Dioxygen in biological reactions
Photosynthesis
In nature, free oxygen is produced by the light-driven
A simplified overall formula for photosynthesis is:[3]
- 6CO
2 + 6H
2O + photons → C
6H
12O
6 + 6O
2
- 6CO
(or simply carbon dioxide + water + sunlight → glucose + oxygen)
Photolytic oxygen evolution during photosynthesis occurs via the light-dependent oxidation of water to molecular oxygen and can be written as the following simplified chemical reaction: 2H2O → 4e− + 4H+ + O2
The reaction occurs in the
Water oxidation is catalyzed by a
Oxygen uptake and transport
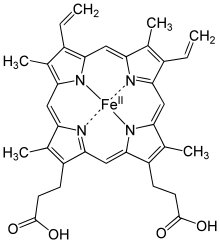
In
Following inhalation into the lungs, oxygen
After being carried in blood to a body tissue in need of oxygen, O2 is handed off from the heme group to
Aerobic respiration
Molecular oxygen, O2, is essential for
- C
6H
12O
6 + 6O
2 → 6CO
2 + 6H
2O + 2880 kJ/mol
- C
Reactive oxygen species
Parts of the
See also
- Oxygen cycle
- Oxygen-haemoglobin dissociation curve
- Oxygen
- Apparent oxygen utilisation
- CO-oximeter
- Oxygen catastrophe
- Oxygen toxicity
- Reactive oxygen species
References
- ISBN 1-4289-2397-7.
- ^ Broeker, W.S. (2006). "Breathing easy, Et tu, O2". Columbia University. Retrieved 2007-10-21.
- ISBN 0-13-048450-4, p. 958
- ^ ISBN 0-7167-1007-2.
- ^
Raval M, Biswal B, Biswal U (2005). "The mystery of oxygen evolution: analysis of structure and function of photosystem II, the water-plastoquinone oxido-reductase". Photosynthesis Research. 85 (3): 267–93. S2CID 12893308.
- ISBN 0-12-352651-5.
- ^ CO2 is released from another part of the hemoglobin molecule, as its acid, which causes CO2 to be released from bicarbonate, its major reservoir in blood plasma (see Bohr effect)
- ^ Stwertka 1998, p. 48.
- ^ a b c d e Emsley 2001, p. 298.
- ^ Cook & Lauer 1968, p. 500.
- ^ Figures given are for values up to 50 miles above the surface
- ^ Emsley 2001, p. 303.
- PMID 35213291.
- ^
Hoffmann, Roald (2004). "The Story of O". American Scientist. 92 (1): 23. doi:10.1511/2004.1.23. Archived from the originalon 2007-02-22. Retrieved 2007-03-03.
- Emsley, John (2001). "Oxygen". Nature's Building Blocks: An A-Z Guide to the Elements. Oxford, England, UK: Oxford University Press. pp. 297–304. ISBN 0-19-850340-7.
- Cook, Gerhard A.; Lauer, Carol M. (1968). "Oxygen". In Clifford A. Hampel (ed.). The Encyclopedia of the Chemical Elements. New York: Reinhold Book Corporation. pp. 499–512. LCCN 68-29938.
- Stwertka, Albert (1998). Guide to the Elements (Revised ed.). Oxford University Press. ISBN 0-19-508083-1.
