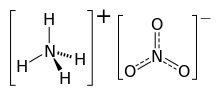
Ammonium nitrate
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Ammonium nitrate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.026.680 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 2426 – liquid
|
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| NH4NO3 | |
| Molar mass | 80.043 g/mol |
| Appearance | white crystalline solid |
| Density | 1.725 g/cm3 (20 °C) |
| Melting point | 169.6 °C (337.3 °F; 442.8 K) |
| Boiling point | approx. 210 °C (410 °F; 483 K) decomposes |
| Endothermic 118 g/100 ml (0 °C) 150 g/100 ml (10 °C) 192 g/100 ml (20 °C) 297 g/100 ml (40 °C) 410 g/100 ml (60 °C) 576 g/100 ml (80 °C) 1024 g/100 ml (100 °C)[1] | |
| −33.6×10−6 cm3/mol | |
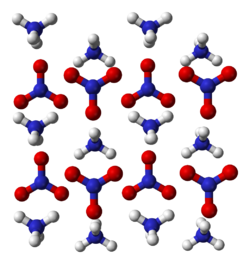
| Structure | |
| orthorhombic[2] | |
| Explosive data | |
| Shock sensitivity | very low |
| Friction sensitivity | very low |
| Detonation velocity | 2500 m/s |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Explosive, Oxidizer |
| GHS labelling: | |
  
| |
| Danger | |
| H201, H271, H319 | |
| P220, P221, P264, P271, P280, P372 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
2085–5300 mg/kg (oral in rats, mice)[3] |
| Related compounds | |
Other anions
|
Ammonium nitrite |
Other cations
|
Sodium nitrate Potassium nitrate Hydroxylammonium nitrate |
Related compounds
|
Ammonium perchlorate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium nitrate is a
Its other major use is as a component of explosive mixtures used in mining, quarrying, and civil construction. It is the major constituent of ANFO, an industrial explosive which accounts for 80% of explosives used in North America; similar formulations have been used in improvised explosive devices.
Many countries are phasing out its use in consumer applications due to concerns over its potential for misuse.[6] Accidental ammonium nitrate explosions have killed thousands of people since the early 20th century.[6][7] Global production was estimated at 21.6 million tonnes in 2017.[8] By 2021, global production of ammonium nitrate was down to 16.7 million tonnes.[7]
Occurrence
Ammonium nitrate is found as the natural mineral
Production, reactions and crystalline phases
The industrial production of ammonium nitrate entails the
- HNO3 + NH3 → NH4NO3
The ammonia required for this process is obtained by the
Another production method is a variant of the nitrophosphate process:
The products, calcium carbonate and ammonium nitrate, may be separately purified or sold combined as calcium ammonium nitrate.
Ammonium nitrate can also be made via metathesis reactions:
- (NH4)2SO4 + Ba(NO3)2 → 2 NH4NO3 + BaSO4
- (NH4)2SO4 + Ca(NO3)2 → 2 NH4NO3 + CaSO4
- NH4Cl + AgNO3 → NH4NO3 + AgCl
Reactions
As ammonium nitrate is a salt, both the cation, NH+4, and the anion, NO−3, may take part in chemical reactions.
Solid ammonium nitrate decomposes on heating. At temperatures below around 300 °C, the decomposition mainly produces nitrous oxide and water:
- NH4NO3 → N2O + 2 H2O
At higher temperatures, the following reaction predominates.[13]
- 2 NH4NO3 → 2 N2 + O2 + 4 H2O
Both decomposition reactions are
The red–orange colour in an explosion cloud is due to nitrogen dioxide, a secondary reaction product.[14]
Crystalline phases
Several crystalline phases of ammonium nitrate have been observed. The following occur under atmospheric pressure.
Phase Temperature (°C) Symmetry (liquid) (above 169.6) I 169.6 to 125.2 cubic II 125.2 to 84.2 tetragonal III 84.2 to 32.3 α-rhombic IV 32.3 to −16.8 β-rhombic V below −16.8 tetragonal[15]
The transition between β-rhombic to α-rhombic forms (at 32.3 °C) occurs at ambient temperature in many parts of the world. These forms have a 3.6% difference in density and hence transition between them causes a change in volume. One practical consequence of this is that ammonium nitrate cannot be used as a
Applications
Fertilizer
Ammonium nitrate is an important fertilizer with
(46-0-0), giving ammonium nitrate a slight transportation disadvantage. Ammonium nitrate's advantage over urea is that it is more stable and does not rapidly lose nitrogen to the atmosphere.Explosives
Ammonium nitrate readily forms explosive mixtures with varying properties when combined with explosives such as TNT or with fuels like aluminium powder or fuel oil. Examples of explosives containing ammonium nitrate include:
- Amatex (ammonium nitrate, TNT and RDX)
- Amatol (ammonium nitrate and TNT)
- Ammonal (ammonium nitrate and aluminum powder)
- ANFO (ammonium nitrate and fuel oil)
- Astrolite (ammonium nitrate and hydrazine rocket fuel)
- Goma-2 (ammonium nitrate, nitroglycol, nitrocellulose, dibutyl phthalate and fuel)
- Minol (explosive) (ammonium nitrate, TNT and aluminum powder)
- Nitrolite (ammonium nitrate, TNT and nitroglycerin +)
- DBX (ammonium nitrate, RDX, TNT and aluminum powder)
- Tovex (ammonium nitrate and methylammonium nitrate)
Mixture with fuel oil
ANFO is a mixture of 94% ammonium nitrate ("AN") and 6%
Terrorism
Ammonium nitrate-based explosives were used in the Sterling Hall bombing in Madison, Wisconsin, 1970, the Oklahoma City bombing in 1995, the 2011 Delhi bombings, the 2011 bombing in Oslo, the Myyrmanni bombing and the 2013 Hyderabad blasts.
In November 2009, the government of the
Niche uses
Ammonium nitrate is used in some
It was once used, in combination with independently explosive "fuels" such as
Safety, handling, and storage
Numerous safety guidelines are available for storing and handling ammonium nitrate. Health and safety data are shown on the safety data sheets available from suppliers and from various governments.[25][26][27]
Pure ammonium nitrate does not burn, but as a strong oxidizer, it supports and accelerates the combustion of organic (and some inorganic) material.[25][28][29] It should not be stored near combustible substances.
While ammonium nitrate is stable at ambient temperature and pressure under many conditions, it may detonate from a strong initiation charge. It should not be stored near high explosives or blasting agents.
Molten ammonium nitrate is very sensitive to shock and detonation, particularly if it becomes contaminated with incompatible materials such as combustibles, flammable liquids, acids, chlorates, chlorides, sulfur, metals, charcoal and sawdust.[30][25]
Contact with certain substances such as
Ammonium nitrate begins decomposition after melting, releasing NOx, HNO3, NH
3 and H2O. It should not be heated in a confined space.[25] The resulting heat and pressure from decomposition increases the sensitivity to detonation and increases the speed of decomposition. Detonation may occur at 80 atmospheres. Contamination can reduce this to 20 atmospheres.[30]
Ammonium nitrate has a critical relative humidity of 59.4% at 30 °C. At higher humidity it will absorb moisture from the atmosphere. Therefore, it is important to store ammonium nitrate in a tightly sealed container. Otherwise, it can coalesce into a large, solid mass. Ammonium nitrate can absorb enough moisture to liquefy. Blending ammonium nitrate with certain other fertilizers can lower the critical relative humidity.[33]
The potential for use of the material as an explosive has prompted regulatory measures. For example, in Australia, the Dangerous Goods Regulations came into effect in August 2005 to enforce licensing in dealing with such substances.[34] Licenses are granted only to applicants (industry) with appropriate security measures in place to prevent any misuse.[35] Additional uses such as education and research purposes may also be considered, but individual use will not. Employees of those with licenses to deal with the substance are still required to be supervised by authorized personnel and are required to pass a security and national police check before a license may be granted.
Health hazards
Ammonium nitrate is not hazardous to health and is usually used in fertilizer products.[36][37][38]
Ammonium nitrate has an LD50 of 2217 mg/kg,[39] which for comparison is about two-thirds that of table salt.
Disasters
Ammonium nitrate decomposes, non-explosively, into the
Ammonium nitrate can explode through two mechanisms:
- Shock induced detonation. An explosive charge within or in contact with a mass of ammonium nitrate causes the ammonium nitrate to detonate. Examples of such disasters are .
- ). The fire must be confined at least to a degree for successful transition from a fire to an explosion.
See also
References
- ISBN 0-07-049439-8
- ISSN 2073-4352.
- ISBN 1-903996-65-1.
- ^ "Hazard Rating Information for NFPA Fire Diamonds". Archived from the original on 17 February 2015. Retrieved 13 March 2015.
- ISBN 9783527303854.
- ^ a b Ammonium nitrate sold by ton as U.S. regulation is stymied. Archived 28 February 2018 at the Wayback Machine – The Dallas Morning News
- ^ a b "Ammonium nitrate production by country, 2023 - knoema.com". Knoema.
- ^ "Ammonium nitrate production by country, 2019". Knoema. Retrieved 14 August 2020.
- ^ "Gwihabaite". www.mindat.org.
- ^ "Niter". www.mindat.org.
- ^ "List of Minerals". www.ima-mineralogy.org. 21 March 2011.
- ^ US 4927617, Villard, Alexandre & Cotonea, Yves, "Process of producing concentrated solutions of ammonium nitrate", published 22 May 1990, assigned to Societe Chimique des Charbonnages S.A.
- ISBN 978-0-08-037941-8.
- ^ a b "The chemistry behind the Beirut explosion". Chemical & Engineering News.
- .
- .
- ^ "Nutrient Content of Fertilizer Materials" (PDF). Archived from the original (PDF) on 24 December 2012. Retrieved 27 June 2012.
- ^ ASIN B0000EGDJT.
- ^ Brook, Tom Vanden. "Afghan bomb makers shifting to new explosives for IEDs". USA TODAY.
- ^ Coxworth, Ben (20 September 2021). "Sunlight and salt water join forces in electricity-free cooling system". New Atlas. Gizmag Pty Ltd. Retrieved 21 September 2021.
- ^ US 5531941, Poole, Donald R., "Process for preparing azide-free gas generant composition", published 2 July 1996, assigned to Automotive Systems Laboratory
- ^ Airbag Compound Has Vexed Takata for Years – The New York Times
- ^ A Cheaper Airbag, and Takata's Road to a Deadly Crisis. – The New York Times
- ^ "Takata Spotlight". NHTSA.gov. Retrieved 31 May 2024.
- ^ a b c d Chemical Advisory: Safe Storage, Handling, and Management of Ammonium Nitrate United States Environmental Protection Agency
- ^ "Storing and handling ammonium nitrate" (PDF). Archived (PDF) from the original on 4 July 2011. Retrieved 22 March 2006.
- ^ "Ammonium nitrate MSDS". Archived from the original on 18 August 2011. Retrieved 25 January 2012.
- ISBN 0-07-049439-8.
- ^ "Ammonium nitrate". PubChem. Retrieved 6 August 2020.
- ^ a b "Report for Kooragang Island Update PHA MOD1 Report". Orica Mining Services. 1 April 2012. Archived from the original on 12 August 2014. Retrieved 6 August 2020.
- ^ "Chemical Engineering Transactions" (PDF). Archived from the original (PDF) on 14 April 2016.
- ^ "Ammonium Nitrate". webwiser.nlm.nih.gov. Retrieved 6 August 2020.
- ^ Fertilizers Europe (2006). "Guidance for Compatibility of Fertilizer Blending Materials" (PDF). Archived (PDF) from the original on 8 June 2015.
- ^ "Dangerous Goods (HCDG) Regulations" (PDF). Archived (PDF) from the original on 23 March 2012.
- ^ Ammonium Nitrate-Regulating its use, Balancing Access & Protection from "Worksafe Victoria". Archived from the original on 11 March 2011.
- ^ CF Industries. "Ammonium nitrate MSDS" (PDF). Archived from the original (PDF) on 27 March 2014.
- ^ "Chemicalland21 – Ammonium Nitrate". Archived from the original on 10 January 2012.
- ^ "Ammonium Nitrate". Paton Fertilizers Pty Ltd. 2005.
- ^ "Material Safety Data Sheet, Ammonium nitrate MSDS". Archived from the original on 18 August 2011. Retrieved 25 January 2012.
- S2CID 94427830.
- ^ "Lebanon's president calls for two-week state of emergency in Beirut after blast". Reuters. Beirut. 4 August 2020. Retrieved 4 August 2020.
Aoun, in remarks published on the Presidency Twitter account, said it was "unacceptable" that 2,750 tonnes of ammonium nitrate was stored in a warehouse for six years without safety measures and vowed that those responsible would face the "harshest punishments".
Sources
- Properties: UNIDO and International Fertilizer Development Center (1998), Fertilizer Manual, Kluwer Academic Publishers, ISBN 0-7923-5032-4.
External links
- International Chemical Safety Card 0216
- "Storing and Handling Ammonium Nitrate", United Kingdom Health and Safety Executive publication INDG230 (1986)
- Chemical Advisory: Safe Storage, Handling, and Management of Ammonium Nitrate United States Environmental Protection Agency
- Calculators: surface tensions, and densities, molarities and molalities of aqueous ammonium nitrate

