the timescale of the process under consideration. For room-temperature liquids, the right-hand side is about 10−14 seconds, which generally means that time-dependent processes involving translational motion can be described classically.[54]
At extremely low temperatures, even the macroscopic behavior of certain liquids deviates from classical mechanics. Notable examples are hydrogen and helium. Due to their low temperature and mass, such liquids have a thermal de Broglie wavelength comparable to the average distance between molecules.[54]
Dynamic phenomena
The expression for the sound velocity of a liquid,
- ,
contains the bulk modulus K. If K is frequency-independent, then the liquid behaves as a linear medium, so that sound propagates without dissipation or mode coupling. In reality, all liquids show some dispersion: with increasing frequency, K crosses over from the low-frequency, liquid-like limit to the high-frequency, solid-like limit . In normal liquids, most of this crossover takes place at frequencies between GHz and THz, sometimes called
At sub-GHz frequencies, a normal liquid cannot sustain
According to
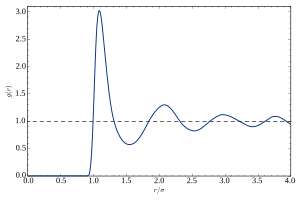
Experimental methods
The absence of long-range order in liquids is mirrored by the absence of
An equivalent representation of these correlations is the radial distribution function , which is related to the Fourier transform of .[49] It represents a spatial average of a temporal snapshot of pair correlations in the liquid.
Prediction of liquid properties
Methods for predicting liquid properties can be organized by their "scale" of description, that is, the length scales and time scales over which they apply.[64][65]
- Macroscopic methods use equations that directly model the large-scale behavior of liquids, such as their thermodynamic properties and flow behavior.
- Microscopic methods use equations that model the dynamics of individual molecules.
- Mesoscopic methods fall in between, combining elements of both continuum and particle-based models.
Macroscopic
Empirical correlations
Empirical correlations are simple mathematical expressions intended to approximate a liquid's properties over a range of experimental conditions, such as varying temperature and pressure.[66] They are constructed by fitting simple functional forms to experimental data. For example, the temperature-dependence of liquid viscosity is sometimes approximated by the function , where and are fitting constants.[67] Empirical correlations allow for extremely efficient estimates of physical properties, which can be useful in thermophysical simulations. However, they require high quality experimental data to obtain a good fit and cannot reliably extrapolate beyond the conditions covered by experiments.
Thermodynamic potentials
Thermodynamic potentials are functions that characterize the
Hydrodynamics
Hydrodynamic theories describe liquids in terms of space- and time-dependent macroscopic
Mesoscopic
Mesoscopic methods operate on length and time scales between the particle and continuum levels. For this reason, they combine elements of particle-based dynamics and continuum hydrodynamics.[64]
An example is the lattice Boltzmann method, which models a fluid as a collection of fictitious particles that exist on a lattice.[64] The particles evolve in time through streaming (straight-line motion) and collisions. Conceptually, it is based on the Boltzmann equation for dilute gases, where the dynamics of a molecule consists of free motion interrupted by discrete binary collisions, but it is also applied to liquids. Despite the analogy with individual molecular trajectories, it is a coarse-grained description that typically operates on length and time scales larger than those of true molecular dynamics (hence the notion of "fictitious" particles).
Other methods that combine elements of continuum and particle-level dynamics include smoothed-particle hydrodynamics,[74][75] dissipative particle dynamics,[76] and multiparticle collision dynamics.[77]
Microscopic
Microscopic simulation methods work directly with the equations of motion (classical or quantum) of the constituent molecules.
Classical molecular dynamics
Classical molecular dynamics (MD) simulates liquids using Newton's law of motion; from
Ab initio (quantum) molecular dynamics
Ab initio quantum mechanical methods simulate liquids using only the laws of quantum mechanics and fundamental atomic constants.[57] In contrast with classical molecular dynamics, the intermolecular force fields are an output of the calculation, rather than an input based on experimental measurements or other considerations. In principle, ab initio methods can simulate the properties of a given liquid without any prior experimental data. However, they are very expensive computationally, especially for large molecules with internal structure.
See also
References
- ISBN 1-57912-814-9
- S2CID 252979251.
- S2CID 22732411.
- ISSN 0009-2819.
- ISSN 0066-4146.
- ISBN 978-0-07-304859-8
- ISSN 0167-7322.
- ISBN 3-527-31497-0
- ISBN 1-895198-24-0
- ISBN 0-8493-9345-0
- ISBN 1-4018-4831-1
- ^ Gerald Wendt The prospects of nuclear power and technology D. Van Nostrand Company 1957 p. 266
- ISBN 1-56347-013-6
- ISBN 0-07-044451-X
- OCLC 8451210.
- ISBN 0-7506-7174-2
- S2CID 205276487.
- S2CID 235568215.
- S2CID 236566966.
- ISBN 0-8493-1081-4
- ISSN 0004-637X.
- S2CID 120735632.
- ISBN 978-0-8053-2736-6
- ISBN 978-0-07-304859-8
- ^ "Compressibility of Liquids". hyperphysics.phy-astr.gsu.edu. Archived from the original on 7 December 2017. Retrieved 8 May 2018.
- ^ Intelligent Energy Field Manufacturing: Interdisciplinary Process Innovations By Wenwu Zhang -- CRC Press 2011 Page 144
- ^ Knight (2008) p. 454
- ^ Fluid Mechanics and Hydraulic Machines by S. C. Gupta -- Dorling-Kindersley 2006 Page 85
- ^ Knight (2008) p. 448
- ^ Knight (2008) pp. 455-459
- ISBN 978-0-07-304859-8
- ISBN 978-3-11-058314-4.
- ISBN 978-0-08-033933-7
- ISBN 978-0-470-11539-8
- ^ Zhmud, Boris (2014), "Viscosity Blending Equations" (PDF), Lube-Tech, 93
- ^ "Viscosity Index". UK: Anton Paar. Archived from the original on March 9, 2020. Retrieved 29 August 2018.
- ^ Honey in Traditional and Modern Medicine by Laid Boukraa -- CRC Press 2014 Page 22--24
- ISBN 978-1-891389-22-1
- ISBN 978-981-3102-53-8
- ^ Siegel, Ethan (2014-12-11). "Does water freeze or boil in space?". Starts With A Bang!. Retrieved 2022-02-10.
- ^ Silberberg, pp. 188 and 502
- ISBN 978-0-544-85019-4
- S2CID 37248336.
- ^ S2CID 42203015.
- OCLC 696342117.
- PMID 26624528.
- ^ )
- OCLC 1259588062.
- ^ OCLC 13946448.
- S2CID 55689631.
- ^ )
- PMID 15991270.
- PMID 19805248.
- ^ )
- ^ OCLC 148639922.
- ^ OCLC 10145548.
- ^ )
- ^ Fisher, I.Z. (1964), Statistical Theory of Liquids, The University of Chicago Press
- PMID 24014589.
- S2CID 4938804.
- PMC 3081025.
- S2CID 104272002.
- doi:10.1063/1.1750497. Archived from the originalon 2016-05-15.
- ^ )
- OCLC 1337924123.
- OCLC 44712950.
- ISBN 978-0-470-11539-8. Archivedfrom the original on 2020-03-02. Retrieved 2019-09-18.
- ISBN 978-3-540-67311-8. Retrieved 2023-04-01.
- S2CID 249968848.
- ISSN 0047-2689.
- ^ Moffatt, H.K. (2015), "Fluid Dynamics", in Nicholas J. Higham; et al. (eds.), The Princeton Companion to Applied Mathematics, Princeton University Press, pp. 467–476
- OCLC 656397653.
- OCLC 812917029.
- S2CID 5987481.
- S2CID 221538477.
- S2CID 961922.
- S2CID 8433369.