Genistin
Appearance
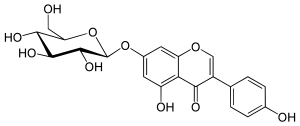
| |
| Names | |
|---|---|
| IUPAC name
7-(β-D-Glucopyranosyloxy)-4′,5-dihydroxyisoflavone
| |
| Systematic IUPAC name
5-Hydroxy-3-(4-hydroxyphenyl)-7-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-4H-1-benzopyran-4-one | |
| Other names
Genistoside
Genistine Genistein 7-glucoside Genistein glucoside Genistein-7-glucoside Genisteol 7-monoglucoside Glucosyl-7-genistein Genistein 7-O-beta-D-glucoside | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.120.406 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H20O10 | |
| Molar mass | 432.37 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Genistin is an
anticancer
effects are analogous.
Metabolism
When ingested along the diet, genistin is readily converted to its
mice can cause the complete conversion.[2]
Biological importance
Estrogenic activity
Genistin, like genistein, is a
tumors to regress.[3]
Antiviral activity
Genistin and other isoflavones are demonstrated to be bioactive within the neonatal intestine and may reduce the severity of rotavirus infections; genistin alone shows inhibition of the viral infectivity by 40-60%.[4]
Bone metabolism
In vitro study have shown that both genistin and
fructooligosaccharides.[6] The amount of new bone produced by grafting genistin in collagen matrix was compared to the bone produced by collagen matrix alone in New Zealand white rabbits, and was observed that genistin caused significant increase in bone formation.[7]
References
External links
- "Genistin (Genistein-7-O-β-D-glucoside)". Natural Compounds. 2013. p. 362. ISBN 978-1-4614-0534-4.
- Chemical compound Review at Wikigenes
