Togni reagent II

| |
| Names | |
|---|---|
| Preferred IUPAC name
1-(Trifluoromethyl)-1λ3,2-benziodoxol-3(1H)-one | |
| Other names
Togni's reagent II; Togni reagent 2
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.214.822 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H4F3IO2 | |
| Molar mass | 316.018 g·mol−1 |
| Appearance | colorless crystalline solid |
| Melting point | 122.4–123.4 °C (252.3–254.1 °F; 395.5–396.5 K) |
| soluble in methylene chloride, chloroform, acetonitrile, methanol, ethanol, acetone | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Togni reagent II (1-trifluoromethyl-1,2-benziodoxol-3(1H)-one) is a chemical compound used in organic synthesis for direct electrophilic trifluoromethylation.[1][2]
History
Synthesis, properties, and reactivity of the compound were first described in 2006 by Antonio Togni and his coworkers at ETH Zurich.[3] The article also contains information on Togni reagent I (1,3-dihydro-3,3-dimethyl-1-(trifluoromethyl)-1,2-benziodoxole).
Preparation
The synthesis consists of three steps. In the first step,
Alternatively,
Properties
Physical properties
The compound crystallized in a monoclinic crystal structure. The space group is P21/n with four molecules in the unit cell.[3] From the crystallographic data, a density of 2.365 g·cm−3 was deduced.[3]
Chemical properties
Pure Togni reagent II is
Uses
Togni reagent II is used for trifluoromethylation of organic compounds. For phenolates, the substitution takes place preferably in the ortho position. It is possible to obtain a second substitution by using an excess of Togni reagent II.[7]
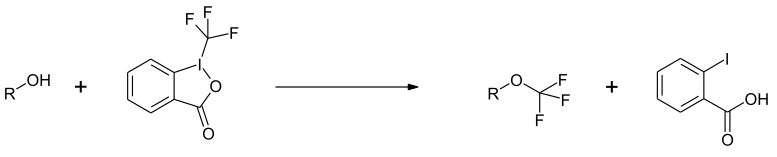
Reactions with alcohols yield the corresponding trifluoromethyl ethers.[8]
Trifluoromethylation of alkenes is possible under copper catalysis.[9]
References
- ^ PubChem. "1-(Trifluoromethyl)-1,2-benziodoxol-3(1H)-one". pubchem.ncbi.nlm.nih.gov. National Center for Biotechnology Information. Retrieved 2020-07-07.
- .
- ^ PMID 16402401.
- ^ ISBN 978-0-471-93623-7
- PMID 23734560.
- ISSN 1083-6160.
- PMID 18771328.
- .
- PMID 21919144.