Nitrogen-vacancy center

The nitrogen-vacancy center (N-V center or NV center) is one of numerous photoluminescent point defects in diamond. Its most explored and useful properties include its spin-dependent photoluminescence (which enables measurement of the electronic spin state using optically detected magnetic resonance), and its relatively long (millisecond) spin coherence at room temperature.[1] The NV center energy levels are modified by magnetic fields,[2] electric fields,[3] temperature,[4] and strain,[5] which allow it to serve as a sensor of a variety of physical phenomena. Its atomic size and spin properties can form the basis for useful quantum sensors.[6] It has also been explored for applications in quantum computing (e.g. for entanglement generation[7]), quantum simulation,[8] and spintronics.[9]
Structure
The nitrogen-vacancy center is a point defect in the diamond lattice. It consists of a nearest-neighbor pair of a nitrogen atom, which substitutes for a carbon atom, and a lattice vacancy.
Two charge states of this defect, neutral NV0 and negative NV−, are known from
The NV0 thus has one unpaired electron and is paramagnetic. However, despite extensive efforts, electron paramagnetic resonance signals from NV0 avoided detection for decades until 2008. Optical excitation is required to bring the NV0 defect into the EPR-detectable excited state; the signals from the ground state are presumably too broad for EPR detection.[18]
The NV0 centers can be converted into NV− by changing the
In the negative charge state NV−, an extra electron is located at the vacancy site forming a spin S=1 pair with one of the vacancy electrons. As in NV0, the vacancy electrons are "exchanging roles" preserving the overall trigonal symmetry. This NV− state is what is commonly, and somewhat incorrectly, called "the nitrogen-vacancy center". The neutral state is not generally used for quantum technology.
The NV centers are randomly oriented within a diamond crystal. Ion implantation techniques can enable their artificial creation in predetermined positions.[19]
Production
Nitrogen-vacancy centers are typically produced from single substitutional nitrogen centers (called C or P1 centers in diamond literature) by irradiation followed by annealing at temperatures above 700 °C.[11] A wide range of high-energy particles is suitable for such irradiation, including electrons, protons, neutrons, ions, and gamma photons. Irradiation produces lattice vacancies, which are a part of NV centers. Those vacancies are immobile at room temperature, and annealing is required to move them. Single substitutional nitrogen produces strain in the diamond lattice;[20] it therefore efficiently captures moving vacancies,[21] producing the NV centers.

During chemical vapor deposition of diamond, a small fraction of single substitutional nitrogen impurity (typically <0.5%) traps vacancies generated as a result of the plasma synthesis. Such nitrogen-vacancy centers are preferentially aligned to the growth direction.[23][24] Delta doping of nitrogen during CVD growth can be used to create two-dimensional ensembles of NV centers near the diamond surface for enhanced sensing[25] or simulation.[26]
Diamond is notorious for having a relatively large lattice strain. Strain splits and shifts optical transitions from individual centers resulting in broad lines in the ensembles of centers.[11][27] Special care is taken to produce extremely sharp NV lines (line width ~10 MHz)[28] required for most experiments: high-quality, pure natural or better synthetic diamonds (type IIa) are selected. Many of them already have sufficient concentrations of grown-in NV centers and are suitable for applications. If not, they are irradiated by high-energy particles and annealed. Selection of a certain irradiation dose allows tuning the concentration of produced NV centers such that individual NV centers are separated by micrometre-large distances. Then, individual NV centers can be studied with standard optical microscopes or, better, near-field scanning optical microscopes having sub-micrometre resolution.[17][29]
Energy level structure
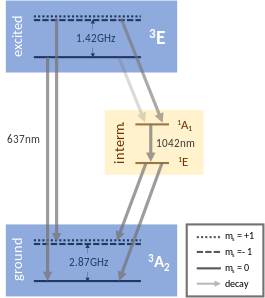
The NV center has a ground-state triplet (3A), an excited-state triplet (3E) and two intermediate-state singlets (1A and 1E).[note 1][33][34] Both 3A and 3E contain ms = ±1 spin states, in which the two electron spins are aligned (either up, such that ms = +1 or down, such that ms = -1), and an ms = 0 spin state where the electron spins are antiparallel. Due to the magnetic interaction, the energy of the ms = ±1 states is higher than that of the ms = 0 state. 1A and 1E only contain a spin state singlet each with ms = 0.
If an external magnetic field is applied along the defect axis (the axis which aligns with the nitrogen atom and the vacancy) of the NV center, it does not affect the ms = 0 states, but it splits the ms = ±1 levels (Zeeman effect). Similarly the following other properties of the environment influence the energy level diagram :
- Amplitude and orientation of a static magnetic field splits the ms = ±1 levels in the ground and excited states.
- Amplitude and orientation of have a much smaller but also more complex effects on the different levels.
- Continuous-wave microwave radiation (applied in resonance with the transition between ms = 0 and (one of the) ms = ±1 states) changes the population of the sublevels within the ground and excited state.[36]
- A tunable laser can selectively excite certain sublevels of the ground and excited states.[36][37]
- Surrounding spins and spin–orbit interaction will modulate the magnetic field experienced by the NV center.
- Temperature and pressure affect different parts of the spectrum including the shift between ground and excited states.
The above-described energy structure[note 2] is by no means exceptional for a defect in diamond or other semiconductor.[38] It was not this structure alone, but a combination of several favorable factors (previous knowledge, easy production, biocompatibility, simple initialisation, use at room temperature etc.) which suggested the use of the NV center as a qubit and quantum sensor.
Optical properties

NV centers emit bright red light (3E→3A transitions), if excited off-resonantly by visible green light (3A →3E transitions). This can be done with convenient light sources such as
As the relaxation time from the excited state is small (~10
An important property of the luminescence from individual NV centers is its high temporal stability. Whereas many single-molecular emitters bleach (i.e. change their charge state and become dark) after emission of 106–108 photons, bleaching is unlikely for NV centers at room temperature.[42][29] Strong laser illumination, however, may also convert some NV− into NV0 centers.[13]
Because of these properties, the ideal technique to address the NV centers is confocal microscopy, both at room temperature and at low temperature.
State manipulation

Optical spin manipulation
Optical transitions must preserve the total spin and occur only between levels of the same total spin. Specifically, transitions between the ground and excited states (with equal spin) can be induced using a green laser with a wavelength of 546 nm. Transitions 3E→1A and 1E→3A are non-radiative, while 1A →1E has both a non-radiative and infrared decay path.
The diagram on the right shows the multi-electronic states of the NV center labeled according to their symmetry (E or A) and their spin state (3 for a triplet (S=1) and 1 for a singlet (S=0)). There are two triplet states and two intermediate singlet states.[47]
Spin-state initialisation
An important property of the non-radiative transition between 3E and 1A is that it is stronger for ms = ±1 and weaker for ms = 0. This provides the basis a very useful manipulation strategy, which is called spin state initialisation (or optical spin-polarization). To understand the process, first consider an off-resonance excitation which has a higher frequency (typically 2.32 eV (532 nm)) than the frequencies of all transitions and thus lies in the vibronic bands for all transitions. By using a pulse of this wavelength, one can excite all spin states from 3A to 3E. An NV center in the ground state with ms = 0 will be excited to the corresponding excited state with ms = 0 due to the conservation of spin. Afterwards it decays back to its original state. For a ground state with ms = ±1, the situation is different. After the excitation, it has a relatively high probability to decay into the intermediate state 1A by non-radiative transition[note 3][48] and further into the ground state with ms = 0. After many cycles, the state of the NV center (independently of whether it started in ms = 0 or ms = ±1) will end up in the ms = 0 ground state. This process can be used to initialize the quantum state of a qubit for quantum information processing or quantum sensing.
Sometimes the polarisability of the NV center is explained by the claim that the transition from 1E to the ground state with ms = ±1 is small, compared to the transition to ms = 0. However, it has been shown that the comparatively low decay probability for ms = 0 states w.r.t. ms = ±1 states into 1A is enough to explain the polarization.[49]
Effects of external fields
Microwave spin manipulation
The energy difference between the ms = 0 and ms = ±1 states corresponds to the
Influence of external factors
If a magnetic field is oriented along the defect axis it leads to Zeeman splitting separating the ms = +1 from the ms = -1 states. This technique is used to lift the degeneracy and use only two of the spin states (usually the ground states with ms = -1 and ms = 0) as a qubit. Population can then be transferred between them using a microwave field. In the specific instance that the magnetic field reaches 1027 G (or 508 G) then the ms = –1 and ms = 0 states in the ground (or excited) state become equal in energy (Ground/Excited State Level Anticrossing). The following strong interaction results in so-called spin polarization, which strongly affects the intensity of optical absorption and luminescence transitions involving those states.[32]
Importantly, this splitting can be modulated by applying an external electric field,[35][36] in a similar fashion to the magnetic field mechanism outlined above, though the physics of the splitting is somewhat more complex. Nevertheless, an important practical outcome is that the intensity and position of the luminescence lines is modulated. Strain has a similar effect on the NV center as electric fields.[60]
There is an additional splitting of the ms = ±1 energy levels, which originates from the hyperfine interaction between surrounding nuclear spins and the NV center. These nuclear spins create magnetic and electric fields of their own leading to further distortions of the NV spectrum (see nuclear Zeeman and quadrupole interaction). Also the NV center's own spin–orbit interaction and orbital degeneracy leads to additional level splitting in the excited 3E state.
Temperature and pressure directly influence the zero-field term of the NV center leading to a shift between the ground and excited state levels.
The Hamiltonian, a quantum mechanical equation describing the dynamics of a system, which shows the influence of different factors on the NV center can be found below.
Although it can be challenging, all of these effects are measurable, making the NV center a perfect candidate for a quantum sensor.[56]
Charge state manipulation
It is also possible to switch the charge state of the NV center (i.e. between NV−, NV+ and NV0) by applying a gate voltage.[61]
Applications
The spectral shape and intensity of the optical signals from the NV− centers are sensitive to external perturbation, such as temperature, strain, electric and magnetic field. However, the use of spectral shape for sensing those perturbation is impractical, as the diamond would have to be cooled to cryogenic temperatures to sharpen the NV− signals. A more realistic approach is to use luminescence intensity (rather than lineshape), which exhibits a sharp resonance when a microwave frequency is applied to diamond that matches the splitting of the ground-state levels. The resulting optically detected magnetic resonance signals are sharp even at room temperature, and can be used in miniature sensors. Such sensors can detect magnetic fields of a few nanotesla[63] or electric fields of about 10 V/cm[64] at kilohertz frequencies after 100 seconds of averaging. This sensitivity allows detecting a magnetic or electric field produced by a single electron located tens of nanometers away from an NV− center.
Using the same mechanism, the NV− centers were employed in
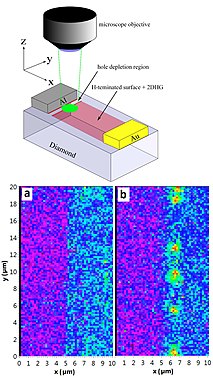
Because the NV center is sensitive to magnetic fields, it is being actively used in scanning probe measurements to study myriad condensed matter phenomena both through measuring a spatially varying magnetic field or inferring local currents in a device.[65][66][67][68][69]
Another possible use of the NV− centers is as a detector to measure the full mechanical stress tensor in the bulk of the crystal. For this application, the stress-induced splitting of the zero-phonon-line is exploited, and its polarization properties.[70] A robust frequency-modulated radio receiver using the electron-spin-dependent photoluminescence that operated up to 350 °C demonstrates the possibility for use in extreme conditions.[71]
In addition to the quantum optical applications, luminescence from the NV− centers can be applied for imaging biological processes, such as fluid flow in living cells.[72][73] This application relies on good compatibility of diamond nano-particles with the living cells and on favorable properties of photoluminescence from the NV− centers (strong intensity, easy excitation and detection, temporal stability, etc.). Compared with large single-crystal diamonds, nanodiamonds are cheap (about 1 USD per gram) and available from various suppliers. NV− centers are produced in diamond powders with sub-micrometre particle size using the standard process of irradiation and annealing described above. Due to the relatively small size of nanodiamond, NV centers can be produced by irradiating nanodiamond of 100 nm or less with medium energy H+ beam. This method reduces the required ion dose and reaction, making it possible to mass produce fluorescent nanodiamonds in ordinary laboratory.[74] Fluorescent nanodiamond produced with such method is bright and photostable, making it excellent for long-term, three dimensional tracking of single particle in living cell.[75] Those nanodiamonds are introduced in a cell, and their luminescence is monitored using a standard fluorescence microscope.[76]
Stimulated emission from the NV− center has been demonstrated, though it could be achieved only from the phonon side-band (i.e. broadband light) and not from the ZPL. For this purpose, the center has to be excited at a wavelength longer than ~650 nm, as higher-energy excitation ionizes the center.[77]
The first continuous-wave room-temperature maser has been demonstrated.[78][79] It used 532-nm pumped NV− centers held within a high Purcell factor microwave cavity and an external magnetic field of 4300 G. Continuous maser oscillation generated a coherent signal at ~9.2 GHz.
The NV center can have a very long spin
Historical remarks
The microscopic model and most optical properties of ensembles of the NV− centers have been firmly established in the 1970s based on the optical measurements combined with uniaxial stress[11] and on the electron paramagnetic resonance.[14][15] However, a minor error in EPR results (it was assumed that illumination is required to observe NV− EPR signals) resulted in the incorrect multiplicity assignments in the energy level structure. In 1991 it was shown that EPR can be observed without illumination,[16] which established the energy level scheme shown above. The magnetic splitting in the excited state has been measured only recently.[32]
The characterization of single NV− centers has become a very competitive field nowadays, with many dozens of papers published in the most prestigious scientific journals. One of the first results was reported back in 1997.[17] In that paper, it was demonstrated that the fluorescence of single NV− centers can be detected by room-temperature fluorescence microscopy and that the defect shows perfect photostability. Also one of the outstanding properties of the NV center was demonstrated, namely room-temperature optically detected magnetic resonance.
See also
Notes
- ^ Group theory results are used to take into account the symmetry of the diamond crystal, and so the symmetry of the NV itself. Followingly, the energy levels are labeled according to group theory, and in particular are labelled after the irreducible representations of the C3V symmetry group of the defect center, A1, A2, and E. The "3" in 3A2 and 3E as well as the "1" in 1A1 and 1E represent the number of allowable ms spin states, or the spin multiplicity, which range from –S to S for a total of 2S+1 possible states. If S = 1, ms can be −1, 0, or 1.
- ^ The energy level structure of the NV center was established by combining optically detected magnetic resonance (ODMR), electron paramagnetic resonance (EPR) and theoretical results, as shown in the figure. In particular, several theoretical works have been done, using the Linear Combination of Atomic Orbitals (LCAO) approach,[citation needed] to build the electronic orbitals to describe the possible quantum states, looking at the NV center as a molecule.
- ^ This is a phenomenon called intersystem crossing (ISC). It happens at an appreciable rate because the energy curve in function of the position of the atoms for the excited ms = ±1 state intersects the curve for the 1A state. Therefore, for some instant during the vibrational relaxation that the ions undergo after the excitement, it is possible for the spin to flip with little or no energy required in the transition.
References
- S2CID 5055366.
- PMID 18833275.
- S2CID 119287960.
- PMID 23903748.
- S2CID 16820460.
- S2CID 2555443.
- S2CID 4383784.
- PMID 28277511.
- S2CID 206545890.
- ^ PMID 26177799.
- ^ a b c d
Davies, G.; Hamer, M. F. (1976). "Optical Studies of the 1.945 eV Vibronic Band in Diamond". S2CID 93303167.
- ^
Mita, Y. (1996). "Change of absorption spectra in type-Ib diamond with heavy neutron irradiation". Physical Review B. 53 (17): 11360–11364. PMID 9982752.
- ^ a b
Iakoubovskii, K.; Adriaenssens, G. J.; Nesladek, M. (2000). "Photochromism of vacancy-related centres in diamond" (PDF). S2CID 250820432.
- ^ a b
Loubser, J. H. N.; van Wyk, J. A. (1977). "Electron Spin Resonance in Annealed Type 1b Diamond". Diamond Research. 11: 4–7. ISSN 0070-4679.
- ^ a b
Loubser, J. H. N.; van Wyk, J. A. (1978). "Electron spin resonance in the study of diamond". S2CID 250898303.
- ^ a b
Redman, D.; Brown, S.; Sands, R.; Rand, S. (1991). "Spin dynamics and electronic states of N-V centers in diamond by EPR and four-wave-mixing spectroscopy". PMID 10044729.
- ^ a b c Gruber, A.; et al. (1997). "Scanning Confocal Optical Microscopy and Magnetic Resonance on Single Defect Centers" (PDF). Science. 276 (5321): 2012–2014. .
- ^
Felton, S.; et al. (2008). "Electron paramagnetic resonance studies of the neutral nitrogen vacancy in diamond". S2CID 122354274.
- ^
Awschalom, D. D.; Epstein, R.; Hanson, R. (2007). "Diamond Age of Spintronics". PMID 17926759.
- ^
Lang, A. R.; et al. (1991). "On the Dilatation of Synthetic Type Ib Diamond by Substitutional Nitrogen Impurity". S2CID 54190787.
- ^
Iakoubovskii, K.; Adriaenssens, G. J. (2001). "Trapping of vacancies by defects in diamond". Journal of Physics: Condensed Matter. 13 (26): 6015. S2CID 250804678.
- S2CID 207968005.
- S2CID 118609894.
- S2CID 3283919.
- S2CID 253370730.
- PMID 37323805.
- S2CID 119422670.
- ^
Tamarat, Ph.; et al. (2006). "Stark Shift Control of Single Optical Centers in Diamond". S2CID 33870769.
- ^ a b
Kuhn, S.; et al. (2001). "Diamond colour centres as a nanoscopic light source for scanning near-field optical microscopy". S2CID 26077916.
- ISSN 0070-4679.
- S2CID 250898303.
- ^ S2CID 24822943.
- .
- S2CID 202236167.
- ^ S2CID 33870769.
- ^ S2CID 37554428.
- S2CID 14264923.
- S2CID 37867189.
- S2CID 97620201.
- .
- .
- .
- .
- S2CID 43745993.
- S2CID 42329227.
- S2CID 119113089.
- S2CID 119113089.
- .
- S2CID 55207459.
- S2CID 5055366.
- S2CID 20697722.[permanent dead link]
- S2CID 18853275.
- PMID 18352594.
- PMID 14995873.[permanent dead link]
- S2CID 136428582.
- ^ S2CID 2555443.
- S2CID 205237059.
- S2CID 53320913.
- S2CID 25096136.
- S2CID 119244679.
- PMID 22395620.
- ^ PMID 26584676.
- S2CID 136428582. Archived from the original(PDF) on 2016-03-07. Retrieved 2015-08-29.
- S2CID 119287960.
- PMID 30006532.
- S2CID 211082922.
- S2CID 249209788.
- S2CID 231709827.
- S2CID 4931477.
- S2CID 119233985.
- S2CID 24851439.
- PMID 18654525. Archived from the original(PDF) on 2016-03-04. Retrieved 2013-03-04.
- S2CID 248422954.
- ISBN 9781119477082.
- PMID 18654525.
- .
- PMID 28128228.
- S2CID 588265.
- S2CID 4143597.
- S2CID 964488.
- S2CID 206545959.
- S2CID 205246446.
- S2CID 139734402.
- PMID 27906340.
- S2CID 27859868.

