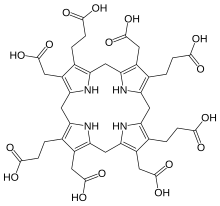
Uroporphyrinogen III
| |
| Identifiers | |
|---|---|
| MeSH | Uroporphyrinogen+III |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C40H44N4O16 | |
| Molar mass | 836.795 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Uroporphyrinogen III is a tetrapyrrole, the first macrocyclic intermediate in the biosynthesis of heme, chlorophyll, vitamin B12, and siroheme. It is a colorless compound, like other porphyrinogens.[1]
Structure
The
Biosynthesis and metabolism
In the general
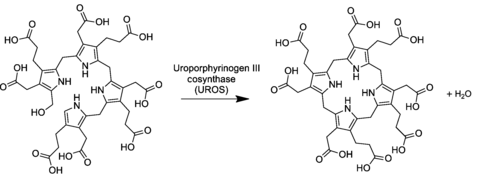
The conversion entails a reversal of the last
In the biosynthesis of hemes and chlorophylls, uroporphyrinogen III is converted into coproporphyrinogen III by the enzyme uroporphyrinogen III decarboxylase. In the biosynthesis of sirohemes, uroporphyrinogen III is converted by two methyl transferases to dihydrosirohydrochlorin, which is subsequently oxidized sirohydrochlorin, a precursor to the siroheme prosthetic group.
Medical significance
If uroporphyrinogen-III synthase is not present or inactive, the hydroxymethylbilane will spontaneously cyclise into the
See also
References
- S2CID 4177167.
- ISBN 978-0-470-04867-2.
- ^
