Phenols
In
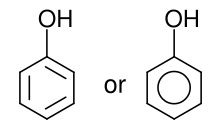
Phenols are both synthesized industrially and produced by plants and microorganisms.[2]
Properties
Acidity
Phenols are more
Condensation with aldehydes and ketones
Phenols are susceptible to Electrophilic aromatic substitutions. Condensation with formaldehyde gives resinous materials, famously Bakelite.
Another industrial-scale electrophilic aromatic substitution is the production of bisphenol A, which is produced by the condensation with acetone.[3]
C-Alkylation with alkenes
Phenol is readily alkylated at the ortho positions using alkenes in the presence of a Lewis acid such as
- CH2=CR2 + C6H5OH → R2CHCH2-2-C6H4OH
More than 100,000 tons of
Other reactions
Phenols undergo
Phenols are oxidized to hydroquinones in the Elbs persulfate oxidation.
Reaction of naphtols and hydrazines and sodium bisulfite in the Bucherer carbazole synthesis.
Synthesis
Many phenols of commercial interest are prepared by elaboration of
- rearrangement of esters in the Fries rearrangement[7][8]
- rearrangement of N-phenylhydroxylamines in the Bamberger rearrangement[9][10]
- ethers
- reduction of quinones
- replacement of an aromatic amine by an hydroxyl group with water and sodium bisulfide in the Bucherer reaction[11]
- thermal decomposition of aryl diazonium salts, the salts are converted to phenol[12]
- by the oxidation of aryl silanes—an aromatic variation of the Fleming-Tamao oxidation[13]
- catalytic synthesis from aryl bromides and iodides using nitrous oxide[14]
Classification
There are various
| Phenol | the parent compound, used as a disinfectant and for chemical synthesis |
| Bisphenol A | and other bisphenols produced from ketones and phenol / cresol |
| BHT | (butylated hydroxytoluene) - a fat-soluble antioxidant and food additive |
4-Nonylphenol |
a breakdown product of detergents and nonoxynol-9
|
Orthophenyl phenol |
a citrus fruits
|
| Picric acid | (trinitrophenol) - an explosive material
|
| Phenolphthalein | pH indicator |
| Xylenol | used in antiseptics & disinfectants |
Drugs and bioactive natural products
More than 371 drugs approved by the FDA between the years of 1951 and 2020 contain either a phenol or a phenolic ether (a phenol with an alkyl), with nearly every class of small molecule drugs being represented, and natural products making up a large portion of this list.[17]
| tyrosine | one of the 20 standard amino acids |
L-DOPA |
dopamine prodrug used to treat Parkinson's disease |
| propofol | short-acting intravenous anesthetic agent |
| vitamin K hydroquinone | blood-clotting agent that converts |
| levothyroxine (L-thyroxine) | Top-selling drug to treat thyroid hormone deficiency. |
| amoxicillin | Top-selling antibiotic |
| estradiol | the major female sex hormone |
References
- PMID 10802549.
- ^ )
- ^ 2,4-Hexadienedioic acid, monomethyl ester, (Z,Z)- Organic Syntheses, Coll. Vol. 8, p. 490 (1993); Vol. 66, p. 180 (1988) Article.
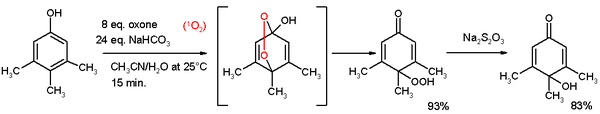
- ^ "2,5-Cyclohexadiene-1,4-dione, 2,3,5-trimethyl". Organic Syntheses. 52: 83. 1972.
- PMID 16548026.
- .
- .
- .
- .
- .
- .
- S2CID 31736757.
- PMID 35478236.
- ^ a b Wilfred Vermerris and Ralph Nicholson. Phenolic Compound Biochemistry Springer, 2008.
- ^ Harborne, J. B. (1980). "Plant phenolics". In Bell, E. A.; Charlwood, B. V. (eds.). Encyclopedia of Plant Physiology, volume 8 Secondary Plant Products. Berlin Heidelberg New York: Springer-Verlag. pp. 329–395.
- S2CID 248667453.