Jasmonic acid
| |

| |
| Names | |
|---|---|
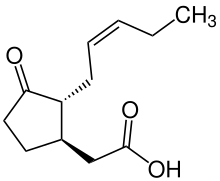
| Preferred IUPAC name
{(1R,2R)-3-Oxo-2-[(2Z)-pent-2-en-1-yl]cyclopentyl}acetic acid | |
| Other names
Jasmonic acid
(−)-Jasmonic acid JA (1R,2R)-3-Oxo-2-(2Z)-2-pentenylcyclopentylethanoic acid {(1R,2R)-3-Oxo-2-[(2Z)-2-penten-1-yl]cyclopentyl}acetic acid | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H18O3 | |
| Molar mass | 210.27 g/mol |
| Density | 1.1 g/cm3 |
| Boiling point | 160 °C (320 °F; 433 K) at 0.7 mmHg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Jasmonic acid (JA) is an organic compound found in several plants including jasmine. The molecule is a member of the jasmonate class of plant hormones. It is biosynthesized from linolenic acid by the octadecanoid pathway. It was first isolated in 1957 as the methyl ester of jasmonic acid by the Swiss chemist Édouard Demole and his colleagues.[1]
Biosynthesis
Its biosynthesis starts from the fatty acid linolenic acid, which is oxygenated by lipoxygenase (13-LOX), forming a hydroperoxide. This peroxide then cyclizes in the presence of allene oxide synthase to form an allene oxide. The rearrangement of allene oxide to form 12-oxophytodienoic acid is catalyzed by the enzyme allene oxide cyclase. A series of β-oxidations results in 7-isojasmonic acid. In the absence of enzyme, this isojasmonic acid isomerizes to jasmonic acid.[2]

Function
The major function of JA and its various metabolites is regulating plant responses to abiotic and biotic stresses as well as plant growth and development.
JA may have a role in pest control.
Derivatives
Jasmonic acid is also converted to a variety of derivatives including the ester methyl jasmonate. This conversion is catalyzed by the jasmonic acid carboxyl methyltransferase enzyme.[14] It can also be conjugated to amino acids in some biological contexts. Decarboxylation affords the related fragrance jasmone.
References
- .
- Chapuis, Christian (December 2011). "The chemistry and creative legacy of methyl jasmonate and hedione". Perfumer & Flavorist. 36: 36–48.
- ISBN 978-0-470-74168-9.
- PMID 16807821.
- PMID 18657055.
- ^ Lutz, Diana (2012). "Key part of plants' rapid response system revealed". Washington University in St. Louis.
- PMID 14976235.
- ^ The Effects of Bacterial and Jasmonic Acid Treatments on Insects of Canola. 2008.
- ^ "Success for plants' pest control". BBC News. 2008-10-07. Retrieved 2010-05-05.
- PMID 22142268.
- PMID 16668777.
- S2CID 89119004.
- S2CID 10778582.
- ^ PMID 21216178.
- PMID 11287667.
- Sankawa, Ushio; Barton, Derek H. R.; Nakanishi, Koji; Meth-Cohn, Otto, eds. (1999). Comprehensive Natural Products Chemistry: Polyketides and Other Secondary Metabolites Including Fatty Acids and Their Derivatives. Pergamon Press. ISBN 978-0-08-043153-6.
