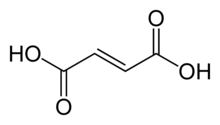
Fumaric acid
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-But-2-enedioic acid | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 605763 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.003.404 |
| EC Number |
|
| E number | E297 (preservatives) |
| 49855 | |
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 9126 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H4O4 | |
| Molar mass | 116.072 g·mol−1 |
| Appearance | White solid |
| Density | 1.635 g/cm3 |
| Melting point | 287 °C (549 °F; 560 K) (decomposes)[2] |
| 4.9 g/L at 20 °C[1] | |
| Acidity (pKa) | pka1 = 3.03, pka2 = 4.44 (15 °C, cis isomer) |
| −49.11·10−6 cm3/mol | |
| non zero | |
| Pharmacology | |
| D05AX01 (WHO) | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H319 | |
| P264, P280, P305+P351+P338, P313 | |
| NFPA 704 (fire diamond) | |
| 375 °C (707 °F; 648 K) | |
| Related compounds | |
Related carboxylic acids
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Fumaric acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297.[3] The salts and esters are known as fumarates. Fumarate can also refer to the C
4H
2O2−
4 ion (in solution). Fumaric acid is the trans isomer of butenedioic acid, while maleic acid is the cis isomer.
Biosynthesis and occurrence
It is produced in
Fumaric acid is found in
Fumarate is an
Human skin naturally produces fumaric acid when exposed to sunlight.[4][5]
Fumarate is also a product of the urea cycle.
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "TCACycle_WP78".
Uses
Food
Fumaric acid has been used as a food acidulant since 1946. It is approved for use as a food additive in the EU,
The European Commission Scientific Committee on Animal Nutrition, part of DG Health, found in 2014 that fumaric acid is "practically non-toxic" but high doses are probably nephrotoxic after long-term use.[11]
Medicine
Fumaric acid was developed as a medicine to treat the autoimmune condition psoriasis in the 1950s in Germany as a tablet containing 3 esters, primarily dimethyl fumarate, and marketed as Fumaderm by Biogen Idec in Europe. Biogen would later go on to develop the main ester, dimethyl fumarate, as a treatment for multiple sclerosis.
In patients with relapsing-remitting multiple sclerosis, the ester dimethyl fumarate (BG-12, Biogen) significantly reduced relapse and disability progression in a phase 3 trial. It activates the
Signaling metabolite in cell biology
In mammals, malic enzyme 2 (ME2) is allosterically activated by fumarate. The
Other uses
Fumaric acid is used in the manufacture of
When fumaric acid is added to their feed, lambs produce up to 70% less methane during digestion.[14]
Synthesis
Fumaric acid is produced based on
Historic and laboratory routes
Fumaric acid was first prepared from
Reactions
The chemical properties of fumaric acid can be anticipated from its component
Safety
Fumaric acid is required for life. The oral
See also
- Citric acid cycle (TCA cycle)
- Photosynthesis
- Maleic acid, the cis isomer of fumaric acid
References
- ^ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ Pubchem. "Fumaric acid". pubchem.ncbi.nlm.nih.gov.
- ^ ISBN 3-527-30673-0.
- ^ Active Ingredients Used in Cosmetics: Safety Survey, Council of Europe. Committee of Experts on Cosmetic Products
- ^ "Fumaric Acid Foods". Retrieved 2018-04-22.[permanent dead link]
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Retrieved 2011-10-27.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". Food and Drug Administration. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 - Labelling of ingredients". 8 September 2011. Retrieved 2011-10-27.
- ^ "Fumaric Acid - The Chemical Company". The Chemical Company. Retrieved 2018-04-22.
- ^ Eats, Serious. "The Science Behind Salt and Vinegar Chips". www.seriouseats.com.
- ^ European Commission: "European Commission Report of the Scientific Committee on Animal Nutrition on the Safety of Fumaric Acid" (PDF). Retrieved 2014-03-07.
- S2CID 6614191.
- PMID 33770508.
- ^ "Scientists look to cut cow flatulence". phys.org. March 21, 2008.
- .
- .


