Pentose
In
Pentoses are very important in
Like some other monosaccharides, pentoses exist in two forms, open-chain (linear) or closed-chain (cyclic), that easily convert into each other in water solutions.
The term "pentose" sometimes is assumed to include deoxypentoses, such as deoxyribose: compounds with general formula C
5H
10O
5-y that can be described as derived from pentoses by replacement of one or more hydroxyl groups with hydrogen atoms.
Classification
The
In the open form, there are eight aldopentoses and four 2-ketopentoses,
Aldopentoses
The aldopentoses have three
 D-Arabinose |
 D-Lyxose |
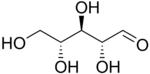 D-Ribose |
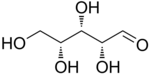 D-Xylose |
 L-Arabinose |
 L-Lyxose |
 L-Ribose |
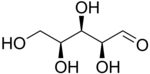 L-Xylose |
Ribose is a constituent of RNA, and the related molecule, deoxyribose, is a constituent of DNA. Phosphorylated pentoses are important products of the pentose phosphate pathway, most importantly ribose 5-phosphate (R5P), which is used in the synthesis of nucleotides and nucleic acids, and erythrose 4-phosphate (E4P), which is used in the synthesis of aromatic amino acids.
Ketopentoses
The 2-ketopentoses have two chiral centers; therefore, four (22) different stereoisomers are possible. The 3-ketopentoses are rare.
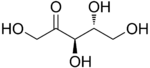 D-Ribulose |
 D-Xylulose |
 L-Ribulose |
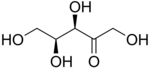 L-Xylulose |
Cyclic form
The closed or cyclic form of a pentose is created when the
The closure turns the carboxyl carbon into a chiral center, which may have any of two configurations, depending on the position of the new hydroxyl. Therefore, each linear form can produce two distinct closed forms, identified by prefixes "α" and "β".
Deoxypentoses
The one deoxypentose has two total stereoisomers.
 D-Deoxyribose |
 L-Deoxyribose |
Properties
In the cell, pentoses have a higher
A polymer composed of pentose sugars is called a pentosan.
Tests for pentoses
The most important tests for pentoses rely on converting the pentose to furfural, which then reacts with a chromophore. In Tollens’ test for pentoses (not to be confused with Tollens' silver-mirror test for reducing sugars), the furfural ring reacts with phloroglucinol to produce a colored compound;[4] in the aniline acetate test with aniline acetate;[5] and in Bial's test, with orcinol.[6] In each of these tests, pentoses react much more strongly and quickly than hexoses.
References
- ^ Pentose, Merriam-Webster
- ^ "D-Ribose". PubChem compound webpage, accessed on 2010-02-06.
- ^ a b Morrison, Robert Thornton; Boyd, Robert Neilson. Organic Chemistry (2nd ed.). Allyn and Bacon. Library of Congress catalog 66-25695
- ISSN 0365-9496.
- ISBN 9781337517140.
- ISBN 0534408338.
