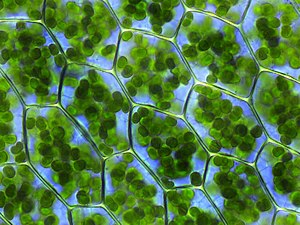
Chlorophyll
Chlorophyll is any of several related green
Chlorophylls absorb light most strongly in the blue portion of the electromagnetic spectrum as well as the red portion.[4] Conversely, it is a poor absorber of green and near-green portions of the spectrum. Hence chlorophyll-containing tissues appear green because green light, diffusively reflected by structures like cell walls, is less absorbed.[1] Two types of chlorophyll exist in the photosystems of green plants: chlorophyll a and b.[5]
History
Chlorophyll was first isolated and named by Joseph Bienaimé Caventou and Pierre Joseph Pelletier in 1817.[6] The presence of magnesium in chlorophyll was discovered in 1906,[7] and was the first detection of that element in living tissue.[8]
After initial work done by German chemist
Photosynthesis

Chlorophyll is vital for photosynthesis, which allows plants to absorb energy from light.[15]
Chlorophyll molecules are arranged in and around photosystems that are embedded in the thylakoid membranes of chloroplasts.[16] In these complexes, chlorophyll serves three functions:
- The function of the vast majority of chlorophyll (up to several hundred molecules per photosystem) is to absorb light.
- Having done so, these same centers execute their second function: The transfer of that energy by reaction centerof the photosystems.
- This specific pair performs the final function of chlorophylls: Charge separation, which produces the unbound protons (H+) and electrons (e−) that separately propel biosynthesis.
The two currently accepted photosystem units are
The function of the reaction center of chlorophyll is to absorb light energy and transfer it to other parts of the photosystem. The absorbed energy of the photon is transferred to an electron in a process called charge separation. The removal of the electron from the chlorophyll is an oxidation reaction. The chlorophyll donates the high energy electron to a series of molecular intermediates called an electron transport chain. The charged reaction center of chlorophyll (P680+) is then reduced back to its ground state by accepting an electron stripped from water. The electron that reduces P680+ ultimately comes from the oxidation of water into O2 and H+ through several intermediates. This reaction is how photosynthetic organisms such as plants produce O2 gas, and is the source for practically all the O2 in Earth's atmosphere. Photosystem I typically works in series with Photosystem II; thus the P700+ of Photosystem I is usually reduced as it accepts the electron, via many intermediates in the thylakoid membrane, by electrons coming, ultimately, from Photosystem II. Electron transfer reactions in the thylakoid membranes are complex, however, and the source of electrons used to reduce P700+ can vary.
The electron flow produced by the reaction center chlorophyll pigments is used to pump H+ ions across the thylakoid membrane, setting up a
Reaction center chlorophyll–protein complexes are capable of directly absorbing light and performing charge separation events without the assistance of other chlorophyll pigments, but the probability of that happening under a given light intensity is small. Thus, the other chlorophylls in the photosystem and antenna pigment proteins all cooperatively absorb and funnel light energy to the reaction center. Besides chlorophyll a, there are other pigments, called accessory pigments, which occur in these pigment–protein antenna complexes.
Chemical structure
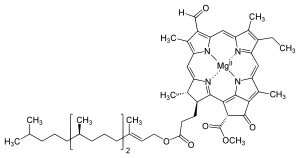
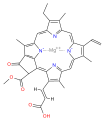
Several chlorophylls are known. All are defined as derivatives of the parent
The structures of chlorophylls are summarized below:[17][18]
| Chlorophyll a | Chlorophyll b | Chlorophyll c1
|
Chlorophyll c2
|
Chlorophyll d | Chlorophyll f[14] | |
|---|---|---|---|---|---|---|
Molecular formula
|
C55H72O5N4Mg | C55H70O6N4Mg | C35H30O5N4Mg | C35H28O5N4Mg | C54H70O6N4Mg | C55H70O6N4Mg |
| C2 group | −CH3 | −CH3 | −CH3 | −CH3 | −CH3 | −CHO |
| C3 group | −CH=CH2 | −CH=CH2 | −CH=CH2 | −CH=CH2 | −CHO
|
−CH=CH2 |
| C7 group | −CH3 | −CHO | −CH3 | −CH3 | −CH3 | −CH3 |
| C8 group | −CH2CH3 | −CH2CH3 | −CH2CH3 | −CH=CH2 | −CH2CH3 | −CH2CH3 |
| C17 group | −CH2CH2COO−Phytyl | −CH2CH2COO−Phytyl | −CH=CHCOOH | −CH=CHCOOH | −CH2CH2COO−Phytyl | −CH2CH2COO−Phytyl |
| C17−C18 bond | Single (chlorin) |
Single (chlorin) |
Double (porphyrin) |
Double (porphyrin) |
Single (chlorin) |
Single (chlorin) |
| Occurrence | Universal | Mostly plants | Various algae | Various algae | Cyanobacteria | Cyanobacteria |
- Structures of chlorophylls
-
chlorophyll a
-
chlorophyll b
-
chlorophyll c1
-
chlorophyll c2
-
chlorophyll d
-
chlorophyll f
Chlorophyll e is reserved for a pigment that has been extracted from algae in 1966 but not chemically described. Besides the lettered chlorophylls, a wide variety of sidechain modifications to the chlorophyll structures are known in the wild. For example, Prochlorococcus, a cyanobacterium, uses 8-vinyl Chl a and b.[19]
Measurement of chlorophyll content

Chlorophylls can be extracted from the protein into organic solvents.[20][21][22] In this way, the concentration of chlorophyll within a leaf can be estimated.[23] Methods also exist to separate chlorophyll a and chlorophyll b.
In
Ratio fluorescence emission can be used to measure chlorophyll content. By exciting chlorophyll a fluorescence at a lower wavelength, the ratio of chlorophyll fluorescence emission at 705±10 nm and 735±10 nm can provide a linear relationship of chlorophyll content when compared with chemical testing. The ratio F735/F700 provided a correlation value of r2 0.96 compared with chemical testing in the range from 41 mg m−2 up to 675 mg m−2. Gitelson also developed a formula for direct readout of chlorophyll content in mg m−2. The formula provided a reliable method of measuring chlorophyll content from 41 mg m−2 up to 675 mg m−2 with a correlation r2 value of 0.95.[27]
Biosynthesis
In some plants, chlorophyll is derived from
Chlorophyll synthase[31] is the enzyme that completes the biosynthesis of chlorophyll a:[32][33]- chlorophyllide a + phytyl diphosphate chlorophyll a + diphosphate
This conversion forms an ester of the carboxylic acid group in chlorophyllide a with the 20-carbon diterpene alcohol phytol. Chlorophyll b is made by the same enzyme acting on chlorophyllide b. The same is known for chlorophyll d and f, both made from corresponding chlorophyllides ultimately made from chlorophyllide a.[34]
In
Chlorophyll is bound to
Senescence and the chlorophyll cycle
The process of plant senescence involves the degradation of chlorophyll: for example the enzyme chlorophyllase (EC 3.1.1.14) hydrolyses the phytyl sidechain to reverse the reaction in which chlorophylls are biosynthesised from chlorophyllide a or b. Since chlorophyllide a can be converted to chlorophyllide b and the latter can be re-esterified to chlorophyll b, these processes allow cycling between chlorophylls a and b. Moreover, chlorophyll b can be directly reduced (via 71-hydroxychlorophyll a) back to chlorophyll a, completing the cycle.[37][38] In later stages of senescence, chlorophyllides are converted to a group of colourless tetrapyrroles known as nonfluorescent chlorophyll catabolites (NCC's) with the general structure:
These compounds have also been identified in ripening fruits and they give characteristic autumn colours to deciduous plants.[38][39]
Distribution
The chlorophyll maps show milligrams of chlorophyll per cubic meter of seawater each month. Places where chlorophyll amounts were very low, indicating very low numbers of
Culinary use
Synthetic chlorophyll is registered as a food additive colorant, and its E number is E140. Chefs use chlorophyll to color a variety of foods and beverages green, such as pasta and spirits. Absinthe gains its green color naturally from the chlorophyll introduced through the large variety of herbs used in its production.[41] Chlorophyll is not soluble in water, and it is first mixed with a small quantity of vegetable oil to obtain the desired solution.[citation needed]
Biological use
A 2002 study found that "leaves exposed to strong light contained degraded major
See also
- Bacteriochlorophyll, related compounds in phototrophic bacteria
- Chlorophyllin, a semi-synthetic derivative of chlorophyll
- Deep chlorophyll maximum
- Chlorophyll fluorescence, to measure plant stress
References
- ^ .
- ^ May P. "Chlorophyll". University of Bristol.
- ^ "chlorophyll". Online Etymology Dictionary.
- PMID 24642884.
- ^ Speer BR (1997). "Photosynthetic Pigments". UCMP Glossary (online). University of California Museum of Paleontology. Retrieved 2010-07-17.
- ^ See:
- .
- Pelletier PJ, Caventou JB (1817). "Notice sur la matière verte des feuilles" [Notice on the green material in leaves]. Journal de Pharmacie (in French). 3: 486–491. On p. 490, the authors propose a new name for chlorophyll. From p. 490: "Nous n'avons aucun droit pour nommer une substance connue depuis long-temps, et à l'histoire de laquelle nous n'avons ajouté que quelques faits ; cependant nous proposerons, sans y mettre aucune importance, le nom de chlorophyle, de chloros, couleur, et φύλλον, feuille : ce nom indiquerait le rôle qu'elle joue dans la nature." (We have no right to name a substance [that has been] known for a long time, and to whose story we have added only a few facts ; however, we will propose, without giving it any importance, the name chlorophyll, from chloros, color, and φύλλον, leaf : this name would indicate the role that it plays in nature.)
- .
From p. 49: "Das Hauptproduct der alkalischen Hydrolyse bilden tiefgrüne Alkalisalze. In ihnen liegen complexe Magnesiumverbindungen vor, die das Metall in einer gegen Alkali auch bei hoher Temperatur merkwürdig widerstandsfähigen Bindung enthalten." (Deep green alkali salts form the main product of alkali hydrolysis. In them, complex magnesium compounds are present, which contain the metal in a bond that is extraordinarily resistant to alkali even at high temperature.)
- ^ ISBN 978-952-10-4846-3.
- (PDF) from the original on 2011-04-10.
- S2CID 4262313.
- .
- ^ Jabr F (August 2010). "A New Form of Chlorophyll?". Scientific American.
- ^ "Infrared chlorophyll could boost solar cells". New Scientist. 19 August 2010. Retrieved 15 April 2012.
- ^ S2CID 206527174.
- ^ Carter JS (1996). "Photosynthesis". University of Cincinnati. Archived from the original on 2013-06-29.
- ^ "Unit 1.3. Photosynthetic cells". Essentials of Cell Biology. Nature. July 5, 2013.
{{cite book}}:|website=ignored (help) - ISBN 978-1-4020-4515-8.
- PMID 27498781.
- S2CID 149632511.
- .
- JSTOR 1539668.
- Napier University. Archived from the originalon April 14, 2008. Retrieved 2010-07-17.
- PMID 12975132.
- ISBN 978-0442006570.
- .
- ISBN 978-0-7923-6333-0.
- .
- PMID 11152419.
- PMID 7842850.
- PMID 12828371.
- S2CID 3099209.
- S2CID 21174896.
- S2CID 23808539.
- PMID 35406896.
- S2CID 4427639.
- ^
Meskauskiene R, Nater M, Goslings D, Kessler F, op den Camp R, Apel K (October 2001). "FLU: a negative regulator of chlorophyll biosynthesis in Arabidopsis thaliana". Proceedings of the National Academy of Sciences of the United States of America. 98 (22): 12826–31. PMID 11606728.
- ^ "Chlorophyll Cycle". IUBMB. 2011. Retrieved 2020-06-04.
- ^ PMID 16669755.
- PMID 17943948.
- ^ "Chlorophyll : Global Maps". Earthobservatory.nasa.gov. Retrieved 2 February 2014.
- ISBN 978-1860649202.
- PMID 12450406.