Acylation
Appearance
Source: Wikipedia, the free encyclopedia.
It has been suggested that Nucleophilic acyl substitution be merged into this article. (Discuss) Proposed since January 2024. |
Chemical reaction which adds an acyl group (R–C=O) to a compound
In
acyl
group (R−C=O) is added to a substrate. The compound providing the acyl group is called the acylating agent. The substrate to be acylated and the product include the following:
A particularly common type of acylation is acetylation, the addition of the acetyl group. Closely related to acylation is formylation, which employ sources of "HCO+ in place of "RCO+".
Examples
Because they form a strong
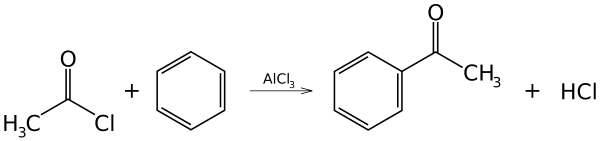
This reaction is an example of electrophilic aromatic substitution.
Acyl halides and acid anhydrides of carboxylic acids are also common acylating agents. In some cases, active esters exhibit comparable reactivity. All react with amines to form amides and with alcohols to form esters by nucleophilic acyl substitution.
Acylation can be used to prevent
rearrangement reactions that would normally occur in alkylation. To do this an acylation reaction is performed, then the carbonyl is removed by Clemmensen reduction or a similar process.[3]
Acylation in biology
See also:
protein lipidation
Protein acylation is the
residues of proteins.[7][8] Palmitoleoylation appears to play a significant role in the trafficking, targeting, and function of Wnt proteins.[9][10]
See also
- Hydroacylation
- Acetyl
- Ketene
