Ugi reaction
| Ugi reaction | |
|---|---|
| Named after | Ivar Karl Ugi |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | ugi-reaction |
| RSC ontology ID | RXNO:0000129 |
In organic chemistry, the Ugi reaction is a multi-component reaction involving a ketone or aldehyde, an amine, an isocyanide and a carboxylic acid to form a bis-amide.[1][2][3][4] The reaction is named after Ivar Karl Ugi, who first reported this reaction in 1959.

The Ugi reaction is
Due to the reaction products being potential protein mimetics there have been many attempts to development an enantioselective Ugi reaction,[13] the first successful report of which was in 2018.[14]
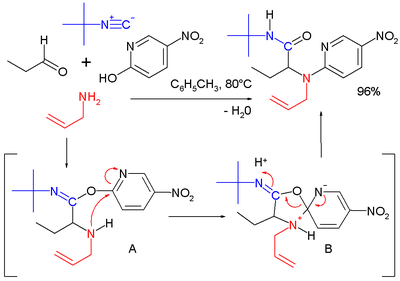
Reaction mechanism
One plausible reaction mechanism is depicted below:[15]

In the related Passerini reaction (lacking the amine) the isocyanide reacts directly with the carbonyl group but other aspects of the reaction are the same. This reaction can take place concurrently with the Ugi reaction, acting as a source of impurities.
Variations
Combination of reaction components
The usage of bifunctional reaction components greatly increases the diversity of possible reaction products. Likewise, several combinations lead to structurally interesting products. The Ugi reaction has been applied in combination with an
A reaction in its own right is the Ugi–Smiles reaction with the carboxylic acid component replaced by a phenol. In this reaction the Mumm rearrangement in the final step is replaced by the Smiles rearrangement.[17]
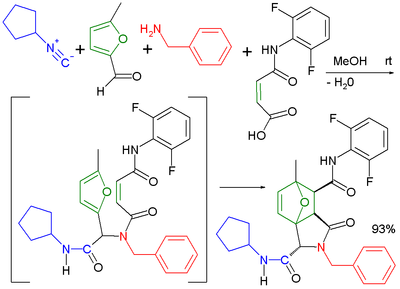 |
| |
| Ugi–Diels–Alder reaction | Ugi–Smiles reaction |
Another combination (with separate workup of the Ugi intermediate) is one with the
 |

| |
| Ugi–Buchwald–Hartwig reaction [20] | Ugi–Heck reaction [21] |
Combination of amine and carboxylic acid
Several groups have used β-amino acids in the Ugi reaction to prepare β-lactams.[22] This approach relies on acyl transfer in the Mumm rearrangement to form the four-membered ring. The reaction proceeds in moderate yield at room temperature in methanol with formaldehyde or a variety of aryl aldehydes. For example, p-nitrobenzaldehyde reacts to form the β-lactam shown in 71% yield as a 4:1 diastereomeric mixture:

Combination of carbonyl compound and carboxylic acid
Zhang et al.[23] have combined aldehydes with carboxylic acids and used the Ugi reaction to create lactams of various sizes. Short et al.[24] have prepared γ-lactams from keto-acids on solid-support.
Applications
Chemical libraries
The Ugi reaction is one of the first reactions to be exploited explicitly to develop chemical libraries. These chemical libraries are sets of compounds that can be tested repeatedly. Using the principles of combinatorial chemistry, the Ugi reaction offers the possibility to synthesize a great number of compounds in one reaction, by the reaction of various ketones (or aldehydes), amines, isocyanides and carboxylic acids. These libraries can then be tested with enzymes or living organisms to find new active pharmaceutical substances. One drawback is the lack of chemical diversity of the products. Using the Ugi reaction in combination with other reactions enlarges the chemical diversity of possible products. Recently, a breakthrough has been made in the field of covalent organic frameworks (COFs), where Ugi reaction is being utilized to introduce different functional handles into the COFs by post-synthetic modification.[25] With this new promising strategy, it is believed a library of COFs can be prepared with useful functional handles for various important applications.
Examples of Ugi reaction combinations:
- Isoquinolines from Ugi and Heck reactions.[26]
Pharmaceutical industry
Additionally, many of the caine-type anesthetics are synthesized using this reaction. Examples include lidocaine and bupivacaine.
See also
References
- .
- .
- .
- .
- S2CID 224890321.
- ISBN 0-08-040593-2.
- S2CID 53949436.
- ISBN 0-471-68260-8.)
- PMID 16312152.
- PMID 11281825.
- PMID 11039522.
- PMID 11028061.
- PMID 29708723.
- PMID 30213886.
- PMID 16292793.
- PMID 17137394.
- PMID 16928063.
- PMID 16956224.
- PMID 16961408.
- phosphineligand Me-Phos
- tetra-n-butylammonium bromide in dimethylformamide
- ^
Gedey S, Van der Eycken J, Fülöp F (May 2002). "Liquid-phase combinatorial synthesis of alicyclic beta-lactams via Ugi four-component reaction". Organic Letters. 4 (11): 1967–9. PMID 12027659.
- PMID 11674195.
- .
- S2CID 257425598.
- PMID 15330611.
- .
