Decompressive craniectomy
| Decompressive craniectomy | |
|---|---|
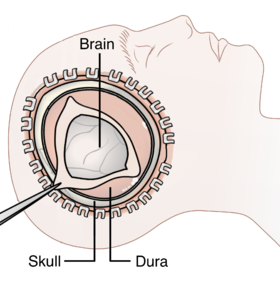 Diagram showing the elements of a decompressive craniectomy | |
| ICD-9-CM | 01.2, 02.02 |
| MeSH | D056424 |
Decompressive craniectomy (
The procedure evolved from a primitive form of surgery known as
Results of clinical trials
Reduction of intracranial pressure
Though the procedure is considered a last resort, some evidence suggests that it does improve outcomes by lowering
DECRA trial
In March 2011, investigators from Australia and several other countries published the results of the DECRA[5] trial in The New England Journal of Medicine. This was a randomized trial comparing decompressive craniectomy to best medical therapy run between 2002 and 2010 to assess the optimal management of patients with medically refractory ICP following diffuse non-penetrating head injury. The study investigators found that decompressive craniectomy was associated with worse functional outcomes, as measured by a standard metric, than best medical care. There were no differences in deaths between groups. However, the results of the DECRA trial have been rejected or at least questioned by many practicing neurosurgeons, and a concurrently published editorial raises several study weaknesses.[6] First, the threshold for defining increased ICP, and the time allowed before declaring ICP medically refractory, are not what many practicing physicians would consider increased or refractory. Second, out of almost 3500 potentially eligible patients, only 155 patients were enrolled, showing that the study cannot be generalized to all patients with severe non-penetrating brain injury.[7] Lastly, more subjects in the craniectomy group had unreactive pupils than patients in the medical therapy group after randomisation and before surgical intervention; thus making this a possible confounding factor.[8]
Other effects
In addition to reducing ICP, studies have found decompressive craniectomy to improve
Decompressive craniectomy is also used to manage major strokes, associated with "malignant" edema and intracranial hypertension. The pooled evidence from three randomised controlled trials in Europe supports the retrospective observations that early (within 48 hours) application of decompressive craniectomy after "malignant" stroke may result in improved survival and functional outcome in patients under the age of 55, compared to conservative management alone.[9]
The procedure is recommended especially for young patients in whom ICP is not controllable by other methods.[1] Age of greater than 50 years is associated with a poorer outcome after the surgery.[3]
Complications
Infections such as meningitis or brain abscess can occur after decompressive craniectomy.[10]
Children
In severely head injured children, a study has shown that decompressive craniectomy resulted in good recovery in all children in the study, suggesting the procedure has an advantage over non-surgical treatment in children.[11] In one of the largest studies on pediatric patients, Jagannathan et al. found a net 65% favorable outcomes rate in pediatric patients for accidental trauma after craniectomy when followed for more than five years. Only three patients were dependent on caregivers.[12] This is the only prospective randomly controlled study to date to support the potential benefit of decompressive craniectomy following traumatic brain injury.[13]
Follow-up treatment
After a craniectomy, the risk of brain injury is increased, particularly after the patient heals and becomes mobile again. Therefore, special measures must be taken to protect the brain, such as a helmet or a temporary implant in the skull.[14]
When the patient has healed sufficiently, the opening in the skull is usually closed with a cranioplasty. If possible, the original skull fragment is preserved after the craniectomy in anticipation of the cranioplasty.[15]
Ongoing trials
The RESCUEicp study is an international multicenter trial that finished recruitment in March 2014. The aim of this study is to determine the effectiveness of decompressive craniectomy, compared to medical management alone, to treat brain swelling and improve outcome. This study is coordinated by the University of Cambridge Academic Neurosurgery Unit[16] and the European Brain Injury Consortium (EBIC).[17][needs update]
The RESCUE-ASDH study Official RESCUE-ASDH Trial Site is a multicenter, pragmatic, parallel group randomised trial that aims to compare the clinical and cost-effectiveness of decompressive craniectomy versus craniotomy for the management of adult head-injured patients undergoing evacuation of an acute subdural haematoma (ASDH). The trial has started recruiting, and is expected to run until 2020. This study is coordinated by the University of Cambridge Academic Neurosurgery Unit. [citation needed]
References
- ^ PMID 9779131.
- S2CID 22490737.
- ^ S2CID 38854921.
- PMID 17020484.
- PMID 21434843.
- PMID 21434843.
- PMID 21434844.
- PMID 24829829.
- S2CID 23509663.
- S2CID 44700600.
- S2CID 10175707.
- PMID 17465359.
- PMID 16437469.
- S2CID 22544660.
- S2CID 6333710.
- ^ University of Cambridge Academic Neurosurgery Unit
- ^ EBIC.nl - European Brain Injury Consortium
