Apoptosome
The apoptosome is a large quaternary
In mammalian cells, once
Once formed, the apoptosome can then recruit and activate the inactive pro-caspase-9. Once activated, this initiator caspase can then activate effector caspases and trigger a cascade of events leading to apoptosis.
History
The term Apoptosome was first introduced in Yoshihide Tsujimoto's 1998 paper "Role of
The criteria for an apoptosome were laid out in 1999. Firstly, it must be a large complex (greater than 1.3 million daltons). Secondly its formation requires the hydrolysis of a high energy bond of ATP or dATP. And lastly it must activate procaspase-9 in its functional form. The formation of this complex is the point of no return, and apoptosis will occur. The stable APAF-1 and cytochrome multimeric complex fit this description, and is now called the apoptosome.[7]
The apoptosome was thought to be a multimeric complex for two reasons. Firstly, to bring multiple procaspase-9 molecules close together for cleavage. And secondly, to raise the threshold for apoptosis, therefore nonspecific leakage of cytochrome c would not result in apoptosis.[7]
Once the apoptosome was established as the procaspase-9 activator, mutations within this pathway became an important research area. Some examples include human leukemia cells, ovarian cancer and viral infections.[8][9][10] Current research areas for this pathway will be discussed in further detail. There are hidden routes for cell death as well, which are independent of APAF-1 and therefore the apoptosome. These routes are also independent of caspase-3 and 9. These hidden pathways for apoptosis are slower, but may prove useful with further research.[11]

(Yuan et al. 2010, Structure of an apoptosome-procaspase-9 CARD complex[1]
Structure
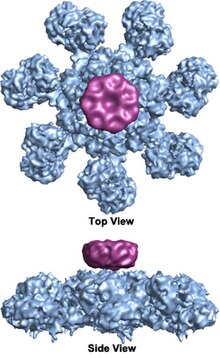
The apoptosome is a multimolecular
The wheel-shaped

Evidence from Wang and colleagues indicates that the stoichiometric ratio of procaspase-9 to Apaf-1 within the complex is approximately 1:1 .[7] This was further proved by quantitative mass spectrometry analysis.[13] The stoichiometry of cytochrome c to Apaf-1 within the complex is proved to be 1:1.[1] There are some debates about whether stable incorporation of cytochrome c into the apoptosome is required following oligomerization, but recent structural data favor the idea that cytochrome c stabilizes the oligomeric human apoptosome.[1] However, cytochrome c may be not required for the assembly of apoptosome in non-mammalian species, such as worms and fruit flies.[14] In addition, several other molecules, most notably caspase-3, have been reported to co-purify with the apoptosome[7] and caspase-3 has been proved to be able to bind the apoptosome-procaspase-9 complex.[13]
Apaf-1 forms the backbone of the apoptosome. It has three distinct regions: the N-terminal caspase-recruitment domain (CARD, residues 1–90), a central nucleotide-binding and oligomerization region (NB-ARC/NOD, 128–586) and a C-terminal WD40 region (613–1248) making up a protein about 140 KDa.[2]
- The CARD domain of Apaf-1 interacts with procaspase-9 and involved with recruitment within the apoptosome.[2]
- The NB-ARC/NOD region exhibits significant sequence similarity to the C. elegans Ced-4 protein.[2]
- The C-terminal WD40 region of Apaf1 contains 15 WD-40 repeats structured into two b-propeller-shaped domains.[1] WD-40 repeats are sequences around 40 amino acids long which end in Trp-Asp and are typically involved in protein–protein interaction.[2]
A short linker and nucleotide binding a/b domains (NBD) that contain conserved Walker boxes A (p-loop 155-161) and B (239-243) follow the N-terminal CARD domain.

Apoptosomes in other organisms
The above descriptions are for the human apoptosome. Apoptosome complex structures from other organisms have many similarities, but are of quite different sizes and numbers of subunits, as shown in the figure. The fruit-fly system, called Dark, has a ring of 8 subunits (PDB 4V4L).[14] The nematode apoptosome, called CED-4, is octameric but much smaller (PDB 3LQQ), and it does not include the regions that would bind cytochrome C.[15]
Mechanism of Action
Initiation
The initiation of apoptosome action corresponds with the first steps in the
Cytochrome c Release
Cytochrome c release is proposed to take place in one of two ways. Firstly, the
Apaf-1
a. Absence of Cytochrome c
In the absence of cytochrome c, Apaf-1 exists in its monomeric form; it is thought that the WD-40 domain remain folded back onto the protein, keeping Apaf-1 in an auto inhibited state.[16] In addition, several areas are so tightly bound that the protein is unable to bind to anything else.[16] It has been determined through mass spectrometry that in the autoinhibited, or "locked" state, ADP is bound to the ATPase domain of Apaf-1.[16] In this state, this protein is singular, and incapable of activating any caspases.
b. Presence of Cytochrome c
Cytochrome c binds to the WD-40 domain of Apaf-1.
Active Apoptosome Action
This functional apoptosome then can provide a platform activation of caspase 9.[1][16] Caspase 9 exists as a zymogen in the cytosol and is thought to be found at 20 nM in cells.[16] Though it is known that the zymogen does not need to be cleaved in order to become active,[16] the activity of procaspase-9 may increase significantly once cleaved.[13] The first hypothesis is that the apoptosome provides a location for the dimerization of two caspase 9 molecules before cleavage; this hypothesis was favoured by Reidl & Salvasen in 2007. The second is that cleavage takes place while caspase 9 is still in its monomeric form.[13][16] In each case, caspase 9 activation leads to the activation of a full caspase cascade and subsequent cell death. It has been suggested that the evolutionary reason for the multimeric protein complex activating the caspase cascade is to ensure trace amounts of cytochrome c do not accidentally cause apoptosis.[7]
Research Areas
What happens when mutations occur?
While apoptosis is required for natural body function, mutations of the apoptosome pathway cause catastrophic effects and changes in the body. Mutations of the cell pathway can either promote cell death or disallow cell death creating a huge amount of disease in the body. Mutated apoptosis pathways causing disease are plentiful and have a wide range from cancer, due to lack of apoptosome activity,
Repression of Apoptosis causing cancer
Genetic and biochemical abnormalities within a cell normally trigger programmed cell death to rid the body of irregular cell function and development; however, cancer cells have acquired mutations that allow them to repress apoptosis and survive. Chemotherapies like ionizing radiation have been developed to activate these repressed PCD pathways by hyper-stimulation to promote normal PCD.[19]
P53 mutations in Apoptosis
Targeting the Apoptosome for Cancer therapy
The inhibition of apoptosis is one of the key features of cancer so finding ways to manipulate and overcome this inhibition to form the apoptosome and activate caspases are important in the development of new cancer treatments.[20] The ability to directly cause apoptosome activation is valuable in cancer therapies because the infected cancerous genes are unable to be destroyed causing a continuation of the cancer to form. By activating the apoptosome by an outside stimulus apoptosis can occur and get rid of the mutated cells. Numerous approaches to achieve this are currently being pursued including recombinant biomolecules, antisense strategies, gene therapy and classic organic combinatorial chemistry to target specific apoptotic regulators in the approach to correct excessive or deficient cell death in human diseases.[18]
In general the up regulation of anti-apoptotic proteins leads to the prevention of apoptosis which can be solved by inhibitors and the down regulation of anti-apoptotic proteins leads to the induction of apoptosis which is reversed by activators that are able to bind and modify their activity. An important target molecule in apoptosis based therapies is
Another targeted molecule for cancer therapy involves the caspase family and their regulators. The inhibition of caspase activity blocks cell death in human disease including neurodegenerative disorders, stroke, heart attack and liver injury. Therefore, caspase inhibitors are a promising pharmacological tool providing treatments for stroke and other human diseases. There are several caspase inhibitors that are currently in the preclinical stage that have shown promising evidence of reversing effects of some neurodegenerative diseases. In a recent study researchers developed a reversible
Apoptosome complex has revealed new potential targets for molecular therapy
The Apaf1/caspase-9 apoptosome formation is a crucial event in the apoptotic cascade. The identification of new potential drugs that prevent or stabilize the formation of active apoptosome complex is the ideal strategy for the treatment of disease characterized by excessive or insufficient apoptosis.[18] Recently taurine has been found to prevent ischemia-induced apoptosis in cardiomyocytes through its ability to inhibit Apaf1/caspase-9 apoptosome formation without preventing mitochondrial dysfunction. The possible mechanism by which taurine inhibits the apoptosome formation was identified as being capable of reducing the expression of caspase-9, a fundamental component of apoptosome. However, there are studies that show Apaf1 and caspase-9 have independent roles other than the apoptosome so altering their levels could alter cell function as well. So despite encouraging experimental data several problems remain unsolved and limit the use of experimental drugs in clinical practice.[18]
The discovery of apoptosome inhibitors will provide a new therapeutical tool for the treatment of apoptosis mediated disease. Of particular importance are those new compounds able to inhibit apoptosome stability and activity, by acting on intracellular protein–protein interactions without altering the transcriptional levels of the apoptosome components.[18] Recent structural studies of apoptosome may provide valuable tools for designing apoptosome-based therapies.[1][13]
See also
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w Yuan S, Yu X, Topf M, Ludtke SJ, Wang X, Akey CW. "Structure of an apoptosome-procaspase-9 CARD complex." Structure. 2010 May;18(5):571-83.
- ^ PMID 11864614.
- PMID 21827944.
- PMID 9990505.
- PMID 9488720.
- PMID 9837928.
- ^ PMID 10206961.
- PMID 11435311.
- PMID 12021264.
- PMID 11429402.
- PMID 12773531.
- ^ )
- ^ a b c d e Yuan S, Yu X, Asara JM, Heuser JE, Ludtke SJ and Akey CW. "The Holo-Apoptosome: Activation of Procaspase-9 and Interactions with Caspase-3." Structure. 2011 August;19(8):1084-1096.
- ^ a b Yuan S, Yu X, Topf M, Dorstyn L, Kumar S, Ludtke SJ and Akey CW. "Structure of the Drosophila Apoptosome at 6.9 Å Resolution." Structure. 2011 January;19(1):128-140.
- PMID 20434985.
- ^ S2CID 3064077.
- ^ Diamond & McCabe (2007). The mitochondrion and plant programmed cell death. In: Logan DC (Ed.), Plant mitochondria. Annual Plant Review 2007, 31:308–334.
- ^ PMID 17674158.
- ^ PMID 20040274.
- S2CID 36028755.
