Betalain

Betalains are a class of red and yellow
Description
The name "betalain" comes from the
There are two categories of betalains:[3]
- Betacyanins include the reddish to violet betalain pigments. Among the betacyanins present in plants include betanin, isobetanin, probetanin, and neobetanin.
- Betaxanthins are those betalain pigments which appear yellow to orange. Among the betaxanthins present in plants include vulgaxanthin, miraxanthin, portulaxanthin, and indicaxanthin.
The physiological function of betalains in plants is uncertain, but there is some evidence that they may have fungicidal properties.[4] Additionally, betalains have been found in fluorescent flowers, though their role in these plants is also uncertain.[5]
-
Swiss chard, showing one plant expressing yellow betaxanthins and another expressing red betacyanins
-
Flowers of the cactus Mammillaria sp. contain betalains.
-
Inflorescences of Amaranthus caudatus (love-lies-bleeding) contain large quantities of betacyanins.
Chemistry
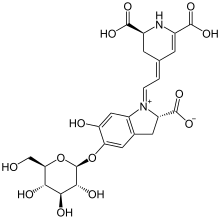
Betalains (betacyanins) were first isolated and its chemical structure discovered in 1960 at the University of Zurich by Dr. Tom Mabry.[6] It was once thought that betalains were related to anthocyanins, the reddish pigments found in most plants. Both betalains and anthocyanins are water-soluble pigments found in the vacuoles of plant cells. However, betalains are structurally and chemically unlike anthocyanins and the two have never been found in the same plant together.[7][8] For example, betalains contain nitrogen whereas anthocyanins do not.[2]
It is now known that betalains are
The most heavily studied betalain is
Other important betacyanins are
Biosynthesis

In the first step of the biosynthetic pathway, L-tyrosine is converted to L-3,4-dihydroxyphenylalanine (l-DOPA) by 3-hydroxylation by a cytochrome P450 enzyme. For dopa, the biosynthesis branches: a) on the one hand, its oxidation occurs by a CYP enzyme to cyclo-dopa;[13] (b) on the other hand, the aromatic ring of dopa is activated by a dopa-4,5-dioxygenase[14] opened to seco-dopa, from which betalamic acid is formed by spantane recyclization. This then reacts spontaneously with cyclo-dopa to form betanidine on the one hand, or on the other hand after its prior glucosylation by a cyclo-dopa glucosyltransferase[15] to red-purple betanin, the simplest betacyan. In addition, betalamic acid reacts spontaneously with various amino acids or amines to form yellow-orange colored betaxanthins (see figure). The diversity of betacyans results from different glucosylation of betanidine and its subsequent acylation with aliphatic and aromatic carboxylic acids.
Semisynthetic derivatives

Betanin extracted from the red beet[16] was used as starting material for the semisynthesis of an artificial coumarinic betalaine. The betanin was hydrolyzed to betalamic acid, and this was coupled to 7-amino-4-methylcoumarin. The resulting betalain was applied as a fluorescent probe for the live-cell imaging of Plasmodium-infected erythrocytes.[17]
Taxonomic significance
Betalain pigments occur only in the Caryophyllales and some
Among the flowering plant
Economic uses
Betanin is commercially used as a natural
See also
References
- PMID 12620337.
- ^ a b c Robinson T (1963). The Organic Constituents of Higher Plants. Minneapolis: Burgess Publishing. p. 292.
- ^ ISBN 978-0-534-15162-1.
- ^ Kimler LM (1975). "Betanin, the red beet pigment, as an antifungal agent". Botanical Society of America, Abstracts of Papers. 36.
- S2CID 4408230.
- ^ "Tom J. MABRY Obituary (1932 - 2015) Austin American-Statesman". Legacy.com. Retrieved 2021-12-03.
- ISBN 978-1-891127-00-7.
- ISSN 0168-9452.
- ISBN 978-0-7167-1007-3.
- .
- S2CID 93924750.
- PMID 24019934.
- .
- PMC 316306.
- .
- .
- PMID 23342028.
- ^ ISBN 978-0-231-03880-5.
- S2CID 220615978.
- ISSN 0168-9452.
- .
- PMID 15447903.
External links
 Media related to Betalains at Wikimedia Commons
Media related to Betalains at Wikimedia Commons- Betalain synthesis diagram



