Crystallographic defects in diamond

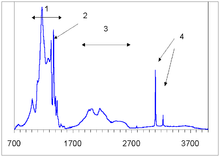
The defects can be detected by different types of
Labeling of diamond centers
There is a tradition in diamond spectroscopy to label a defect-induced spectrum by a numbered acronym (e.g. GR1). This tradition has been followed in general with some notable deviations, such as A, B and C centers. Many acronyms are confusing though:[2]
- Some symbols are too similar (e.g., 3H and H3).
- Accidentally, the same labels were given to different centers detected by EPR and optical techniques (e.g., N3 EPR center and N3 optical center have no relation).[3]
- Whereas some acronyms are logical, such as N3 (N for natural, i.e. observed in natural diamond) or H3 (H for heated, i.e. observed after irradiation and heating), many are not. In particular, there is no clear distinction between the meaning of labels GR (general radiation), R (radiation) and TR (type-II radiation).[2]
Defect symmetry
The symmetry of defects in crystals is described by the
The defect symmetry allows predicting many optical properties. For example, one-phonon (infrared) absorption in pure diamond lattice is forbidden because the lattice has an inversion center. However, introducing any defect (even "very symmetrical", such as N-N substitutional pair) breaks the crystal symmetry resulting in defect-induced infrared absorption, which is the most common tool to measure the defect concentrations in diamond.[2]
In synthetic diamond grown by the high-pressure high-temperature synthesis[5] or chemical vapor deposition,[6][7] defects with symmetry lower than tetrahedral align to the direction of the growth. Such alignment has also been observed in gallium arsenide[8] and thus is not unique to diamond.
Extrinsic defects
Various elemental analyses of diamond reveal a wide range of impurities. They mostly originate, however, from inclusions of foreign materials in diamond, which could be nanometer-small and invisible in an optical microscope. Also, virtually any element can be hammered into diamond by ion implantation. More essential are elements that can be introduced into the diamond lattice as isolated atoms (or small atomic clusters) during the diamond growth. By 2008, those elements are nitrogen, boron, hydrogen, silicon, phosphorus, nickel, cobalt and perhaps sulfur. Manganese[9] and tungsten[10] have been unambiguously detected in diamond, but they might originate from foreign inclusions. Detection of isolated iron in diamond[11] has later been re-interpreted in terms of micro-particles of ruby produced during the diamond synthesis.[12] Oxygen is believed to be a major impurity in diamond,[13] but it has not been spectroscopically identified in diamond yet.[citation needed] Two electron paramagnetic resonance centers (OK1 and N3) have been initially assigned to nitrogen–oxygen complexes, and later to titanium-related complexes.[14] However, the assignment is indirect and the corresponding concentrations are rather low (few parts per million).[15]
Nitrogen
The most common impurity in diamond is nitrogen, which can comprise up to 1% of a diamond by mass.[13] Previously, all lattice defects in diamond were thought to be the result of structural anomalies; later research revealed nitrogen to be present in most diamonds and in many different configurations. Most nitrogen enters the diamond lattice as a single atom (i.e. nitrogen-containing molecules dissociate before incorporation), however, molecular nitrogen incorporates into diamond as well.[16]
Absorption of light and other material properties of diamond are highly dependent upon nitrogen content and aggregation state. Although all aggregate configurations cause absorption in the infrared, diamonds containing aggregated nitrogen are usually colorless, i.e. have little absorption in the visible spectrum.[2] The four main nitrogen forms are as follows:

C-nitrogen center
The C center corresponds to electrically neutral single substitutional nitrogen
The C center produces a characteristic infrared absorption spectrum with a sharp peak at 1344 cm−1 and a broader feature at 1130 cm−1. Absorption at those peaks is routinely used to measure the concentration of single nitrogen.[20] Another proposed way, using the UV absorption at ~260 nm, has later been discarded as unreliable.[19]
Acceptor defects in diamond ionize the fifth nitrogen electron in the C center converting it into C+ center. The latter has a characteristic IR absorption spectrum with a sharp peak at 1332 cm−1 and broader and weaker peaks at 1115, 1046 and 950 cm−1.[21]

A-nitrogen center
The A center is probably the most common defect in natural diamonds. It consists of a neutral nearest-neighbor pair of nitrogen atoms substituting for the carbon atoms. The A center produces UV absorption threshold at ~4 eV (310 nm, i.e. invisible to eye) and thus causes no coloration. Diamond containing nitrogen predominantly in the A form as classed as type IaA.[22]
The A center is
The A center shows an IR absorption spectrum with no sharp features, which is distinctly different from that of the C or B centers. Its strongest peak at 1282 cm−1 is routinely used to estimate the nitrogen concentration in the A form.[24]
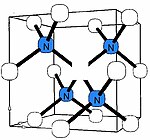
B-nitrogen center
There is a general consensus that B center (sometimes called B1) consists of a carbon vacancy surrounded by four nitrogen atoms substituting for carbon atoms.[1][2][25] This model is consistent with other experimental results, but there is no direct spectroscopic data corroborating it. Diamonds where most nitrogen forms B centers are rare and are classed as type IaB; most gem diamonds contain a mixture of A and B centers, together with N3 centers.
Similar to the A centers, B centers do not induce color, and no UV or visible absorption can be attributed to the B centers. Early assignment of the N9 absorption system to the B center have been disproven later.[26] The B center has a characteristic IR absorption spectrum (see the infrared absorption picture above) with a sharp peak at 1332 cm−1 and a broader feature at 1280 cm−1. The latter is routinely used to estimate the nitrogen concentration in the B form.[27]
Many optical peaks in diamond accidentally have similar spectral positions, which causes much confusion among gemologists. Spectroscopists use the whole spectrum rather than one peak for defect identification and consider the history of the growth and processing of individual diamond.[1][2][25]

N3 nitrogen center
The N3 center consists of three nitrogen atoms surrounding a vacancy. Its concentration is always just a fraction of the A and B centers.[28] The N3 center is paramagnetic, so its structure is well justified from the analysis of the EPR spectrum P2.[3] This defect produces a characteristic absorption and luminescence line at 415 nm and thus does not induce color on its own. However, the N3 center is always accompanied by the N2 center, having an absorption line at 478 nm (and no luminescence).[29] As a result, diamonds rich in N3/N2 centers are yellow in color.
Boron
Diamonds containing boron as a substitutional impurity are termed type IIb. Only one percent of natural diamonds are of this type, and most are blue to grey.
Boron-doped diamonds transmit light down to ~250 nm and absorb some red and infrared light (hence the blue color); they may phosphoresce blue after exposure to shortwave ultraviolet light.[31] Apart from optical absorption, boron acceptors have been detected by electron paramagnetic resonance.[32]
Phosphorus
Phosphorus could be intentionally introduced into diamond grown by chemical vapor deposition (CVD) at concentrations up to ~0.01%.
Hydrogen
Hydrogen is one of the most technological important impurities in semiconductors, including diamond. Hydrogen-related defects are very different in natural diamond and in synthetic diamond films. Those films are produced by various chemical vapor deposition (CVD) techniques in an atmosphere rich in hydrogen (typical hydrogen/carbon ratio >100), under strong bombardment of growing diamond by the plasma ions. As a result, CVD diamond is always rich in hydrogen and lattice vacancies. In polycrystalline films, much of the hydrogen may be located at the boundaries between diamond 'grains', or in non-diamond carbon inclusions. Within the diamond lattice itself, hydrogen-vacancy[37] and hydrogen-nitrogen-vacancy[38] complexes have been identified in negative charge states by electron paramagnetic resonance. In addition, numerous hydrogen-related IR absorption peaks are documented.[39]
It is experimentally demonstrated that hydrogen passivates electrically active boron[40] and phosphorus[41] impurities. As a result of such passivation, shallow donor centers are presumably produced.[42]
In natural diamonds, several hydrogen-related IR absorption peaks are commonly observed; the strongest ones are located at 1405, 3107 and 3237 cm−1 (see IR absorption figure above). The microscopic structure of the corresponding defects is yet unknown and it is not even certain whether or not those defects originate in diamond or in foreign inclusions. Gray color in some diamonds from the Argyle mine in Australia is often associated with those hydrogen defects, but again, this assignment is yet unproven.[43]
Nickel, cobalt and chromium
When diamonds are grown by the high-pressure high-temperature technique, nickel, cobalt, chromium or some other metals are usually added into the growth medium to facilitate catalytically the conversion of graphite into diamond. As a result, metallic inclusions are formed. Besides, isolated nickel and cobalt atoms incorporate into diamond lattice, as demonstrated through characteristic hyperfine structure in electron paramagnetic resonance, optical absorption and photoluminescence spectra,[44] and the concentration of isolated nickel can reach 0.01%.[45] This fact is by all means unusual considering the large difference in size between carbon and transition metal atoms and the superior rigidity of the diamond lattice.[2][45]
Numerous Ni-related defects have been detected by electron paramagnetic resonance,[5][46] optical absorption and photoluminescence,[5][46] both in synthetic and natural diamonds.[43] Three major structures can be distinguished: substitutional Ni,[47] nickel-vacancy[48] and nickel-vacancy complex decorated by one or more substitutional nitrogen atoms.[46] The "nickel-vacancy" structure, also called "semi-divacancy" is specific for most large impurities in diamond and silicon (e.g., tin in silicon[49]). Its production mechanism is generally accepted as follows: large nickel atom incorporates substitutionally, then expels a nearby carbon (creating a neighboring vacancy), and shifts in-between the two sites.
Although the physical and chemical properties of cobalt and nickel are rather similar, the concentrations of isolated cobalt in diamond are much smaller than those of nickel (parts per billion range). Several defects related to isolated cobalt have been detected by electron paramagnetic resonance[50] and photoluminescence,[5][51] but their structure is yet unknown.
A chromium-related optical center has been detected in diamond after ion implantation and subsequent annealing.[52][53]
Silicon, germanium, tin and lead

Silicon is a common impurity in diamond films grown by chemical vapor deposition and it originates either from silicon substrate or from silica windows or walls of the CVD reactor. It was also observed in natural diamonds in dispersed form.[54] Isolated silicon defects have been detected in diamond lattice through the sharp optical absorption peak at 738 nm[55] and electron paramagnetic resonance.[56] Similar to other large impurities, the major form of silicon in diamond has been identified with a Si-vacancy complex (semi-divacancy site).[56] This center is a deep donor having an ionization energy of 2 eV, and thus again is unsuitable for electronic applications.[57]
Germanium, tin and lead are normally absent in diamond, but they can be introduced during the growth or by subsequent ion implantation. Those impurities can be detected optically via the
Similar to N-V centers, Si-V, Ge-V, Sn-V and Pb-V complexes all have potential applications in quantum computing.[62][60]
Sulfur
Around the year 2000, there was a wave of attempts to dope synthetic CVD diamond films by sulfur aiming at n-type conductivity with low activation energy. Successful reports have been published,[63] but then dismissed[64] as the conductivity was rendered p-type instead of n-type and associated not with sulfur, but with residual boron, which is a highly efficient p-type dopant in diamond.
So far (2009), there is only one reliable evidence (through hyperfine interaction structure in electron paramagnetic resonance) for isolated sulfur defects in diamond. The corresponding center called W31 has been observed in natural type-Ib diamonds in small concentrations (parts per million). It was assigned to a sulfur-vacancy complex – again, as in case of nickel and silicon, a semi-divacancy site.[65]
Intrinsic defects
The easiest way to produce intrinsic defects in diamond is by displacing carbon atoms through irradiation with high-energy particles, such as alpha (helium), beta (electrons) or gamma particles, protons, neutrons, ions, etc. The irradiation can occur in the laboratory or in nature (see
Vacancies and interstitials can also be produced in diamond by plastic deformation, though in much smaller concentrations.
Isolated carbon interstitial

Isolated interstitial has never been observed in diamond and is considered unstable. Its interaction with a regular carbon lattice atom produces a "split-interstitial", a defect where two carbon atoms share a lattice site and are covalently bonded with the carbon neighbors. This defect has been thoroughly characterized by electron paramagnetic resonance (R2 center)[67] and optical absorption,[68] and unlike most other defects in diamond, it does not produce photoluminescence.
Interstitial complexes

The isolated split-interstitial moves through the diamond crystal during irradiation. When it meets other interstitials it aggregates into larger complexes of two and three split-interstitials, identified by electron paramagnetic resonance (R1 and O3 centers),[69][70] optical absorption and photoluminescence.[71]
Vacancy-interstitial complexes
Most high-energy particles, beside displacing carbon atom from the lattice site, also pass it enough surplus energy for a rapid migration through the lattice. However, when relatively gentle gamma irradiation is used, this extra energy is minimal. Thus the interstitials remain near the original vacancies and form vacancy-interstitials pairs identified through optical absorption.[71][72][73]
Vacancy-di-interstitial pairs have been also produced, though by electron irradiation and through a different mechanism:[74] Individual interstitials migrate during the irradiation and aggregate to form di-interstitials; this process occurs preferentially near the lattice vacancies.
Isolated vacancy

Isolated
The vacancy behaves as a deep electron donor/acceptor, whose electronic properties depend on the charge state. The energy level for the +/0 states is at 0.6 eV and for the 0/- states is at 2.5 eV above the
Multivacancy complexes
Upon annealing of pure diamond at ~700 °C, vacancies migrate and form divacancies, characterized by optical absorption and electron paramagnetic resonance.[76] Similar to single interstitials, divacancies do not produce photoluminescence. Divacancies, in turn, anneal out at ~900 °C creating multivacancy chains detected by EPR[77] and presumably hexavacancy rings. The latter should be invisible to most spectroscopies, and indeed, they have not been detected thus far.[77] Annealing of vacancies changes diamond color from green to yellow-brown. Similar mechanism (vacancy aggregation) is also believed to cause brown color of plastically deformed natural diamonds.[78]
Dislocations
Dislocations are the most common structural defect in natural diamond. The two major types of dislocations are the glide set, in which bonds break between layers of atoms with different indices (those not lying directly above each other) and the shuffle set, in which the breaks occur between atoms of the same index. The dislocations produce dangling bonds which introduce energy levels into the band gap, enabling the absorption of light.[79] Broadband blue photoluminescence has been reliably identified with dislocations by direct observation in an electron microscope, however, it was noted that not all dislocations are luminescent, and there is no correlation between the dislocation type and the parameters of the emission.[80]
Platelets

Most natural diamonds contain extended planar defects in the <100> lattice planes, which are called "platelets". Their size ranges from nanometers to many micrometers, and large ones are easily observed in an
Platelets produce sharp absorption peaks at 1359–1375 and 330 cm−1 in IR absorption spectra; remarkably, the position of the first peak depends on the platelet size.[81][84] As with dislocations, a broad photoluminescence centered at ~1000 nm was associated with platelets by direct observation in an electron microscope. By studying this luminescence, it was deduced that platelets have a "bandgap" of ~1.7 eV.[85]
Voidites

Voidites are
Interaction between intrinsic and extrinsic defects
Extrinsic and intrinsic defects can interact producing new defect complexes. Such interaction usually occurs if a diamond containing extrinsic defects (impurities) is either plastically deformed or is irradiated and annealed.

Most important is the interaction of vacancies and interstitials with nitrogen. Carbon interstitials react with substitutional nitrogen producing a bond-centered nitrogen interstitial showing strong IR absorption at 1450 cm−1.[88] Vacancies are efficiently trapped by the A, B and C nitrogen centers. The trapping rate is the highest for the C centers, 8 times lower for the A centers and 30 times lower for the B centers.[89] The C center (single nitrogen) by trapping a vacancy forms the famous nitrogen-vacancy center, which can be neutral or negatively charged;[90][91] the negatively charged state has potential applications in quantum computing. A and B centers upon trapping a vacancy create corresponding 2N-V (H3[92] and H2[93] centers, where H2 is simply a negatively charged H3 center[94]) and the neutral 4N-2V (H4 center[95]). The H2, H3 and H4 centers are important because they are present in many natural diamonds and their optical absorption can be strong enough to alter the diamond color (H3 or H4 – yellow, H2 – green).
Boron interacts with carbon interstitials forming a neutral boron–interstitial complex with a sharp optical absorption at 0.552 eV (2250 nm).[75] No evidence is known so far (2009) for complexes of boron and vacancy.[25]
In contrast, silicon does react with vacancies, creating the described above optical absorption at 738 nm.[96] The assumed mechanism is trapping of migrating vacancy by substitutional silicon resulting in the Si-V (semi-divacancy) configuration.[97]
A similar mechanism is expected for nickel, for which both substitutional and semi-divacancy configurations are reliably identified (see subsection "nickel and cobalt" above). In an unpublished study, diamonds rich in substitutional nickel were electron irradiated and annealed, with following careful optical measurements performed after each annealing step, but no evidence for creation or enhancement of Ni-vacancy centers was obtained.[48]
See also
- Chemical vapor deposition of diamond
- Crystallographic defect
- Diamond color
- Diamond enhancement
- Gemstone irradiation
- Material properties of diamond
- Nitrogen-vacancy center
- Synthetic diamond
References
- ^ .
- ^ (PDF) from the original on July 23, 2018. Retrieved February 12, 2019.
- ^ .
- ISBN 978-3-540-66582-3.
- ^ S2CID 250836558.
- S2CID 118609894.
- .
- doi:10.1063/1.116043.
- .
- PMID 9985239.
- S2CID 250871851.
- S2CID 250752181.
- ^ .
- from the original on June 30, 2023. Retrieved November 24, 2020.
- S2CID 250823139.
- S2CID 250888444.
- .
- ISBN 0-87311-016-1, p. 191
- ^ S2CID 250734213.
- ^ I. Kiflawi et al. "Infrared-absorption by the single nitrogen and a defect centers in diamond" Philos. Mag. B 69 (1994) 1141
- S2CID 250841408.
- .
- PMID 9975922.
- .
- ^ .
- .
- .
- ISBN 0-7198-0261-X
- S2CID 98179513.
- ISBN 0-7506-3173-2, p. 52
- ^ S2CID 202574625.
- .
- ^ S2CID 96415838.
- .
- .
- S2CID 10675358.
- PMID 15089622.
- PMID 12786024.
- doi:10.1063/1.113126.
- .
- .
- S2CID 21797000.
- ^ .
- .
- ^ .
- ^ S2CID 250759333.
- PMID 9994206.
- ^ .
- .
- .
- doi:10.1063/1.361744.
- S2CID 56165312.
- S2CID 54887620.
- .
- PMID 9978673.
- ^ .
- .
- .
- PMID 26250337.
- ^ Wikidata Q105746117.
- S2CID 206305034.
- S2CID 53959613.
- .
- doi:10.1063/1.125885.
- .
- .
- .
- .
- PMID 9984317.
- .
- ^ .
- ^ S2CID 97742332.
- .
- .
- ^ S2CID 2988243.
- .
- ^ .
- .
- ^ Kolodzie, A.T. and Bleloch, A.L. Investigation of band gap energy states at dislocations in natural diamond. Cavendish Laboratory, University of Cambridge; Cambridge, England.
- S2CID 120959202.
- ^ (PDF) from the original on February 12, 2019. Retrieved February 12, 2019.
- S2CID 4277090.
- .
- from the original on June 30, 2023. Retrieved August 11, 2019.
- S2CID 138243923.
- .
- .
- PMID 9985801.
- S2CID 250804678.
- S2CID 250820432.
- PMID 9982752.
- S2CID 93034755.
- S2CID 250824280.
- S2CID 250878505.
- S2CID 98842822.
- .
- PMID 10062116.


