Vinyl iodide functional group
This article may be too technical for most readers to understand. (December 2013) |

In
Properties
Vinyl iodides are generally stable under

In cross-

Other applications

Besides using vinyl iodides as useful substrates in transition metal cross-
Methods of synthesis

Vinyl iodides are synthesized by methods such as
Synthesis from alkynes
The common and simplest approach to make vinyl iodide is addition of one equivalent
α-vinyl iodides
Introducing an α-vinyl iodide from a terminal position of an alkyne is a difficult step. in addition, the vinyl metal intermediate can be mildly

Another method doesn't involve hydrometalation but hydroiodation with I2/hydrophosphine binary system, which was developed by Ogawa's group.[12]

The hydroiodation proceeds by Markovnikov-type adduct, no reaction is observed without addition of hydrophoshine. In a plausible mechanism proposed by Ogawa's group, the hydrophosphine reacts with HI to form an intermediate complex that coordinate HI to do Markovnikov hydroiodation on the alkene. The advantage of this system is the conditions are mild, can tolerate wide range of functional groups.

β-vinyl iodides
They are generally more methods in making β-vinyl iodides versus α-vinyl iodides using
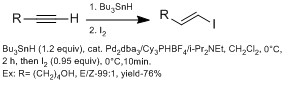
Z selective β-vinyl iodides are slightly more difficult to introduce than E-β-vinyl iodides, often requiring more than one step. Hydroalumination and hydroboration usually proceed by syn fashion, therefore selectively favors E geometry. The Oshima group have demonstrated using hydroindation with HInCl selectively favors Z geometry.[14] They suggested that the reaction proceeds by a radical mechanism. They predict that HInCl adds to alkyne by radical addition in a Z geometry. It does not isomerized to E geometry because of low reactivity of radical InCl2 with intermediate complex (no second addition). If second addition occurs then isomerization will occur through diindium intermediate. They confirm a radical mechanism in a mechanistic study with alkyne and alkene cyclization.

Substitution
Substitution is perhaps most useful method in introducing vinyl iodide into the molecule. Halogen-exchange can be useful since vinyl iodides are more reactivity than other

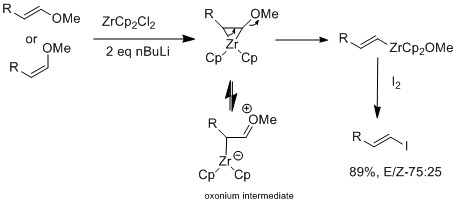
Halogen-exchange can also be done with zirconium derivatives that retain

The Marek group have further investigated using zirconium catalyst on E or Z

An interesting substitution reaction is vinyl boronic acid to vinyl iodide done by Brown's group.
Radical substitution of carboxylic acid to iodide is demonstrated by a modified Hunsdiecker reaction.[19] Homolytic cleavage of O-I bond generates CO2 and vinyl radical. Vinyl radical recombines with iodide radical to form vinyl iodide.
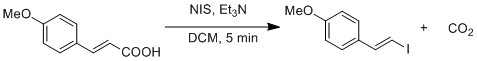
Iododesilylation
Iododesilylation is a substitution reaction of
The Kishi's group reported a mild preparation of vinyl iodide from vinyl silyl using NIS in mixture of acetonitrile and chloroacetonitrile.[20] They observed retention of olefin geometry in some vinyl silyl substrates while inversion in others. They reasoned that the R group's size had an effect on the geometry of the olefin. If the R group is small, the solvent acetonitrile can participate in the reaction leading to inversion of the olefin's geometry. If the R group is big, the solvent is unable to participate, leading to retention of olefin's geometry

Zakarian's group then decided to run the reaction in

Unfortunately, iododesilylation under those conditions (above) can potentially yield multiple byproducts in highly functionalized molecules with oxygen
Name reactions
Some famous vinyl iodide synthesis methods involve conversion of
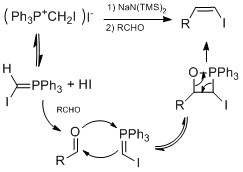
Below is example of employing both Takai olefination and Stork-Zhao olefination in total synthesis of (+)-3-(E)- and (+)-3-(Z)-Pinnatifidenyne.[29]

Elimination method
Vinyl iodides are rarely by made an elimination reaction of vicinal diiodide because it tends to decompose to alkene and iodide.[30] The Baker group have shown using decarboxylation, elimination can occur.[31]

See also
- List of functional groups
- Group contribution method
References
- ISSN 0022-328X.
- ^ ISBN 978-1-118-13750-5.
- ISBN 978-81-203-2441-1.
- ISSN 0001-4842.
- ISSN 0022-3832.
- ISSN 0040-4020.
- PMID 19476372.
- ^ ISSN 0022-3263.
- ISSN 1523-7060.
- ISSN 0022-3263.
- PMID 20698643.
- ISSN 1523-7060.
- ^ ISSN 0040-4020.
- ISSN 0022-3263.Takami, Kazuaki, et al. "Triethylborane-mediated hydrogallation and hydroindation: Novel access to organogalliums and organoindiums." The Journal of Organic Chemistry 68.17 (2003): 6627-6631.
- ISSN 0002-7863.
- ^ ISSN 0022-3263.
- ISSN 0009-2975.
- ISSN 0040-4039.
- ISSN 0022-3263.
- ISSN 0040-4039.
- ISSN 1523-7060.
- ISSN 1523-7060.
- ISSN 0368-1769.
- ISSN 0040-4020.
- ISSN 0002-7863.Danishefsky, Samuel J., et al. "Total synthesis of baccatin III and taxol." Journal of the American Chemical Society 118.12 (1996): 2843-2859
- ISSN 0002-7863.
- ISSN 0002-7863.
- ISSN 0040-4039.
- ISSN 0002-7863.
- ISBN 978-0-08-042323-4.
- ISSN 1364-5463.
