(Diacetoxyiodo)benzene

| |
| Names | |
|---|---|
| Preferred IUPAC name
Phenyl-λ3-iodanediyl diacetate | |
| Other names
Bis(acetoxy)(phenyl)iodane
Bis(acetato-O)phenyliodine Bis(acetoxy)iodobenzene (BAIB) (Diacetoxyiodo)benzene I,I-Diacetatoiodobenzene Iodobenzene diacetate Iodosobenzene I,I-diacetate Phenyliodine(III) diacetate (PIDA) Phenyliodo diacetate | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.019.826 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H11IO4 | |
| Molar mass | 322.098 g·mol−1 |
| Appearance | white powder |
| Melting point | 163–165 °C (325–329 °F; 436–438 K) |
| reacts | |
| Solubility | soluble in acetic acid, acetonitrile, dichloromethane |
| Structure[1][2] | |
| orthorhombic | |
| Pnn2 | |
a = 15.693(3) Å, b = 8.477(2) Å, c = 8.762(2) Å[2]
| |
| T-shaped molecular geometry | |
| Related compounds | |
Related compounds
|
(Bis(trifluoroacetoxy)iodo)benzene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
(Diacetoxyiodo)benzene, also known as
6H
5I(OCOCH
3)
2. It is used as an oxidizing agent in organic chemistry
Preparation
This reagent was originally prepared by Conrad Willgerodt[3] by reacting iodobenzene with a mixture of acetic acid and peracetic acid:[4][5]
PIDA can also be prepared from
More recent preparations direct from
The PIDA molecule is termed
Unconventional reactions
One use of PIDA is in the preparation of similar reagents by
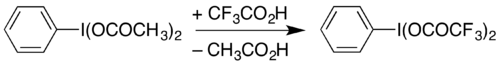
PIFA can be used to carry out the Hofmann rearrangement under mildly acidic conditions,[11] rather than the strongly basic conditions traditionally used.[12][13] The Hofmann decarbonylation of an N-protected asparagine has been demonstrated with PIDA, providing a route to β-amino-L-alanine derivatives.[14]
PIDA is also used in Suárez oxidation, where photolysis of hydroxy compounds in the presence of PIDA and iodine generates cyclic ethers.[15][16][17] This has been used in several total syntheses, such as the total synthesis of (−)-majucin, (−)-Jiadifenoxolane A,[18] and cephanolide A.[19]
References
- ^ .
- ^ .
- .
- ; Collected Volumes, vol. 5, p. 660.
- ^ ISBN 9780470842898.
- PMID 16095332.
- .
- ^ ISBN 9781118878651.
- ISBN 9781118878651.
- ; Collected Volumes, vol. 8, p. 132.
- ISBN 9780080977430.
- .
- ISBN 9781483258683.
- .
- .
- .
- .
- PMID 29148748.
- S2CID 249314606.
