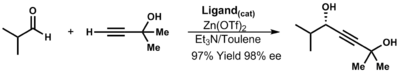Organozinc chemistry
Organozinc chemistry is the study of the physical properties, synthesis, and reactions of organozinc compounds, which are
Organozinc compounds were among the first organometallic compounds made. They are less reactive than many other analogous organometallic reagents, such as
Organozincs can be categorized according to the number of carbon substituents that are bound to the metal.[2][3]
- Diorganozinc (R2Zn): A class of organozinc compounds in which two alkyl ligands. These may be further divided into subclasses depending on the other ligandsattached
- Heteroleptic (RZnX): Compounds which an electronegative or monoanionic ligand (X), such as a halide, is attached to the zinc center with another alkyl or aryl substituent (R).
- Ionic organozinc compounds: This class is divided into organozincates (RnZn−) and organozinc cations(RZnL+
n).
Bonding
In its
Typical diorganozinc complexes have the formula R2Zn. Dialkylzinc compounds are monomeric with a linear coordination at the zinc atom.
These structures cause zinc to have two bonding d-orbitals and three low-lying non-bonding d-orbitals (see

Synthesis
Several methods exist for the generation of organozinc compounds. Commercially available diorganozinc compounds are
-
(2.1)
From zinc metal
Frankland's original synthesis of
-
2 EtI + 2 Zn0 → Et
2Zn + ZnI
2(2.2)
-
(2.3)
-
(2.4)
Functional group exchange
The two most common zinc functional group interconversion reactions are with halides and boron, which is catalyzed by
-
(2.5)
This group transfer reaction can be used in
-
(2.6)
β-Silyl diorganozinc compounds
One of the major drawbacks of diorganozinc alkylations is that only one of the two alkyl substituents is transferred. This problem can be solved by using Me3SiCH2- (TMSM), which is a non-transferable group.[14]
-
(2.7)
Transmetallation
-
HgPh2 + Zn → ZnPh2 + Hg(2.8)
The benefit of transmetalling to zinc it is often more tolerant of other functional groups in the molecule due to the low reactivity which increases selectivity.[15]
- In the synthesis of Maoecrystal V, a Esters are significantly stable against organozinc reagents.[16]
-
(2.9)
Organozinc can be obtained directly from zinc metal:[17][18]
-
(2.10)
- In this method zinc is activated by 1,2-dibromoethane and trimethylsilyl chloride. A key ingredient is lithium chloridewhich quickly forms a soluble adduct with the organozinc compound thus removing it from the metal surface.
Reactions
In many of their reactions organozincs appear as intermediates.
- In the Frankland–Duppa reaction (1863) an alkyl halide R'X, zinc and hydrochloric acid to the α-hydroxycarboxylic esters RR'COHCOOR[19]
Reformatsky reaction
This organic reaction can be employed to convert α-haloester and ketone or aldehyde to a β-hydroxyester. Acid is needed to protonate the resulting alkoxide during work up. The initial step is an oxidative addition of zinc metal into the carbon-halogen bond, thus forming a carbon-zinc enolate. This C-Zn enolate can then rearrange to the Oxygen-Zinc enolate via coordination. Once this is formed the other carbonyl containing starting material will coordinate in the manner shown below and give the product after protonation.[20] The benefits of the Reformatsky reaction over the conventional aldol reaction protocols is the following:
- Allows for exceedingly derivatized ketone substrates
- The ester enolate intermediate can be formed in the presence of enolizable moieties
- Well suited for intramolecular reactions
Below shows the six-membered transition state of the Zimmerman–Traxler model (Chelation Control, see Aldol reaction), in which R3 is smaller than R4.[21]
-
(3.1)
The Reformatsky reaction has been employed in numerous total syntheses such as the synthesis of C(16),C(18)-bis-epi-cytochalasin D:[22]
-
(3.2)
The Reformatsky reaction even allows for with zinc homo-enolates.[23] A modification of the Reformatsky reaction is the Blaise reaction.[21]
-
(3.3)
Simmons–Smith reaction
The
-
(3.4)
-
(3.5)
Although the mechanism has not been fully elaborated it is hypothesized that the organozinc intermediate is a metal-
-
(3.6)
Titanium–zinc methylidenation
Organozinc compounds derived from
Negishi coupling
This powerful carbon-carbon bond forming
Alkylzinc species require the presence of at least a stoichiometric amount of halide ions in solution to form a "zincate" species of the form RZnX32−, before it can undergo transmetalation to the palladium centre.[28] This behavior contrasts greatly with the case of aryl zinc species. A key step in the catalytic cycle is a transmetalation in which a zinc halide exchanges its organic substituent for another halogen with the metal center.
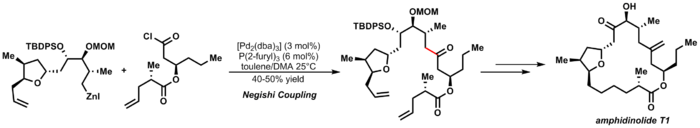
An elegant example of Negishi coupling is Furstner's synthesis of amphidinolide T1:[29]
-
(3.7)
Fukuyama coupling
Fukuyama coupling is a palladium-catalyzed reaction involving the coupling of an aryl, alkyl, allyl, or α,β- unsaturated thioester compound. This thioester compound can be coupled to a wide range of organozinc reagents in order to reveal the corresponding ketone product. This protocol is useful due to its sensitivity to functional groups such as ketone, acetate, aromatic halides, and even aldehydes. The chemoselectivity observed indicates ketone formation is more facile than oxidative addition of palladium into these other moieties.[30]
-
(3.8)
A further example of this coupling method is the synthesis of (+)-biotin. In this case, the Fukuyama coupling takes place with the thiolactone:[31]
-
(3.9)
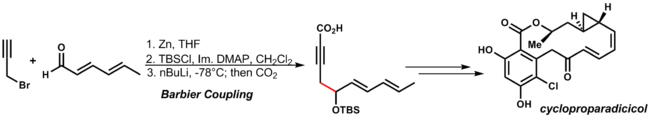
Barbier reaction
The
-
(3.10)
The mechanism resembles the
-
(3.11)
Zinc acetylides
The formation of the zinc
-
(3.12)
In the absence of ligands, the
-
(3.13)
-
(3.14)
- The α-stereocenter of the ligand dictates observed stereochemistry of the propargylic alcohols
- The steric effects between the aldehyde substituent and the ligand are less important but still dictate the favored conformation
Zinc-acetylides are used in the
-
(3.15)
Organozincates
The first organozinc ate complex (organozincate) was reported in 1858 by James Alfred Wanklyn,[38] an assistant to Frankland and concerned the reaction of elemental sodium with diethylzinc:
-
2 Na + 3 ZnEt2 → 2 NaZnEt3 + Zn(4.1)
Organozinc compounds that are strongly
Synthesis
Tetraorganozincates such as [Me4Zn]Li2 can be formed by mixing Me2Zn and MeLi in a 1:2 molar ratio of the rectants. Another example synthetic route to forming spriocyclic organozincates is shown below:[3]
-
(4.2)
Triorganozincates compounds are formed by treating a diorganozinc such as (Me3SiCH2)2Zn with an
-
2 NaZnEt3 → Na2Zn2H2Et4 + 2 C2H4(4.3)
The product is an edge-shared bitetrahedral structure, with
Reactions
Although less commonly studied, organozincates often have increased reactivity and selectivity compared to the neutral diorganozinc compounds. They have been useful in stereoselective alkylations of ketones and related carbonyls, ring opening reactions. Aryltrimethylzincates participate in vanadium mediated C-C forming reactions.[3]
-
(4.4)
Organozinc(I) compounds
Low valent organozinc compounds having a Zn–Zn bond are also known. The first such compound, decamethyldizincocene, was reported in 2004.[40]
See also
- Compounds of zinc
References
- ISBN 0471264180.
- ^ ISBN 0-470-09337-4.
- ^ ISBN 0-19-850121-8.
- ISBN 3-13-103061-5
- ^ a b E. Frankland, Liebigs Ann. Chem.,1849, 71, 171
- ISBN 978-3-527-29390-2
- PMID 21919175.
- PMID 16721894.
- tetraethylethylene diamine (TEEDA) resulting in an enantiomeric excessof 92%.
- S2CID 92794.
- .
- PMID 11667810.
- ^ Naka,H; et al.New J. Chem., 2010, 34, 1700–1706
- ^ Knochel,P.; et al. Angel. Chem. Int. Ed. Engl. 1997, volume 36, 1496-1498
- .
- PMID 24047444.
- PMID 16900548.
- nucleophilic displacement
- ISBN 9780911910131. Frankland–Duppa reaction.
- S2CID 94339252.)
- ^ a b c d Kurti, L.; Czako, B. Strategic Applications of Named Reactions in Organic Synthesis; Elsevier: Burlington, 2005.
- PMID 10987942.
- ^ Kumwaijima,I.; et al. J. Am. Chem. 1987, 109, 8056
- .
- ISBN 9780080405926.
- ^ S. Sase, M. Jaric, A. Metzger, V. Malakhov, P. Knochel, J. Org. Chem., 2008, 73, 7380-7382
- PMID 15991198.)
- PMID 22685029.
- PMID 14664598.
- .
- .
- PMID 15212536.
- ^ Li, Z.; Upadhyay, V.; DeCamp, A. E.; DiMichele, L.; Reider, P. J. Synthesis 1999, 1453-1458.
- .
- .
- PMID 3358127.
- ^ Thompson, A. S.; Corley, E. G.; Huntington, M. F.; Grabowski, E. J. J. Tetrahedron Lett. 1995, 36, 8937-8940
- .
- PMID 17665387.
- PMID 20486240.


![Reicke zinc allows for an activated zinc species for oxidative addition {\displaystyle {\ce {{ZnCl2}+2K->[{\ce {THF}}][{\ce {-2KCl}}]}}\overbrace {\ce {Zn^{0}}} ^{\ce {Riecke\ zinc}}+{\ce {R-X->[{\ce {THF}}][20-60^{\circ }{\ce {C}}]R-Zn-I}}\qquad {\begin{cases}\mathbf {R} :&{\text{Allyl, Aryl, Alkyl, Benzyl}}\\\mathbf {X} :&{\text{Bromide, Iodide}}\end{cases}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/18c7cba23f482f24117a7664d614603a1271e9f2)


![Knochel and coworkers beta silyl group transfer addition {\displaystyle {\begin{array}{l}{}\\{\ce {{R2Zn}+(TMSM)2Zn}}\ {\overset {\ce {THF}}{\ce {<=>>}}}\ {\ce {2R(TMSM)Zn}}\\{}\\{\ce {{RZnI}+(TMSM)Li->[{\ce {THF}}][-80^{\circ }\!{\ce {C}}]{R(TMSM)Zn}+LiI}}\\{}\end{array}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/0fdd1941ffd29949716def994df7c380dd816d96)