Anthocyanin

Anthocyanins (from
Anthocyanins belong to a parent class of
Although approved as food and beverage
Anthocyanin-rich plants
Coloration
In flowers, the coloration that is provided by anthocyanin accumulation may attract a wide variety of animal pollinators, while in fruits, the same coloration may aid in seed dispersal by attracting herbivorous animals to the potentially-edible fruits bearing these red, blue, or purple colors.
Plant physiology
Anthocyanins may have a protective role in plants against extreme temperatures.[7][8] Tomato plants protect against cold stress with anthocyanins countering reactive oxygen species, leading to a lower rate of cell death in leaves.[7]
Light absorbance

The absorbance pattern responsible for the red color of anthocyanins may be complementary to that of green chlorophyll in photosynthetically active tissues such as young Quercus coccifera leaves. It may protect the leaves from attacks by herbivores that may be attracted by green color.[9]
Occurrence
Anthocyanins are found in the cell vacuole, mostly in flowers and fruits, but also in leaves, stems, and roots. In these parts, they are found predominantly in outer cell layers such as the epidermis and peripheral mesophyll cells.
Most frequently occurring in nature are the
Sometimes
In flowers
Anthocyanins occur in the flowers of many plants, such as the blue poppies of some Meconopsis species and cultivars.[13] Anthocyanins have also been found in various tulip flowers, such as Tulipa gesneriana, Tulipa fosteriana and Tulipa eichleri.[14]
In food
| Food source | Anthocyanin content in mg per 100 g |
|---|---|
Açaí |
410[15] |
| Blackcurrant | 190–270 |
| Aronia (chokeberry) | 1,480[16] |
| Marion blackberry | 317[17] |
Black crowberry |
4,180[18] |
| Black raspberry | 589[19] |
| Raspberry | 365 |
| Wild blueberry | 558[20] |
| Cherry | 122[21] |
| Queen Garnet plum | 277[22] |
| Redcurrant | 80–420 |
| Black rice | 60 [23] |
| Black bean | 213[24] |
| Blue corn (Maize) | 71[25] |
| Purple corn | 1,642 |
| Purple corn husks (dried) | 10× more than in kernels |
| Purple tomato (fresh) | 283 ± 46[26] |
Concord grape |
326[27] |
| Norton grape | 888[27] |
| Red cabbage (fresh) | c. 150[28] |
| Red cabbage (dried) | c. 1442[28] |

Plants rich in anthocyanins are
The highest recorded amount appears to be specifically in the
Nature, traditional agriculture methods, and plant breeding have produced various uncommon crops containing anthocyanins, including
Some tomatoes also have been modified genetically with
In leaves of plant foods
Content of anthocyanins in the leaves of colorful plant foods such as purple corn, blueberries, or
The color spectrum of grape berry leaves may be analysed to evaluate the amount of anthocyanins. Fruit maturity, quality, and harvest time may be evaluated on the basis of the spectrum analysis.[43]
Autumn leaf color

The reds, purples, and their blended combinations responsible for autumn foliage are derived from anthocyanins. Unlike carotenoids, anthocyanins are not present in the leaf throughout the growing season, but are produced actively, toward the end of summer.[2] They develop in late summer in the sap of leaf cells, resulting from complex interactions of factors inside and outside the plant. Their formation depends on the breakdown of sugars in the presence of light as the level of phosphate in the leaf is reduced.[1] Orange leaves in autumn result from a combination of anthocyanins and carotenoids.
Anthocyanins are present in approximately 10% of tree species in temperate regions, although in certain areas such as New England, up to 70% of tree species may produce anthocyanins.[2]
Colorant safety
Anthocyanins are approved for use as
Anthocyanin
In humans
Although anthocyanins have been shown to have antioxidant properties in vitro,[49] there is no evidence for antioxidant effects in humans after consuming foods rich in anthocyanins.[5][50][51] Unlike controlled test-tube conditions, the fate of anthocyanins in vivo shows they are poorly conserved (less than 5%), with most of what is absorbed existing as chemically modified metabolites that are excreted rapidly.[52] The increase in antioxidant capacity of blood seen after the consumption of anthocyanin-rich foods may not be caused directly by the anthocyanins in the food, but instead by increased uric acid levels derived from metabolizing flavonoids (anthocyanin parent compounds) in the food.[52] It is possible that metabolites of ingested anthocyanins are reabsorbed in the gastrointestinal tract from where they may enter the blood for systemic distribution and have effects as smaller molecules.[52]
In a 2010 review of
Chemical properties
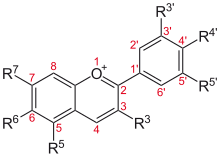
Flavylium cation derivatives
| Basic structure | Anthocyanidin | R3′ | R4′ | R5′ | R3 | R5 | R6 | R7 |
|---|---|---|---|---|---|---|---|---|

|
Aurantinidin | −H | −OH | −H | −OH | −OH | −OH | −OH |
| Cyanidin | −OH | −OH | −H | −OH | −OH | −H | −OH | |
| Delphinidin | −OH | −OH | −OH | −OH | −OH | −H | −OH | |
| Europinidin | −OCH 3 |
−OH | −OH | −OH | −OCH 3 |
−H | −OH | |
| Pelargonidin | −H | −OH | −H | −OH | −OH | −H | −OH | |
| Malvidin | −OCH 3 |
−OH | −OCH 3 |
−OH | −OH | −H | −OH | |
| Peonidin | −OCH 3 |
−OH | −H | −OH | −OH | −H | −OH | |
| Petunidin | −OH | −OH | −OCH 3 |
−OH | −OH | −H | −OH | |
| Rosinidin | −OCH 3 |
−OH | −H | −OH | −OH | −H | −OCH 3 |
Glycosides of anthocyanidins
The anthocyanins, anthocyanidins with sugar group(s), are mostly 3-
Stability
Anthocyanins are thought to be subject to
pH

Anthocyanins generally are degraded at higher pH. However, some anthocyanins, such as
Use as environmental pH indicator

Anthocyanins may be used as pH indicators because their color changes with pH; they are red or pink in acidic solutions (pH < 7), purple in neutral solutions (pH ≈ 7), greenish-yellow in alkaline solutions (pH > 7), and colorless in very alkaline solutions, where the pigment is completely reduced.[57]
Biosynthesis

- Anthocyanin pigments are assembled like all other flavonoids from two different streams of chemical raw materials in the cell:
- One stream involves the shikimate pathway to produce the amino acid phenylalanine, (see phenylpropanoids)
- The other stream produces three molecules of malonyl-CoA, a C3 unit from a C2 unit (acetyl-CoA),[58]
- One stream involves the
- These streams meet and are coupled together by the enzyme chalcone synthase, which forms an intermediate chalcone-like compound via a polyketide folding mechanism that is commonly found in plants,
- The chalcone is subsequently isomerized by the enzyme chalcone isomerase to the prototype pigment naringenin,
- Naringenin is subsequently oxidized by enzymes such as flavanone hydroxylase, flavonoid 3'-hydroxylase, and flavonoid 3',5'-hydroxylase,
- These oxidation products are further reduced by the enzyme dihydroflavonol 4-reductase to the corresponding colorless leucoanthocyanidins,[59]
- Leucoanthocyanidins once were believed to be the immediate precursors of the next enzyme, a dioxygenase referred to as anthocyanidin synthase, or, leucoanthocyanidin dioxygenase. Flavan-3-ols, the products of leucoanthocyanidin reductase (LAR), recently have been shown to be their true substrates,
- The resulting unstable anthocyanidins are further coupled to sugar molecules by enzymes such as UDP-3-O-glucosyltransferase,[60] to yield the final relatively-stable anthocyanins.
Thus, more than five enzymes are required to synthesize these pigments, each working in concert. Even a minor disruption in any of the mechanisms of these enzymes by either genetic or environmental factors, would halt anthocyanin production. While the biological burden of producing anthocyanins is relatively high, plants benefit significantly from the environmental adaptation, disease tolerance, and pest tolerance provided by anthocyanins.
In anthocyanin biosynthetic pathway, L-phenylalanine is converted to naringenin by phenylalanine ammonialyase, cinnamate 4-hydroxylase, 4-coumarate CoA ligase, chalcone synthase, and chalcone isomerase. Then, the next pathway is catalyzed, resulting in the formation of complex aglycone and anthocyanin through composition by flavanone 3-hydroxylase, flavonoid 3'-hydroxylase, dihydroflavonol 4-reductase,
Genetic analysis
The phenolic metabolic pathways and enzymes may be studied by mean of
Dye-sensitized solar cells
Anthocyanins have been used in
Visual markers
Anthocyanins
See also
- Phenolic compounds in wine
- p-Coumaroylated anthocyanin
References
- ^ ISBN 978-1-4051-1737-1.
- ^ PMID 19178979.
- ISBN 978-0470016176.
- ^ .
- ^ doi:10.2903/j.efsa.2010.1752.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ^ "Flavonoids". Micronutrient Information Center. Corvallis, Oregon: Linus Pauling Institute, Oregon State University. 2016. Retrieved 27 March 2022.
- ^ PMID 26943362.
- PMID 16760492.
- PMID 16452075.
- ISBN 978-1-891127-00-7.
- .
- ^ Stommel J, Griesbach RJ (September 2006). "Twice as Nice Breeding Versatile Vegetables". Agricultural Research Magazine, US Department of Agriculture. Retrieved 2 February 2016.
- ^ "Colour range within genus". Meconopsis Group. Archived from the original on 4 May 2020. Retrieved 30 June 2018.
- ^ N. Marissen, W. G. van Doorn and U. van Meeteren, International Society for Horticultural Science Proceedings of the Eighth International Symposium on Postharvest Physiology of Ornamental Plants, 2005, p. 248, at Google Books
- .
- ^ PMID 15612766.
- PMID 15612791.
- PMID 18522397.
- PMID 12033817.
- PMID 18052240.
- PMID 16719536.
- .
- PMID 19256557.
- .
- PMID 29086902.
- S2CID 14895646.
- ^ PMID 15506816.
- ^ PMID 24991694.
- .
- .
- PMID 27508114.
- S2CID 11221328.
- PMID 11743773.
- PMID 18065095.
- PMID 15612791.
- ^ a b Scott J (27 January 2012). "Purple tomato debuts as 'Indigo Rose'". Oregon State University Extension Service, Corvallis. Retrieved 9 September 2014.
- PMID 23707429.
- S2CID 14895646.
- ^ PMID 15740006.
- ISBN 978-0-643-09203-7.
- PMID 19007127.
- PMID 23875756.
- doi:10.1111/j.1755-0238.2011.00158.x. Archived from the original(PDF) on 19 October 2017. Retrieved 7 March 2015.
- ^ "Current EU approved additives and their E Numbers". United Kingdom: Food Standards Agency. 2010. Retrieved 12 August 2017.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 – Labelling of ingredients". 8 September 2011. Retrieved 27 October 2011.
- ^ "Summary of Color Additives for Use in the United States in Foods, Drugs, Cosmetics, and Medical Devices". US Food and Drug Administration. May 2015. Retrieved 29 June 2017.
- ^ "Summary of Color Additives for Use in the United States in Foods, Drugs, Cosmetics, and Medical Devices". US Food and Drug Administration. May 2015. Retrieved 13 August 2017.
- S2CID 233070680.
- S2CID 21174667.
- PMID 17157175.
- PMID 15019969.
- ^ a b c "Studies force new view on biology of flavonoids", by David Stauth, EurekAlert!. Adapted from a news release issued by Oregon State University
- PMID 14561507.
- ISBN 978-952-10-4846-3.
- PMID 19435353.
- hdl:10198/3206.
- .
- ^ Jack Sullivan (1998). "Anthocyanin". Carnivorous Plant Newsletter. Archived from the original on 1 November 2009. Retrieved 6 October 2009.
- PMID 11316805.
- PMID 20621794.
- .
- PMID 20073469.
- .
- .
- PMID 19762337.
- S2CID 15957685.
Further reading
- Andersen, O.M. (2006). Flavonoids: Chemistry, Biochemistry and Applications. Boca Raton FL: CRC Press. ISBN 978-0-8493-2021-7.
- Gould, K.; Davies, K.; Winefield, C., eds. (2008). Anthocyanins: Biosynthesis, Functions, and Applications. Springer. ISBN 978-0-387-77334-6.
