Retinoic acid
| |

| |
| Names | |
|---|---|
| IUPAC name
Retinoic acid
| |
| Systematic IUPAC name
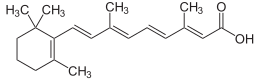
(2E,4E,6E,8E)-3,7-Dimethyl-9-(2,6,6-trimethylcyclohex-1-en-1-yl)nona-2,4,6,8-tetraenoic acid | |
| Other names
vitamin A acid; RA
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
IUPHAR/BPS |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C20H28O2 | |
| Molar mass | 300.43512 g/mol |
| Appearance | yellow to light orange crystalline powder with a characteristic of a floral scent[1] |
| Melting point | 180 to 182 °C (356 to 360 °F; 453 to 455 K) crystals from ethanol[1] |
| nearly insoluble | |
| Solubility in fat | soluble |
| Related compounds | |
Related compounds
|
beta-carotene
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Retinoic acid (used simplified here for all-trans-retinoic acid) is a
All-trans-retinoic acid (ATRA) is the major occurring retinoic acid, while isomers like 13-cis- and 9-cis-retinoic acid are also present in much lower levels.[4]
The key role of all-trans-retinoic acid in embryonic development mediates the high
Mechanism of biological action
All-trans-retinoic acid acts by binding to the
The molecular basis for the interaction between all-trans-retinoic acid and the Hox genes has been studied by using deletion analysis in
Biosynthesis
All-trans-retinoic acid can be produced in the body by two sequential oxidation steps that convert all-trans-retinol to
Function in the absence of precursors
All-trans-retinoic acid is responsible for most of the activity of vitamin A1, save visual pigment effects that require retinal (retinaldehyde), and cell metabolism effects that may require retinol itself. Also, some biochemical functions necessary for fertility in vitamin A deficient male and female mammals originally appeared to require all-trans-retinol for rescue, but this is due to a requirement for local conversion of all-trans-retinol to all-trans-retinoic acid, as administered all-trans-retinoic acid does not reach some critical tissues unless given in high amounts. Thus, if animals are fed only all-trans-retinoic acid but no vitamin A1 (all-trans-retinol or retinal), they suffer none of the growth-stunting or epithelial-damaging effects of lack of vitamin A1 (including no xerophthalmia—dryness of the cornea). They do suffer retina degeneration and blindness, due to retinal deficiency.
In addition, vitamin A1-deprived but all-trans-retinoic acid-supplemented male rats exhibit hypogonadism and infertility due to lack of local retinoic acid synthesis in the testis; similar treatment of female rats causes infertility due to fetal resorption caused by a lack of local retinoic acid synthesis in the embryo.[10][11] The retinoic acid synthesis in testes is catalyzed primarily by the ALDH1A2 (RALDH2) aldehyde dehydrogenase. Suppressing this enzyme has been proposed as a possible way to make a male contraceptive pill, because retinoic acid is necessary for spermatogenesis in humans, much as in rats.[12]
Function in embryonic development
All-trans-retinoic acid (ATRA) is a
Related pharmaceuticals
- Talarozole
- Tretinoin / all-trans-retinoic acid (Tradename: Retin-A)
- Isotretinoin / 13-cis-retinoic acid (Tradename: Accutane(US), Roaccutane)
References
- ^ a b Merck Index, 13th Edition, 8251.
- ^ PMID 18805086.
- ^ S2CID 5549210.
- PMID 30358857.
- PMID 24005729.
- ISBN 1-56081-706-2.
- ^
Marshall H, Morrison A, Studer M, Pöpperl H, Krumlauf R (1996). "Retinoids and Hox genes". S2CID 16062049.
- ^ "ALDH 1 Family". Dr. Vasilis Vasiliou's laboratory at the University of Colorado's Health Sciences Center. Archived from the original on 13 January 2013. Retrieved 22 October 2012.
- PMID 15193143.
- S2CID 34221571.
- PMID 1989855.
- PMID 23087225.
- PMID 25371368.
- ^ PMID 25560970.
- PMID 22318625.
