Lipid-anchored protein
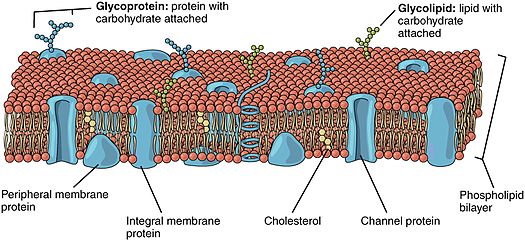
Lipid-anchored proteins (also known as lipid-linked proteins) are
The lipid groups play a role in protein interaction and can contribute to the function of the protein to which it is attached.[2] Furthermore, the lipid serves as a mediator of membrane associations or as a determinant for specific protein-protein interactions.[3] For example, lipid groups can play an important role in increasing molecular hydrophobicity. This allows for the interaction of proteins with cellular membranes and protein domains.[4] In a dynamic role[clarification needed], lipidation can sequester a protein away from its substrate to inactivate the protein and then activate it by substrate presentation.
Overall, there are three main types of lipid-anchored proteins which include prenylated proteins, fatty acylated proteins and glycosylphosphatidylinositol-linked proteins (GPI).[2][5] A protein can have multiple lipid groups covalently attached to it, but[clarification needed] the site where the lipids bind to the protein depends both on the lipid group and protein.[2]
Prenylated proteins


The prenylation motif “CaaX box” is the most common prenylation site in proteins, that is, the site where farnesyl or geranylgeranyl covalently attach.[2][3] In the CaaX box sequence, the C represents the cysteine that is prenylated, the A represents any aliphatic amino acid and the X determines the type of prenylation that will occur. If the X is an Ala, Met, Ser or Gln the protein will be farnesylated via the farnesyltransferase enzyme and if the X is a Leu then the protein will be geranylgeranylated via the geranylgeranyltransferase I enzyme.[3][4] Both of these enzymes are similar with each containing two subunits.[7]
Roles and function

Prenylated proteins are particularly important for eukaryotic cell growth, differentiation and morphology.
Some important prenylation chains that are involved in the HMG-CoA reductase metabolic pathway[1] are geranylgeraniol, farnesol and dolichol. These isoprene polymers (e.g. geranyl pyrophosphate and farnesyl pyrophosphate) are involved in the condensations via enzymes such as prenyltransferase that eventually cyclizes to form cholesterol.[2]
Fatty acylated proteins
Fatty acylated proteins are proteins that have been post-translationally modified to include the covalent attachment of fatty acids at certain amino acid residues.[11][12] The most common fatty acids that are covalently attached to the protein are the saturated myristic (14-carbon) acid and palmitic acid (16-carbon). Proteins can be modified to contain either one or both of these fatty acids.[11]

N-myristoylation
N-myristoylation (i.e. attachment of myristic acid) is generally an irreversible protein modification that typically occurs during protein synthesis
S-palmitoylation

S-palmitoylation (i.e. attachment of palmitic acid) is a reversible protein modification in which a palmitic acid is attached to a specific cysteine residue via
Palmitoylation mediates the affinity of a protein for
GPI proteins

Glycosylphosphatidylinositol-anchored proteins (GPI-anchored proteins) are attached to a GPI complex molecular group via an
Roles and function
The sugar residues in the tetrasaccaride and the fatty acid residues in the
References
- ^ ISBN 978-0-470-48337-4. Retrieved 13 November 2010.
- ^ ISBN 978-0470-54784-7.
- ^ PMID 8621375.
- ^ S2CID 11555502.
- .
- ^ isoprene (2003). "Miller-Keane Encyclopedia and Dictionary of Medicine, Nursing, and Allied Health, Seventh Ed". Retrieved 28 November 2015.
- ^ PMID 16477080.
- PMID 25768003.
- PMID 10394594.
- PMID 10961860.
- ^ S2CID 9734759.
- ^ PMID 21076176.
- ^ PMID 11527981.
- PMID 21056615.
- PMID 21819967.
- ^ Dityatev, Alexander (2006). El-Husseini, Alaa (ed.). Molecular Mechanisms of Synaptogenesis. New York: Springer. pp. 72–75.
- PMID 21131568.
- PMID 27976674.
- PMID 31060927.
- PMID 31672538.
- ^ PMID 26563290.
- ^ PMID 11995915.
- PMID 26563290.
- PMID 24727937.
- PMID 10068467.
External links
 Media related to Lipid-anchored protein at Wikimedia Commons
Media related to Lipid-anchored protein at Wikimedia Commons
