Ammonium oxalate
| |
| Names | |
|---|---|
| IUPAC name
Ammonium oxalate
| |
| Systematic IUPAC name
Ammonium ethanedioate | |
| Other names
Diammonium oxalate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.012.912 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
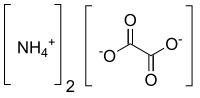
| [NH4]2C2O4 | |
| Molar mass | 124.096 g·mol−1 |
| Appearance | Colorless or white crystalline solid |
| Density | 1.5 g/cm3[1] |
| Melting point | 70 C (158 F, 343.15 K) |
| 5.20 g/(100 ml) (25 °C)[1] | |
| Hazards | |
| GHS labelling: | |
| H302, H312, H319 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium oxalate is a
monohydrate
([NH4]2C2O4·H2O). It is a colorless or white salt under standard conditions and is odorless and non-volatile. It occurs in many plants and vegetables.
Vertebrate
It is produced in the body of
.Mineralogy
Oxammite is a natural mineral form of ammonium oxalate. This mineral is extremely rare. It is an organic mineral derived from guano.[5]
Chemistry
Ammonium oxalate is used as an analytical reagent and general reducing agent.[2] It and other oxalates are used as anticoagulants, to preserve blood outside the body.[citation needed]
Earth sciences
Acid ammonium oxalate (ammonium oxalate acidified to pH 3 with oxalic acid) is commonly employed in soil chemical analysis to extract iron and aluminium from poorly-crystalline minerals (such as ferrihydrite), iron(II)-bearing minerals (such as magnetite) and organic matter.[6][page needed]
References
- ^ ISBN 978-1138561632.
- ^ a b National Center for Biotechnology Information. PubChem Compound Database; CID 14213 (accessed 15 November 2016).
- ISBN 92-4-156301-X.
- ^ N G Coley, "The collateral sciences in the work of Golding Bird (1814-1854)", Medical History, iss.4, vol.13, October 1969, pp.372.
- ^ "Home". mindat.org.
- ISBN 9780643101364.
