Rotavirus
| Rotavirus | |
|---|---|
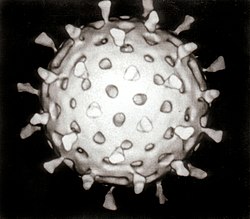
| |
| Computer–aided reconstruction of a rotavirus based on several electron micrographs | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Duplornaviricota |
| Class: | Resentoviricetes |
| Order: | Reovirales
|
| Family: | Sedoreoviridae |
| Genus: | Rotavirus |
| Species | |
| |
Rotaviruses are the most common cause of
The virus is transmitted by the faecal–oral route. It infects and damages the cells that line the small intestine and causes gastroenteritis (which is often called "stomach flu" despite having no relation to influenza). Although rotavirus was discovered in 1973 by Ruth Bishop and her colleagues by electron micrograph images[4] and accounts for approximately one third of hospitalisations for severe diarrhoea in infants and children,[5] its importance has historically been underestimated within the public health community, particularly in developing countries.[6] In addition to its impact on human health, rotavirus also infects other animals, and is a pathogen of livestock.[7]
Rotaviral enteritis is usually an easily managed disease of childhood, but among children under 5 years of age rotavirus caused an estimated 151,714 deaths from diarrhoea in 2019.[8] In the United States, before initiation of the rotavirus vaccination programme in the 2000s, rotavirus caused about 2.7 million cases of severe gastroenteritis in children, almost 60,000 hospitalisations, and around 37 deaths each year.[9] Following rotavirus vaccine introduction in the United States, hospitalisation rates have fallen significantly.[10][11] Public health campaigns to combat rotavirus focus on providing oral rehydration therapy for infected children and vaccination to prevent the disease.[12] The incidence and severity of rotavirus infections has declined significantly in countries that have added rotavirus vaccine to their routine childhood immunisation policies.[13][14][15]
Rotavirus is a
Virology
Types of rotavirus
There are 11 species of rotavirus (sometimes informally called groups) referred to as RVA, RVB, RVC, RVD, RVF, RVG, RVH, RVI, RVJ, RVK and RVL.[17][18] Humans are primarily infected by rotaviruses in the species RVA. This one and the other species cause disease in other animals,[19] for example, species RVH in pigs, RVD, RVF and RVG in birds, RVI in cats and RVJ in bats.[20][21][22][23]
Within group A rotaviruses there are different strains, called
Structure
The genome of rotaviruses consists of 11 unique double helix molecules of RNA (dsRNA) which are 18,555 nucleotides in total. Each helix, or segment, is a gene, numbered 1 to 11 by decreasing size. Each gene codes for one protein, except genes 9, which codes for two.[30] The RNA is surrounded by a three-layered icosahedral protein capsid. Viral particles are up to 76.5 nm in diameter[31][32] and are not enveloped.[33]
Proteins

There are six viral proteins (VPs) that form the virus particle (
At least six of the twelve proteins encoded by the rotavirus genome bind RNA.[35] The role of these proteins in rotavirus replication is not entirely understood; their functions are thought to be related to RNA synthesis and packaging in the virion, mRNA transport to the site of genome replication, and mRNA translation and regulation of gene expression.[36]
Structural proteins

VP1 is located in the core of the virus particle and is an RNA-dependent RNA polymerase enzyme.[37] In an infected cell this enzyme produces mRNA transcripts for the synthesis of viral proteins and produces copies of the rotavirus genome RNA segments for newly produced virus particles.[38]
VP2 forms the core layer of the virion and binds the RNA genome.[39]
VP3 is part of the inner core of the virion and is an enzyme called
VP4 is on the surface of the virion that protrudes as a spike.
VP6 forms the bulk of the capsid. It is highly antigenic and can be used to identify rotavirus species.[48] This protein is used in laboratory tests for rotavirus infections.[49]
VP7 is a
Nonstructural viral proteins
NSP1, the product of gene 5, is a
Targets for degradation include several IRF transcription factors required for interferon gene transcription.[51]
NSP2 is an RNA-binding protein that accumulates in cytoplasmic inclusions (viroplasms) and is required for genome replication.[52][39]
NSP3 is bound to viral mRNAs in infected cells and it is responsible for the shutdown of cellular protein synthesis.[53] NSP3 inactivates two translation initiation factors essential for synthesis of proteins from host mRNA.
First, NSP3 ejects
NSP4 is a viral enterotoxin that induces diarrhoea and was the first viral enterotoxin discovered.[56] It is a viroporin that elevates cytosolic Ca2+ in mammalian cells.[57]
NSP5 is encoded by genome segment 11 of rotavirus A. In virus-infected cells NSP5 accumulates in the viroplasm.[58]
NSP6 is a nucleic acid binding protein[59] and is encoded by gene 11 from an out-of-phase open reading frame.[60]
| RNA Segment (Gene) | Size (base pairs) | Protein | UniProt | Molecular weight kDa |
Location | Copies per particle | Function |
|---|---|---|---|---|---|---|---|
| 1 | 3302 | VP1 | P22678 | 125 | At the vertices of the core | 12 | RNA-dependent RNA polymerase |
| 2 | 2690 | VP2 | A2T3R5 | 102 | Forms inner shell of the core | 120 | RNA binding |
| 3 | 2591 | VP3 | A2T3S5 | 88 | At the vertices of the core | 12 | methyltransferase mRNA capping enzyme |
| 4 | 2362 | VP4 | A2T3T2 | 87 | Surface spike | 180 (60 trimers)[61] | Cell attachment, virulence |
| 5 | 1611 | NSP1 | Q99FX5 | 59 | Nonstructural | 0 | 5'RNA binding, interferon antagonist |
| 6 | 1356 | VP6 | Q6LE89 | 45 | Inner Capsid | 780 (260 trimers)[61] | Structural and species-specific antigen |
| 7 | 1104 | NSP3 | P03536 | 37 | Nonstructural | 0 | Enhances viral mRNA activity and shut-offs cellular protein synthesis |
| 8 | 1059 | NSP2 | A2T3P0 | 35 | Nonstructural | 0 | NTPase involved in RNA packaging |
| 9 | 1062 | VP71 VP72 | P03533 | 38 and 34 | Surface | 780 (260 trimers)[61] | Structural and neutralisation antigen |
| 10 | 751 | NSP4 | P04512 | 20 | Nonstructural | 0 | Viroporin (enterotoxin) |
| 11 | 667 | NSP5 NSP6 | A2T3Q9 P11203 | 22 | Nonstructural | 0 | ssRNA and dsRNA binding modulator of NSP2, phosphoprotein |
This table is based on the simian rotavirus strain SA11. RNA-protein coding assignments differ in some strains.
Replication

- Attachment of the virus to the host cells, which is mediated by VP4 and VP7
- Penetration of the cell by the virus and uncoating of the viral capsid
- Plus strand ssRNA synthesis (this acts as the mRNA) synthesis, which is mediated by VP1, VP3 and VP2
- Formation of the viroplasm, viral RNA packaging and minus strand RNA synthesis and formation of the double-layered virus particles
- Virus particle maturation and release of progeny virions
The attachment of the virus to the host cell is initiated by VP4, which attaches to molecules, called
The eleven dsRNA strands remain within the protection of the two protein shells and the viral
During the infection, rotaviruses produce mRNA for both
Transmission
Rotaviruses are transmitted by the faecal–oral route, via contact with contaminated hands, surfaces and objects,[68] and possibly by the respiratory route.[69] Viral diarrhoea is highly contagious. The faeces of an infected person can contain more than 10 trillion infectious particles per gram;[48] fewer than 100 of these are required to transmit infection to another person.[3]
Rotaviruses are stable in the environment and have been found in
Signs and symptoms
Rotaviral enteritis is a mild to severe disease characterised by nausea, vomiting, watery diarrhoea and low-grade fever. Once a child is infected by the virus, there is an incubation period of about two days before symptoms appear.[71] The period of illness is acute. Symptoms often start with vomiting followed by four to eight days of profuse diarrhoea. Dehydration is more common in rotavirus infection than in most of those caused by bacterial pathogens, and is the most common cause of death related to rotavirus infection.[72]
Rotavirus infections can occur throughout life: the first usually
Disease mechanisms

Rotaviruses replicate mainly in the
The diarrhoea is caused by multiple activities of the virus.[81] Malabsorption occurs because of the destruction of gut cells called enterocytes. The toxic rotavirus protein NSP4 induces age- and calcium ion-dependent chloride secretion, disrupts SGLT1 (sodium/glucose cotransporter 2) transporter-mediated reabsorption of water, apparently reduces activity of brush-border membrane disaccharidases, and activates the calcium ion-dependent secretory reflexes of the enteric nervous system.[56] The elevated concentrations of calcium ions in the cytosol (which are required for the assembly of the progeny viruses) is achieved by NSP4 acting as a viroporin. This increase in calcium ions leads to autophagy (self destruction) of the infected enterocytes.[82]
NSP4 is also secreted. This extracellular form, which is modified by protease enzymes in the gut, is an enterotoxin which acts on uninfected cells via integrin receptors, which in turn cause and increase in intracellular calcium ion concentrations, secretory diarrhoea and autophagy.[83]
The vomiting, which is a characteristic of rotaviral enteritis, is caused by the virus infecting the enterochromaffin cells on the lining of the digestive tract. The infection stimulates the production of 5' hydroxytryptamine (serotonin). This activates vagal afferent nerves, which in turn activates the cells of the brain stem that control the vomiting reflex.[84]
Healthy enterocytes secrete lactase into the small intestine; milk intolerance due to lactase deficiency is a symptom of rotavirus infection,[85] which can persist for weeks.[86] A recurrence of mild diarrhoea often follows the reintroduction of milk into the child's diet, due to bacterial fermentation of the disaccharide lactose in the gut.[87]
Immune responses
Specific responses
Rotaviruses elicit both B and T cell immune responses. Antibodies to the rotavirus VP4 and VP7 proteins neutralise viral infectivity in vitro and in vivo.[88] Specific antibodies of the classes IgM, IgA and IgG are produced, which have been shown to protect against rotavirus infection by the passive transfer of the antibodies in other animals.[89] Maternal trans-placental IgG might play a role in the protection neonates from rotavirus infections, but on the other hand might reduce vaccine efficacy.[90]
Innate responses
Following infection by rotaviruses there is a rapid innate immune response involving types I and III interferons and other cytokines (particularly Th1 and Th2)[91] which inhibit the replication of the virus and recruit macrophages and natural killer cells to the rotavirus infected cells.[92] The rotavirus dsRNA activates pattern recognition receptors such toll-like receptors that stimulate the production of interferons.[93] The rotavirus protein NSP1 counteracts the effects of type 1 interferons by suppressing the activity of the interferon regulatory proteins IRF3, IRF5 and IRF7.[93]
Markers of protection
The levels of IgG and IgA in the blood and IgA in the gut correlate with protection from infection.
Diagnosis and detection
Diagnosis of infection with a rotavirus normally follows diagnosis of gastroenteritis as the cause of severe diarrhoea. Most children admitted to hospital with gastroenteritis are tested for rotavirus.[96][97]
Specific
Treatment and prognosis
Treatment of acute rotavirus infection is nonspecific and involves management of symptoms and, most importantly,
Prevention
Rotaviruses are highly contagious and cannot be treated with antibiotics or other drugs. Because improved
In 2006, two new vaccines against rotavirus A infection were shown to be safe and effective in children,[111] and in 2009, the WHO recommended that rotavirus vaccine be included in all national immunisation programmes.[112]
The incidence and severity of rotavirus infections has declined significantly in countries that have acted on this recommendation.[13][14][15] A 2014 review of available clinical trial data from countries routinely using rotavirus vaccines in their national immunisation programs found that rotavirus vaccines have reduced rotavirus hospitalisations by 49–92% and all cause diarrhoea hospitalisations by 17–55%.[113] In Mexico, which in 2006 was among the first countries in the world to introduce rotavirus vaccine, diarrhoeal disease death rates dropped during the 2009 rotavirus season by more than 65 percent among children age two and under.[114] In Nicaragua, which in 2006 became the first developing country to introduce a rotavirus vaccine, severe rotavirus infections were reduced by 40 percent and emergency room visits by a half.[115] In the United States, rotavirus vaccination since 2006 has led to drops in rotavirus-related hospitalisations by as much as 86 percent.[116] The vaccines may also have prevented illness in non-vaccinated children by limiting the number of circulating infections.[116][117] In developing countries in Africa and Asia, where the majority of rotavirus deaths occur, a large number of safety and efficacy trials as well as recent post-introduction impact and effectiveness studies of Rotarix and RotaTeq have found that vaccines dramatically reduced severe disease among infants.[15][118][119][120] In September 2013, the vaccine was offered to all children in the UK, aged between two and three months.[121] A 2022 study found that the number of rotavirus cases in infants in England under one year of age was reduced by 77–88%. In all age groups, the number of laboratory-confirmed rotavirus infections was reduced by 69–83%.[122] In Europe, hospitalisation rates following infection by rotaviruses have decreased by 65% to 84% following the introduction of the vaccine.[123] Globally, vaccination has reduced hospital admissions and emergency department visits by a median of 67%.[124]
Rotavirus vaccines are licensed in over 100 countries, and more than 80 countries have introduced routine rotavirus vaccination, almost half with the support of the GAVI vaccine alliance.[125] To make rotavirus vaccines available, accessible, and affordable in all countries—particularly low- and middle-income countries in Africa and Asia where the majority of rotavirus deaths occur, PATH (formerly Program for Appropriate Technology in Health), the WHO, the U.S. Centers for Disease Control and Prevention, and GAVI have partnered with research institutions and governments to generate and disseminate evidence, lower prices, and accelerate introduction.[126]
The vaccine may prevent
Epidemiology
Rotavirus A, which accounts for more than 90% of rotavirus gastroenteritis in humans,[16] is endemic worldwide. Each year rotaviruses cause millions of cases of diarrhoea in developing countries, almost 2 million of which result in hospitalisation.[6] In 2019, an estimated 151,714 children younger than five died from rotavirus infections, 90 percent of whom were in developing countries.[8] Almost every child has been infected with rotaviruses by age five.[2][129] Rotaviruses are the leading single cause of severe diarrhoea among infants and children, are responsible for about a third of the cases requiring hospitalisation,[10] and cause 37% of deaths attributable to diarrhoea and 5% of all deaths in children younger than five.[130] Boys are twice as likely as girls to be admitted to hospital for rotavirus infections.[131][132] In the pre-vaccination era, rotavirus infections occurred primarily during cool, dry seasons.[133][134] The number attributable to food contamination is unknown.[135]
Outbreaks of rotavirus A diarrhoea are common among hospitalised infants, young children attending day care centres, and elderly people in nursing homes.[76][136] An outbreak caused by contaminated municipal water occurred in Colorado in 1981.[137] During 2005, the largest recorded epidemic of diarrhoea occurred in Nicaragua. This unusually large and severe outbreak was associated with mutations in the rotavirus A genome, possibly helping the virus escape the prevalent immunity in the population.[138] A similar large outbreak occurred in Brazil in 1977.[139]
Rotavirus B, also called adult diarrhoea rotavirus or ADRV, has caused major epidemics of severe diarrhoea affecting thousands of people of all ages in China. These epidemics occurred as a result of sewage contamination of drinking water.[140][141] Rotavirus B infections also occurred in India in 1998; the causative strain was named CAL. Unlike ADRV, the CAL strain is endemic.[142][143] To date, epidemics caused by rotavirus B have been confined to mainland China, and surveys indicate a lack of immunity to this species in the United States.[144] Rotavirus C has been associated with rare and sporadic cases of diarrhoea in children, and small outbreaks have occurred in families.[145]
-
The seasonal variation of rotavirus A infections in England: rates of infection peak during the winter months.[146]
-
Preventable child deaths from rotavirus vaccination, 2016. Annual number of preventable deaths in children under five years old from rotavirus if full coverage of the rotavirus vaccine was achieved.[147]
Other animals
Rotaviruses infect the young of many species of animals and they are a major cause of diarrhoea in wild and reared animals worldwide.[7] As a pathogen of livestock, notably in young calves and piglets, rotaviruses cause economic loss to farmers because of costs of treatment associated with high morbidity and mortality rates.[148] These rotaviruses are a potential reservoir for genetic exchange with human rotaviruses.[148] There is evidence that animal rotaviruses can infect humans, either by direct transmission of the virus or by contributing one or several RNA segments to reassortants with human strains.[149][150][151]
History

In 1943, Jacob Light and Horace Hodes proved that a filterable agent in the faeces of children with infectious diarrhoea also caused scours (livestock diarrhoea) in cattle.[152] Three decades later, preserved samples of the agent were shown to be rotavirus.[153] In the intervening years, a virus in mice[154] was shown to be related to the virus causing scours.[155] In 1973, Ruth Bishop and colleagues described related viruses found in children with gastroenteritis.[4]
In 1974,
References
- PMID 26337738.
- ^ S2CID 30544613.
- ^ S2CID 31164630.
- ^ PMID 19799704.
- PMID 34904636.
- ^ S2CID 424503.
- ^ ISBN 978-0-12-375158-4.
- ^ S2CID 249072461.
- PMID 17357047.
- ^ PMID 24913793.
- S2CID 20940659.
- ^ PMID 18026034.
- ^ S2CID 23996836.
- ^ PMID 20622508.
- ^ PMID 27059361.
- ^ S2CID 39847059.
- ^ "Virus Taxonomy: 2024 Release". International Committee on Taxonomy of Viruses. Retrieved 22 April 2025.
- PMID 31447474.
- ^ PMID 20684716.
- PMID 21801631.
- PMID 24960190.
- PMID 28255929.
- PMID 27932285.
- S2CID 22421988.
- ^ PMID 22284787.
- PMID 28748110.
- PMID 2556435.
- PMID 33482744.
- PMID 38015834.
- PMID 2556635.
- ^ S2CID 11290382.
- PMID 8050286.
- ^ S2CID 197541267.
- OCLC 55684328.
- PMID 7595370.
- PMID 11444036.
- PMID 17657346.
- PMID 22595300.
- ^ PMID 15010217.
- ISBN 978-0-12-375147-8.
- PMID 20025612.
- PMID 16571811.
- PMID 12234525.
- ^ PMID 15010218.
- PMID 12167342.
- PMID 24523471.
- PMID 32192193.
- ^ PMID 9015109.
- PMID 6321549.
- PMID 8395125.
- PMID 27009959.
- PMID 8030243.
- PMID 8388495.
- PMID 26727111.
- PMID 22658208.
- ^ PMID 19114772.
- PMID 28256607.
- PMID 8811003.
- PMID 17658646.
- S2CID 21538632.
- ^ PMID 36996819.
- OCLC 55684328.
- PMID 20397068.
- PMID 27009959.
- PMID 15220450.
- PMID 15579070.
- PMID 20024520.
- S2CID 20327842.
- ^ S2CID 28625697.
- PMID 6091548.
- PMID 10532018.
- PMID 1962726.
- S2CID 34569166.
- ISBN 978-0-471-49663-2.
- PMID 21318862.
- ^ PMID 14871633.
- PMID 32918943.
- PMID 19457420.
- PMID 8050281.
- PMID 16641274.
- PMID 15367586.
- PMID 21151776.
- PMID 16731945.
- PMID 22722079.
- PMID 18492865.
- PMID 12811680.
- PMID 6436397.
- PMID 19252425.
- PMID 22880110.
- PMID 27847365.
- S2CID 3568330.
- PMID 23486667.
- ^ PMID 27994593.
- PMID 7817873.
- PMID 23596320.
- ^ S2CID 10992309.
- PMID 16650331.
- ISBN 978-0-12-375147-8.
- ISBN 978-0-471-49663-2.
- PMID 15027000.
- S2CID 26076784.
- PMID 8855579.
- ^ World Health Organization, UNICEF. "Joint Statement: Clinical Management of Acute Diarrhoea" (PDF). Retrieved 3 May 2012.
- S2CID 27763488.
- PMID 26644891.
- S2CID 4845135.
- PMID 10219046.
- PMID 11444025.
- PMID 15687880. Archived from the originalon 11 May 2013. Retrieved 21 January 2008.
- PMID 16099078.
- PMID 18202442.
- S2CID 28963507.
- PMID 25048849.
- S2CID 27287753.
- S2CID 7723807.
- ^ PMID 35632617.
- S2CID 1893099.
- PMID 20684687.
- ^ Nelson EA, Widdowson MA, Kilgore PE, Steele D, Parashar UD, eds. (2009). "Rotavirus in Asia: Updates on Disease Burden, Genotypes and Vaccine Introduction". Vaccine. 27 (Suppl 5): F1 – F138.
- ^ World Health Organization (2009). "Rotavirus vaccines: an update" (PDF). Weekly Epidemiological Record. 51–52 (84): 533–540. Retrieved 8 May 2012.
- ^ "New vaccine to help protect babies against rotavirus". UK Department of Health. 10 November 2012. Retrieved 10 November 2012.
- PMID 34043765.
- PMID 25795258.
- PMID 28430997.
- ^ "Rotavirus Deaths & Rotavirus Vaccine Introduction Maps – ROTA Council". rotacouncil.org. Archived from the original on 12 July 2016. Retrieved 29 July 2016.
- S2CID 7567316.
- ^ "Rotavirus vaccination tied to lower rates of type 1 diabetes". Reuters. 22 January 2019. Retrieved 10 February 2019.
- ISSN 0362-4331. Retrieved 10 February 2019.
- PMID 16494759.
- PMID 22030330.
- S2CID 3272810.
- PMID 8752285.
- PMID 19939844.
- PMID 19056806.
- S2CID 10969637.
- PMID 25944726.
- PMID 6320684.
- PMID 17229854.
- PMID 6263087.
- S2CID 54346351.
- PMID 2555422.
- S2CID 34463384.
- S2CID 21258083.
- PMID 2479654.
- PMID 20491826.
- ^ "Rotavirus vaccination programme for infants". www.gov.uk. Public Health England. 26 July 2013.
- ^ Dadonaite B, Ritchie H (2019). "Rotavirus vaccine – an effective tool that prevents children dying from diarrhea". Our World in Data.
- ^ PMID 19781872.
- PMID 17416132.
- PMID 15066329.
- S2CID 42693014.
- PMID 18015921.
- PMID 184047.
- PMID 4998842.
- ^ PMID 965097.
- ^ PMID 77663.
- PMID 4137164.
- PMID 43850.
- S2CID 11547573.
- S2CID 25276891.
- PMID 19072246.
External links
 Medicine portal
Medicine portal Viruses portal
Viruses portal Media related to Rotavirus at Wikimedia Commons
Media related to Rotavirus at Wikimedia Commons

![The seasonal variation of rotavirus A infections in England: rates of infection peak during the winter months.[146]](http://upload.wikimedia.org/wikipedia/commons/thumb/4/4e/Rotavirus_seasonal_distribution.png/650px-Rotavirus_seasonal_distribution.png)
![Preventable child deaths from rotavirus vaccination, 2016. Annual number of preventable deaths in children under five years old from rotavirus if full coverage of the rotavirus vaccine was achieved.[147]](http://upload.wikimedia.org/wikipedia/commons/thumb/8/81/Avertable-deaths-from-rotavirus-with-full-vaccine-coverage.png/960px-Avertable-deaths-from-rotavirus-with-full-vaccine-coverage.png)