Boronic acid

A boronic acid is an
Boronic acids act as
Boronic acids are used extensively in
The compound
The boronic acid functional group is reputed to have low inherent toxicity. This is one of the reasons for the popularity of the Suzuki coupling in the development and synthesis of pharmaceutical agents. However, a significant fraction of commonly used boronic acids and their derivatives were recently found to gives a positive Ames test and act as chemical mutagens. The mechanism of mutagenicity is thought to involve the generation of organic radicals via oxidation of the boronic acid by atmospheric oxygen.[6]
Structure and synthesis
In 1860, Edward Frankland was the first to report the preparation and isolation of a boronic acid. Ethylboronic acid was synthesized by a two-stage process. First, diethylzinc and triethyl borate reacted to produce triethylborane. This compound then oxidized in air to form ethylboronic acid.[7][8][9] Several synthetic routes are now in common use, and many air-stable boronic acids are commercially available.
Boronic acids typically have high melting points. They are prone to forming
| Boronic acid | R | Structure | Molar mass | CAS number |
Melting point °C |
|---|---|---|---|---|---|
| Phenylboronic acid | Phenyl |
 |
121.93 | 98-80-6 | 216–219 |
| 2-Thienylboronic acid | Thiophen |
 |
127.96 | 6165-68-0 | 138–140 |
| Methylboronic acid | Methyl |
 |
59.86 | 13061-96-6 | 91–94 |
| cis-Propenylboronic acid | propene |
85.90 | 7547-96-8 | 65–70 | |
| trans-Propenylboronic acid | propene |
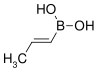 |
85.90 | 7547-97-9 | 123–127 |
Synthesis
Boronic acids can be obtained via several methods. The most common way is reaction of organometallic compounds based on lithium or magnesium (
- PhMgBr + B(OMe)3 → PhB(OMe)2 + MeOMgBr
- PhB(OMe)2 + 2 H2O → PhB(OH)2 + 2 MeOH
Another method is reaction of an
A third method is by palladium catalysed reaction of aryl halides and triflates with diboronyl esters in a coupling reaction known as the Miyaura borylation reaction. An alternative to esters in this method is the use of diboronic acid or tetrahydroxydiboron ([B(OH2)]2).[15][16][17]
Boronic esters (also named boronate esters)
Boronic esters are esters formed between a boronic acid and an alcohol.
| Compound | General formula | General structure |
|---|---|---|
| Boronic acid | RB(OH)2 |  |
| Boronic ester | RB(OR)2 |  |
The compounds can be obtained from
| Boronic ester | Diol | Structural formula | Molar mass | CAS number
|
Boiling point (°C) |
|---|---|---|---|---|---|
| Allylboronic acid pinacol ester | pinacol |  |
168.04 | 72824-04-5 | 50–53 (5 mmHg) |
| Phenyl boronic acid trimethylene glycol ester | trimethylene glycol |
 |
161.99 | 4406-77-3 | 106 (2 mm Hg) |
| Diisopropoxymethylborane | isopropanol |
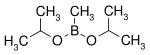 |
144.02 | 86595-27-9 | 105 -107 |
Compounds with 5-membered cyclic structures containing the C–O–B–O–C linkage are called dioxaborolanes and those with 6-membered rings dioxaborinanes.
Organic chemistry applications
Suzuki coupling reaction
Boronic acids are used in organic chemistry in the Suzuki reaction. In this reaction the boron atom exchanges its aryl group with an alkoxy group from palladium.
-
(1)
Chan–Lam coupling
In the Chan–Lam coupling the alkyl, alkenyl or aryl boronic acid reacts with a N–H or O–H containing compound with Cu(II) such as
The
Liebeskind–Srogl coupling
In the
Conjugate addition
The boronic acid organic residue is a nucleophile in
- The catalyst system in this reaction is tris(dibenzylideneacetone)dipalladium(0) / tricyclohexylphosphine.
Another conjugate addition is that of gramine with phenylboronic acid catalyzed by cyclooctadiene rhodium chloride dimer:[23]
Oxidation
Boronic esters are
Homologation
- In boronic ester
-
Boronic ester homologization
-
Homologization application
In this reaction
Electrophilic allyl shifts
Allyl boronic esters engage in
Hydrolysis
Hydrolysis of boronic esters back to the boronic acid and the alcohol can be accomplished in certain systems with thionyl chloride and pyridine.[26] Aryl boronic acids or esters may be hydrolyzed to the corresponding phenols by reaction with hydroxylamine at room temperature.[27]
C–H coupling reactions
The diboron compound
In one modification the arene reacts using only a
Unlike in ordinary
Protonolysis
Protodeboronation is a chemical reaction involving the protonolysis of a boronic acid (or other organoborane compound) in which a carbon-boron bond is broken and replaced with a carbon-hydrogen bond. Protodeboronation is a well-known undesired side reaction, and frequently associated with metal-catalysed coupling reactions that utilise boronic acids (see Suzuki reaction). For a given boronic acid, the propensity to undergo protodeboronation is highly variable and dependent on various factors, such as the reaction conditions employed and the organic substituent of the boronic acid:

Supramolecular chemistry
Saccharide recognition
The covalent pair-wise interaction between boronic acids and hydroxy groups as found in alcohols and acids is rapid and reversible in aqueous solutions. The equilibrium established between boronic acids and the hydroxyl groups present on saccharides has been successfully employed to develop a range of sensors for saccharides.[34] One of the key advantages with this dynamic covalent strategy[35] lies in the ability of boronic acids to overcome the challenge of binding neutral species in aqueous media. If arranged correctly, the introduction of a tertiary amine within these supramolecular systems will permit binding to occur at physiological pH and allow signalling mechanisms such as photoinduced electron transfer mediated fluorescence emission to report the binding event.
Potential applications for this research include blood glucose monitoring systems to help manage diabetes mellitus. As the sensors employ an optical response, monitoring could be achieved using minimally invasive methods, one such example is the investigation of a contact lens that contains a boronic acid based sensor molecule to detect glucose levels within ocular fluids.[36]
Notes
- 2,6-lutidine. The final product is a vinyl cyclopropane. Note: ee stands for enantiomeric excess
- ^ In situ second step reaction of boronate ester with copper(II) bromide
References
- PMID 7372625.
- PMID 4364061.
- PMID 12779325.
- S2CID 205557212.
- ISSN 1083-6160.
- .
- .
- .
- ISBN 978-3-527-30991-7.
- ^ Example: Kristensen, Jesper Langgaard; Lysén, Morten; Vedsø, Per; Begtrup, Mikael (2005). "Synthesis of Ortho Substituted Arylboronic Esters by in situ Traping of Unstable Lithio Intermediates: 2-(5,5-Dimethyl-1,3,2-dioxaborinan-2-yl)benzoic acid ethyl ester". Organic Syntheses. 81: 134; Collected Volumes, vol. 11, pp. 1015 prep= v81p0134.
- ^ Example: Li, Wenjie; Nelson, Dorian P.; Jensen, Mark S.; Scott Hoerrner, R.; Cai, Dongwei; Larsen, Robert D. (2005). "Synthesis of 3-Pyridylboronic Acid and its Pinacol Ester. Application of 3-Pyridylboronic acid in Suzuki Coupling to Prepare 3-Pyridin-3-ylquinoline". Organic Syntheses. 81: 89; Collected Volumes, vol. 11, p. 393.
- ^ Charette, André B.; Lebel, Hélène (1999). "(2S,3S)-(+)-(3-Phenylcyclopropyl)methanol". Organic Syntheses. 76: 86; Collected Volumes, vol. 10, p. 613.
- ^ Washburn, Robert M.; Levens, Ernest; Albright, Charles F.; Billig, Franklin A. (1959). "Benzeneboronic anhydride". Organic Syntheses. 39: 3; Collected Volumes, vol. 4, p. 68.
- PMID 21721088.
- PMID 21105666.
- S2CID 98029876.
- ^ Kidwell, R. L.; Murphy, M.; Darling, S. D. (1969). "Phenols: 6-Methoxy-2-Naphthol". Organic Syntheses. 49: 90; Collected Volumes, vol. 5, p. 918.
- ^ Washburn, Robert M.; Levens, Ernest; Albright, Charles F.; Billig, Franklin A. (1959). "Benzeneboronic anhydride". Organic Syntheses. 39: 3; Collected Volumes, vol. 4, p. 68.
- .
- .
- PMID 17266312.
- PMID 17305348.
- .
- PMID 17315879.
- S2CID 15944330.
- .
- ^ Ishiyama, Tatsuo; Murata, Miki; Ahiko, Taka-aki; Miyaura, Norio (2000). "Bis(pinacolato)diboron". Organic Syntheses. 77: 176; Collected Volumes, vol. 10, p. 115.
- hdl:2115/56222.
- PMID 11792205.
- S2CID 34802662.
- PMID 18027947.
- PMID 15584754.
- ISBN 978-0-85404-537-2.
- PMID 12491278.
- ^ US 6850786, Wayne Front March, "Ocular analyte sensor", issued 2005-02-01

![The Suzuki reaction {\displaystyle {\begin{matrix}{}\\{\ce {{R1-BY2}+R2-X->[{\underset {\text{catalyst}}{\text{Pd}}}][{\text{Base}}]R1-R2}}\\{}\end{matrix}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/c35df36f38fec8abbc8e9d1d9f04e9b2687ae245)








