Ribose
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
D-Ribose
| |||
| Systematic IUPAC name
(2R,3R,4S,5R)-5-(hydroxymethyl)oxolane-2,3,4-triol | |||
| Other names
d-Ribose
| |||
| Identifiers | |||
3D model (
JSmol ) |
| ||
| ChEMBL | |||
| ChemSpider |
| ||
| DrugBank | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties[1][2] | |||
| C5H10O5 | |||
| Molar mass | 150.13 | ||
| Appearance | White solid | ||
| Melting point | 95 °C (203 °F; 368 K) | ||
| 100 g/L (25 °C, 77 °F) | |||
Chiral rotation ([α]D)
|
−21.5° (H2O) | ||
| Related compounds | |||
Related aldopentoses
|
Arabinose Xylose Lyxose | ||
Related compounds
|
Deoxyribose | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||

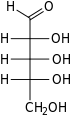
Ribose is a
Like most sugars, ribose exists as a mixture of
The
Synthesis and sources
Ribose as its 5-phosphate ester is typically produced from glucose by the pentose phosphate pathway. In at least some archaea, alternative pathways have been identified.[13]
Ribose can be synthesized chemically, but commercial production relies on fermentation of glucose. Using genetically modified strains of
Ribose has been detected in meteorites.[15][16]
Structure
Ribose is an
The "d-" in the name d-ribose refers to the
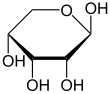
-
α-d-Ribopyranose
-
β-d-Ribopyranose
-
α-d-Ribofuranose
-
β-d-Ribofuranose
Relative abundance of forms of ribose in solution: β-d-ribopyranose (59%), α-d-ribopyranose (20%), β-d-ribofuranose (13%), α-d-ribofuranose (7%) and open chain (0.1%).[11]
For ribose residues in
In closed ring riboses, the observed flexibility mentioned above is not observed because the ring cycle imposes a limit on the number of torsion angles possible in the structure.
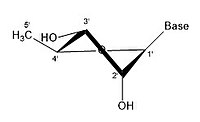
- Some pucker configurations of Ribose
-
2' endo
-
2' endo 3' exo
-
3' endo 2' exo
-
3' endo
A ribose molecule is typically represented as a planar molecule on paper. Despite this, it is typically non-planar in nature. Even between hydrogen atoms, the many constituents on a ribose molecule cause
Functions
ATP is derived from ribose; it contains one ribose, three phosphate groups, and an adenine base. ATP is created during cellular respiration from adenosine diphosphate (ATP with one less phosphate group).
Signaling pathways
Ribose is a building block in secondary signaling molecules such as

Metabolism
Ribose is referred to as the "molecular currency" because of its involvement in intracellular energy transfers.[citation needed] For example, nicotinamide adenine dinucleotide (NAD), flavin adenine dinucleotide (FAD), and nicotinamide adenine dinucleotide phosphate (NADP) all contain the d-ribofuranose moiety. They can each be derived from d-ribose after it is converted to d-ribose 5-phosphate by the enzyme ribokinase.[22][23] NAD, FAD, and NADP act as electron acceptors in biochemical redox reactions in major metabolic pathways including glycolysis, the citric acid cycle, fermentation, and the electron transport chain.

Nucleotide biosynthesis
Nucleotides are synthesized through salvage or
Modifications
Modifications in nature
One important modification occurs at the C2' position of the ribose molecule. By adding an
Synthetic modifications
Along with phosphorylation, ribofuranose molecules can exchange their oxygen with selenium and sulfur to produce similar sugars that only vary at the 4' position. These derivatives are more lipophilic than the original molecule. Increased lipophilicity makes these species more suitable for use in techniques such as PCR, RNA aptamer post-modification, antisense technology, and for phasing X-ray crystallographic data.[27]
Similar to the 2' modifications in nature, a synthetic modification of ribose includes the addition of
Medical uses
d-ribose has been suggested for use in management of
Supplemental d-ribose can bypass part of the
References
- ISBN 091191028X, 8205
- ISBN 0-8493-0462-8.
- from the original on 4 June 2020. Retrieved 12 March 2020.
- .
- .
- ^ from the original on 26 October 2023. Retrieved 15 December 2019.
- .
- ^ ISBN 9781118681961. Archivedfrom the original on 26 October 2023. Retrieved 15 December 2019.
- ^ Leigh, Jeffery (July–August 2012). "Non-IUPAC Nomenclature Systems". Chemistry International. 34 (4). International Union of Pure and Applied Chemistry. Archived from the original on 5 December 2019. Retrieved 15 December 2019.
- ^ ISBN 9781000650907. Archivedfrom the original on 26 October 2023. Retrieved 15 December 2019.
- ^ .
- ISBN 9780120026449.
- PMID 9324245.
- S2CID 34340369.
- ^ Steigerwald, Bill; Jones, Nancy; Furukawa, Yoshihiro (18 November 2019). "First Detection of Sugars in Meteorites Gives Clues to Origin of Life". NASA. Archived from the original on 15 January 2021. Retrieved 18 November 2019.
- PMID 31740594.
- ^ ISBN 9780935702491.
- ISBN 978-0470570951.
- ISSN 1520-6106.
- ^ "Nucleic acid architecture". fbio.uh.cu. Archived from the original on 17 May 2018. Retrieved 8 October 2019.
- ISBN 9780123695079.
- PMID 8382990.
- S2CID 11439854.
- ^ ISBN 9780323357623.
- ^ "Herbal Remedies, Supplements A-Z Index". PDRHealth.com. PDR, LLC. Archived from the original on 11 October 2008.
- ^ .
- ^ S2CID 91052034.
- PMID 21612237.
- PMID 19641697.
- PMID 17109576.
- ^ "Ribose". wa.kaiserpermanente.org. Archived from the original on 3 March 2021. Retrieved 7 October 2019.