Trifluoromethylation
Trifluoromethylation in
History
The first to investigate trifluoromethyl groups in relationship to biological activity was F. Lehmann in 1927.[5] An early review appeared in 1958.[6] An early synthetic method was developed by Frédéric Swarts in 1892,[7] based on antimony fluoride. In this reaction benzotrichloride was reacted with SbF3 to form PhCF2Cl and PhCF3. In the 1930s
. The McLoughlin-Thrower reaction (1968) is an early coupling reaction using iodofluoroalkanes, iodoaromatic compounds and copper.[8] In 1969 Kobayashi & Kumadaki adapted their protocol for trifluoromethylations.[9][10]
|
| McLoughlin-Thrower reaction (1968) |
Reagents
Trifluoromethyltrimethylsilane
Preparation of the trifluoromethyltrimethylsilane was reported by Ingo Ruppert in 1984.[11] In 1989, Prakash and Olah first reported activation of TMSCF3 by fluoride to perform nucleophilic trifluoromethylation of carbonyl compounds.[12] In the same year, Stahly described similar reactions for the synthesis of trifluoromethylated phenols and anilines.[13] Since then TMSCF3 has been widely used as a nucleophilic trifluoromethylating agent.[14][15]
An example is the trifluoromethylation of

|
| Trifluoromethylation using
trifluoromethyltrimethylsilane[16] |
The substrates can be aryl halides.[17][18] Potassium (trifluoromethyl)trimethoxyborate for this purpose has been synthesised from B(OMe)3, CF3SiMe3 and KF.[19] Aryl functionalization by C-H activation has also been reported.[20][21]
Sodium trifluoroacetate
Sodium trifluoroacetate as a reagent for trifluoromethylations was introduced by Matsui in 1981. In the original scope the substrate was an aromatic halide and the metal salt copper(I)iodide.[22][23]
Trifluoromethane
Fluoroform (CF3H) has been employed as a trifluoromethylation reagent for aldehydes in combination with a strong base.[24]

|
| Trifluoromethylation fluoroform folleas 1998[24] |
Trifluoroiodomethane
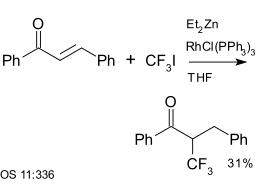
|
| Trifluoromethylation using diethyl zinc and Wilkinson's catalyst[25]
|
Trifluoromethyl sulfone
Trifluoromethyl sulfone (PhSO2CF3) and trifluoromethyl sulfoxide (PhSOCF3) can be used for trifluoromethylations of electrophiles[26]
Trifluoromethanesulfonyl chloride
Trifluoromethanesulfonyl chloride (or
Sodium trifluoromethanesulfinate

|
| Trifluorination Langlois reagent 2011[29] |
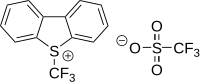
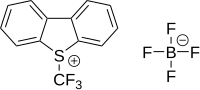
Umemoto reagents
Umemoto reagents are (trifluoromethyl)dibenzoheterocyclic salts, such as 5-(trifluoromethyl)dibenzothiophenium triflate and 5-(trifluoromethyl)dibenzothiophenium tetrafluoroborate.[30][31]
Trifluoromethyl-metal reagents
Many CF3-containing metal complexes have been prepared, and some are useful for trifluoromethylation. The most obvious reagent is CF3Li, which can be generated by lithium-iodide exchange. This compound is however unstable even at low temperatures. It degrades to
Reaction types
Aromatic coupling reactions
In
A

|

|
| Aromatic trifluoromethylation Kitazume 1982[35] | Aromatic catalytic
trifluoromethylation Oishi 2009[37] |
Radical trifluoromethylation
In radical trifluoromethylation the active species is the trifluoromethyl

|
| Trifluoromethylation using CF3I and triethylborane.
The base is 2,6-lutidine[50]
|
In the CF3 radical the fluorine atom is an
Nucleophilic trifluoromethylation
In nucleophilic trifluoromethylation the active species is the CF3− anion.[54] It was, however, widely believed that the trifluoromethyl anion is a transient species and thus cannot be isolated or observed in the condensed phase. Contrary to the popular belief, the CF3 anion, with [K(18-crown-6)]+ as a countercation, was produced and characterized by Prakash and coworkers.[55] The challenges associated with observation of CF3 anion are alluded to its strong basic nature and its tendency to form pentacoordinated silicon species, such as [Me3Si(CF3)2]− or [Me3Si(F)(CF3)]−.
The reactivity of

|
| trifluoromethylation using methyl fluorosulfonyldifluoroacetate.
The intermediate is CF3Cu[59] |
Electrophilic trifluoromethylation
In electrophilic trifluoromethylation the active trifluoromethyl donor group carries a positive charge.
Another group of trifluoromethyl donors are

|
| Trifluoromethylation at a thiol group using hypervalent iodine [71] |
The
Asymmetric trifluoromethylation
In
. Because Ruppert's reagent requires a tetraalkylammonium fluoride, chiral ammonium fluorides have been employed in In the field of electrophilic trifluoromethylation an early contribution involved reaction of a metal enolate with a trifluoromethyl chalcogen salt in presence of a chiral boron catalyst.[80]
|
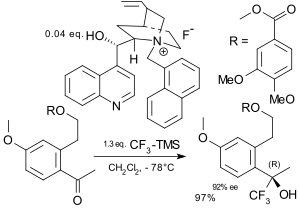
|
| Asymmetric trifluoromethylation Iseki 1994[78] | Asymmetric trifluormethylation Caron 2003[79] |
More recent examples of highly enantioselective methods for the α-trifluoromethylation of carbonyls are available through enamine catalysis of aldehydes (photoredox[81] or iodonium[82]), copper catalysis of β-ketoesters,[83] and radical addition to zirconium enolates.[84]
References
- ^ PMID 21456523.
- PMID 21614074.
- PMID 22488902.
- PMID 25635524.
- ^ Lehmann, F. "Chemical constitution and activity. Aromatic fluorine compounds." Arch. exp. Pathol. Pharmakol 130 (1928): 250-255.
- PMID 13665284.
- ^ Swarts (1892). Acad. Roy. Belg. 3 (24): 474.
{{cite journal}}: Missing or empty|title=(help) - .
- .
- .
- .
- .
- .
- PMID 11848888.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 24754488.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ .
- PMID 21442711.
- PMID 19319442.
- PMID 21274956.
- PMID 21932827.
- PMID 22318969.
- .
- .
- ^ .
- ^ .
- PMID 12943400.
- PMID 22158245.
- .
- ^ PMID 21844378.
- PMID 25011917.
- .
- ^ Donald J. Burton, Long Lu "Fluorinated Organometallic Compounds" Topics in Current Chemistry, 1997, Vol. 193, p. 45.
- .
- ^ .
- .
- ^ PMID 19319442.
- PMID 21274956.
- PMID 20923196.
- PMID 21235259.
- PMID 21328665.
- PMID 21442711.
- PMID 20576888.
- .
- .
- .
- .
- PMID 1133630.
- .
- ^ .
- .
- PMID 22890985.
- .
- ^ .
- PMID 25146595.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- doi:10.1039/A801406J.
- PMID 11149825.
- .
- PMID 20703379.
- PMID 11848810.
- ^ PMID 25335765.
- ^ Yagupolskii, L. M.; Kondratenko, N. V.; Timofeeva, G. N. J. Org. Chem. USSR 1984, 20, 103–106.
- .
- .
- ^ Here, iodine is considered to be less electronegative (2.3) than carbon (2.5), per the IUPAC definition that de facto mandates that Allen electronegativity be used; the more commonly encountered Pauling electronegativity scale would imply that the molecule in question would be that of iodine(I), not iodine(III).
- PMID 16402401.
- PMID 17154193.
- S2CID 205709340.
- PMID 18771328.
- ^ .
- PMID 21919144.
- PMID 22407851.
- PMID 15584697.
- .
- PMID 1542092.
- .
- ^ .
- ^ .
- .
- PMID 19722670.
- PMID 20297822.
- PMID 22693950.
- PMID 22486383.