Biological aspects of fluorine
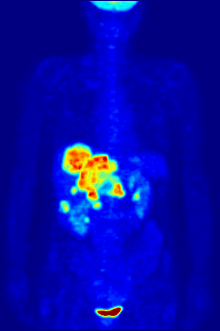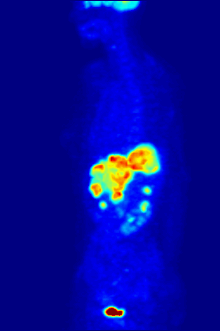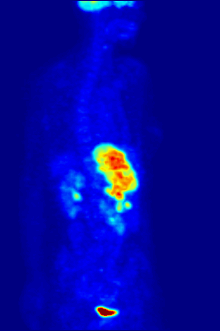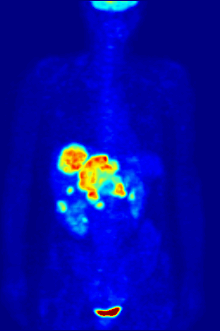
Aside from their use in medicine, man-made fluorinated compounds have also played a role in several noteworthy environmental concerns.
Fluorine biology is also relevant to a number of cutting-edge technologies. PFCs (
Dental care
Since the mid-20th century, it has been discerned from population studies (though incompletely understood) that fluoride reduces

When teeth begin to decay from the acid produced by sugar-consuming bacteria, calcium is lost (demineralization). However, teeth have a limited ability to recover calcium if decay is not too far advanced (remineralization). Fluoride appears to reduce demineralization and increase remineralization. Also, there is some evidence that fluoride interferes with the bacteria that consume sugars in the mouth and make tooth-destroying acids.[3] In any case, it is only the fluoride that is directly present in the mouth (topical treatment) that prevents cavities; fluoride ions that are swallowed do not benefit the teeth.[3]
Sodium fluoride, tin difluoride, and, most commonly, sodium monofluorophosphate, are used in toothpaste. In 1955, the first fluoride toothpaste was introduced in the United States. Now, almost all toothpaste in developed countries is fluoridated. For example, 95% of European toothpaste contains fluoride.[9] Gels and foams are often advised for special patient groups, particularly those undergoing radiation therapy to the head (cancer patients). The patient receives a four-minute application of a high amount of fluoride. Varnishes, which can be more quickly applied, exist and perform a similar function. Fluoride is also often present in prescription and non-prescription mouthwashes and is a trace component of foods manufactured using fluoridated water supplies.[11]
Medical applications
Pharmaceuticals
Of all commercialized pharmaceutical drugs, twenty percent contain fluorine, including important drugs in many different pharmaceutical classes.[12] Fluorine is often added to drug molecules during drug design, as even a single atom can greatly change the chemical properties of the molecule in desirable ways.
Because of the considerable stability of the
Adding fluorine to biologically active organic compounds increases their lipophilicity (ability to dissolve in fats), because the carbon–fluorine bond is even more hydrophobic than the carbon–hydrogen bond. This effect often increases a drug's bioavailability because of increased cell membrane penetration.[14] Although the potential of fluorine being released in a fluoride leaving group depends on its position in the molecule,[15] organofluorides are generally very stable, since the carbon–fluorine bond is strong.
Fluorines also find their uses in common mineralocorticoids, a class of drugs that increase the blood pressure. Adding a fluorine increases both its medical power and anti-inflammatory effects.[16] Fluorine-containing fludrocortisone is one of the most common of these drugs.[17] Dexamethasone and triamcinolone, which are among the most potent of the related synthetic corticosteroid class of drugs, contain fluorine as well.[17]
Several inhaled general anesthetic agents, including the most commonly used inhaled agents, also contain fluorine. The first fluorinated anesthetic agent, halothane, proved to be much safer (neither explosive nor flammable) and longer-lasting than those previously used. Modern fluorinated anesthetics are longer-lasting still and almost insoluble in blood, which accelerates the awakening.[18] Examples include sevoflurane, desflurane, enflurane, and isoflurane, all hydrofluorocarbon derivatives.[19]
Prior to the 1980s,

|

|

|

|
|---|---|---|---|
| Lipitor (atorvastatin) | 5-FU (fluorouracil) | Florinef (fludrocortisone) | Isoflurane |
Scanning

Compounds containing fluorine-18, a radioactive isotope that emits
Natural fluorine is monoisotopic, consisting solely of
Oxygen transport research
Liquid fluorocarbons have a very high capacity for holding gas in solution. They can hold more oxygen or carbon dioxide than blood does. For that reason, they have attracted ongoing interest related to the possibility of artificial blood or of liquid breathing.[28]

Blood substitutes are the subject of research because the demand for blood transfusions grows faster than donations. In some scenarios, artificial blood may be more convenient or safe. Because fluorocarbons do not normally mix with water, they must be mixed into emulsions (small droplets of perfluorocarbon suspended in water) in order to be used as blood.
Possible medical uses of liquid breathing (which uses pure perfluorocarbon liquid, not a water emulsion) involve assistance for premature babies or for burn patients (if normal lung function is compromised). Both partial and complete filling of the lungs have been considered, although only the former has undergone any significant tests in humans. Several animal tests have been performed and there have been some human partial liquid ventilation trials.[33] One effort, by Alliance Pharmaceuticals, reached clinical trials but was abandoned because of insufficient advantage compared to other therapies.[34]
Nanocrystals represent a possible method of delivering water- or fat-soluble drugs within a perfluorochemical fluid. The use of these particles is being developed to help treat babies with damaged lungs.[35]
Perfluorocarbons are banned from sports, where they may be used to increase oxygen use for endurance athletes. One cyclist, Mauro Gianetti, was investigated after a near-fatality where PFC use was suspected.[36][37] Other posited applications include deep-sea diving and space travel, applications that both require total, not partial, liquid ventilation.[38][39] The 1989 film The Abyss depicted a fictional use of perfluorocarbon for human diving but also filmed a real rat surviving while cooled and immersed in perfluorocarbon.[40] (See also list of fictional treatments of perfluorocarbon breathing.)
Agrichemicals
An estimated 30% of agrichemical compounds contain fluorine.[41] Most of them are used as poisons, but a few stimulate growth instead.

Trifluralin was widely used in the 20th century, for example, in over half of U.S. cotton field acreage in 1998.[46] Because of its suspected carcinogenic properties some Northern European countries banned it in 1993.[47] As of 2015, the European Union has banned it, although Dow made a case to cancel the decision in 2011.[48]
Biochemistry
Biologically synthesized organofluorines are few in number, although some are widely produced.
Fluoride is considered a semi-essential element for humans: not necessary to sustain life, but contributing (within narrow limits of daily intake) to dental health and bone strength. Daily requirements for fluorine in humans vary with age and sex, ranging from 0.01 mg in infants below 6 months to 4 mg in adult males, with an upper tolerable limit of 0.7 mg in infants to 10 mg in adult males and females.
Hazards
| NFPA 704 fire diamond | |
|---|---|
The fire diamond hazard sign for elemental fluorine.[55] |

Fluorine gas
Elemental fluorine is highly toxic. Above a concentration of 25 ppm, it causes significant irritation while attacking the eyes, airways and lungs and affecting the liver and kidneys. At a concentration of 100 ppm, human eyes and noses are seriously damaged.
Hydrofluoric acid

Once in the blood, hydrogen fluoride reacts with calcium and magnesium, resulting in electrolyte imbalances, potentially including
Symptoms of exposure to hydrofluoric acid may not be immediately evident, with an eight-hour delay for 50% HF and up to 24 hours for lower concentrations. Hydrogen fluoride interferes with nerve function, meaning that burns may not initially be painful. If the burn has been initially noticed, then HF should be washed off with a forceful stream of water for ten to fifteen minutes to prevent its further penetration into the body. Clothing used by the person burned may also present a danger.[63] Hydrofluoric acid exposure is often treated with calcium gluconate, a source of Ca2+ that binds with the fluoride ions. Skin burns can be treated with a water wash and 2.5 percent calcium gluconate gel[64][65] or special rinsing solutions.[66] Because HF is absorbed, further medical treatment is necessary. Calcium gluconate may be injected or administered intravenously. Use of calcium chloride is contraindicated and may lead to severe complications. Sometimes surgical excision of tissue or amputation is required.[62][67]
Fluoride ion

Soluble fluorides are moderately toxic. For sodium fluoride, the lethal dose for adults is 5–10 g, which is equivalent to 32–64 mg of elemental fluoride per kilogram of body weight.[68] The dose that may lead to adverse health effects is about one fifth of the lethal dose.[69] Chronic excess fluoride consumption can lead to skeletal fluorosis, a disease of the bones that affects millions in Asia and Africa.[69][70]
The fluoride ion is readily absorbed by the stomach and intestines. Ingested fluoride forms hydrofluoric acid in the stomach. In this form, fluoride crosses cell membranes and then binds with calcium and interferes with various enzymes. Fluoride is excreted through urine. Fluoride exposure limits are based on urine testing, which is used to determine the human body's capacity for ridding itself of fluoride.[69][71]
Historically, most cases of fluoride poisoning have been caused by accidental ingestion of insecticides containing inorganic fluoride.[72] Most calls to poison control centers for possible fluoride poisoning come from the ingestion of fluoride-containing toothpaste.[69] Malfunction of water fluoridation equipment has occurred several times, including an Alaskan incident that sickened nearly 300 people and killed one.[73]
Biopersistence
Because of the strength of the carbon–fluorine bond, organofluorines endure in the environment. Perfluorinated compounds (PFCs) have attracted particular attention as persistent global contaminants. These compounds can enter the environment from their direct uses in waterproofing treatments and firefighting foams or indirectly from leaks from fluoropolymer production plants (where they are intermediates). Because of the acid group, PFCs are water-soluble in low concentrations.[74] While there are other PFAAs, the lion's share of environmental research has been done on the two most well-known: perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA). The U.S. Environmental Protection Agency classifies these materials as "emerging contaminants" based on the growing but still incomplete understanding of their environmental impact.[75][76][77]
Trace quantities of PFCs have been detected worldwide, in organisms from polar bears in the Arctic to the global human population. Both PFOS and PFOA have been detected in breast milk and the blood of newborns. A 2013 review showed widely varying amounts of PFOS and PFOA in different soils and groundwater, with no clear pattern of one chemical dominating. PFC concentrations were generally higher in areas with more human population or industrial activity, and areas with more PFOS generally also had more PFOA.[78] the two chemicals have been found at different concentrations in different populations; for example, one study showed more PFOS than PFOA in Germans, while another study showed the reverse for Americans. PFCs may be starting to decrease in the biosphere: one study indicated that PFOS levels in wildlife in Minnesota were decreasing, presumably because 3M discontinued its production.[75][76]

In the body, PFCs bind to proteins such as serum albumin. Their tissue distribution in humans is unknown, but studies in rats suggest it is present mostly in the liver, kidney, and blood. They are not metabolized by the body but are excreted by the kidneys. Dwell time in the body varies greatly by species. Rodents have half-lives of days, while in humans they remain for years. Many animals show sex differences in the ability to rid the body of PFAAs, but without a clear pattern. Gender differences of half lives vary by animal species.[75][76][79]
The potential health impact of PFCs is unclear. Unlike chlorinated hydrocarbons, PFCs are not
The biochemical causes of toxicity are also unclear and may differ by molecule, health effect, and even animal.
Less fluorinated chemicals (i.e. not perfluorinated compounds) can also be detected in the environment. Because biological systems do not metabolize fluorinated molecules easily, fluorinated pharmaceuticals like antibiotics and antidepressants can be found in treated city sewage and wastewater.[80] Fluorine-containing agrichemicals are measurable in farmland runoff and nearby rivers.[81]
See also
- Fluorine absorption dating (a relative method for archeological dating of bone or other organics)
- Fluorine deficiency
- Fluoride toxicity
References
- ISBN 978-3527306732
- ^ S2CID 13189520.
- PMID 11521913.
- PMID 8474047. Archived from the original(PDF) on 2009-03-04.
- ^ PMID 17916854. Archived from the original(PDF) on 2019-12-09. Retrieved 2013-05-12.
- ISBN 9789350252161.
- PMID 18067684.
- ^ ISBN 978-1-4051-3889-5.
- PMID 18584000.
- ^ Cracher, Connie Myers (2009). "Current concepts in preventive dentistry" (PDF). dentalcare.com. p. 12. Archived from the original (PDF) on 14 October 2013. Retrieved 20 January 2012.
- ISBN 978-0-19-960563-7.
- ISBN 9780841209244.
- ^ Swinson, Joel (2005). "Fluorine – A vital element in the medicine chest" (PDF). PharmaChem: 26–27. Archived from the original (PDF) on 8 February 2012. Retrieved 26 August 2010.
- ISBN 9783540326236.
- ISBN 9783764360597.
- ^ ISBN 9781118300459.
- ISBN 9780470281871.
- PMID 21426080. Archived from the original(PDF) on 2013-10-22.
- ISBN 978-1-4381-0192-7.
- PMID 17342653.
- PMID 11132989.
- PMID 198801.
- ISBN 9780123742124.
- ISBN 978-0-13-033451-0.
- PMID 8800468.
- S2CID 35741393.
- S2CID 31161098.
- PMID 19742251.
- ^ Schimmeyer, S. (2002). "The search for a blood substitute". Illumin. 5 (1). Archived from the original on 2 October 2011. Retrieved 2 December 2010.
- ^ Tasker, Fred (2008-03-19). "Miami Herald: Artificial blood goes from science fiction to science fact". Miami Herald (at noblood.org). Archived from the original on 24 July 2008. Alt URL
- ^ Davis, Nicole (2006). "Better than blood". Popular Science. Archived from the original on 2011-06-04. Retrieved 30 September 2012.
- S2CID 222167378.
- PMID 16254269.
- PMID 10587539.
- ^ Gains, Paul (October 18, 1998). "A New Threat in Blood Doping". New York Times.
- ^ "Dying to ride". 1999-04-21.
- ^ Kylstra, J. A. (1977). The feasibility of liquid breathing in man. Duke University. Archived from the original on 7 July 2008. Retrieved 5 May 2008.
{{cite book}}: CS1 maint: unfit URL (link) - ^ The Global Oneness Commitment. "Liquid breathing – Space travel". experiencefestival.com. Archived from the original on 17 April 2010. Retrieved 17 May 2008.
- ^ Aljean Harmetz (1989). "FILM; 'The Abyss': A foray into deep waters". The New York Times. Retrieved 2 October 2012.
- ^ "Fluorine's treasure trove". ICIS news. 2006-10-02. Retrieved 20 February 2011.
- ^ Eisler, Ronald (1995). Biological report 27: Sodium monofluoroacetate (1080) Hazards to fish, wildlife and invertebrates: A synoptic review (PDF) (Report). Patuxent Environmental Science Center (U.S. National Biological Service). Archived from the original (PDF) on 12 June 2010. Retrieved 5 June 2011.
- ^ S2CID 29189551.
- ^ "Class I ozone-depleting substances". Sodium fluoride – pesticidal uses. Scorecard. Archived from the original on 11 July 2011. Retrieved 20 February 2011.
- ISBN 9781859570333.
- ^ "Fact sheet: Trifluralin". Pesticides News. 52: 20–21. 2001.
- ^ European Commission (2007). Trifluralin (PDF) (Report).
- ^ Case T-475/07, Dow AgroSciences Ltd vs. European Commission (2011). The General Court of European Union (Third Camber).
- ISBN 978-3-540-42064-4.
- ^ PMID 12738270.
- PMID 11907567.
- ^ Olivares, M.; Uauy, R. (2004). Essential nutrients in drinking water: Tables 2,6,7,8. (Draft) (PDF) (Report). WHO. Archived from the original (PDF) on 19 October 2012. Retrieved 30 December 2008.
- ^ Food and Nutrition Board, Institute of Medicine, National Academies. Dietary Reference Intakes (DRIs): Recommended Dietary Allowances and Adequate Intakes, Elements. http://www.nationalacademies.org/hmd/~/media/Files/Activity%20Files/Nutrition/DRI-Tables/2_%20RDA%20and%20AI%20Values_Vitamin%20and%20Elements.pdf?la=en Archived 2018-11-13 at the Wayback Machine Accessed Jan 2 2019.
- PMID 19874950.
- ^ "FLUORINE | CAMEO Chemicals | NOAA".
- ^ NOAA 9F data sheet.
- ^ Keplinger & Suissa 1968.
- ^ "CDC - NIOSH Pocket Guide to Chemical Hazards - Fluorine". www.cdc.gov. Retrieved 2015-11-03.
- ^ Eaton, Charles. "Figure hfl". E-Hand.com: the electronic textbook of hand surgery. The Hand Center (former practice of Dr. Eaton). Retrieved 28 September 2013.
- ^ a b Blodgett, Suruda & Crouch 2001.
- ^ Hoffman et al. 2007, p. 1333.
- ^ a b HSM 2006.
- ^ Fischman 2001, pp. 458–459.
- ^ El Saadi et al. 1989.
- ^ Roblin et al. 2006.
- ^ Hultén et al. 2004.
- ^ Zorich 1991, pp. 182–3.
- ^ Liteplo et al. 2002, p. 100.
- ^ a b c d Shin & Silverberg 2013.
- ^ Reddy 2009.
- ^ Baez, Baez & Marthaler 2000.
- ^ Augenstein et al. 1991.
- ^ Gessner et al. 1994.
- PMID 11999053.
- ^ PMID 20423814.
- ^ PMID 18007977.
- ^ "Emerging Contaminants Fact Sheet- PFOS and PFOA" (PDF). 2013-04-23. Archived from the original (PDF) on October 29, 2013. Retrieved November 1, 2013.
- ^ P. Zareitalabad, J. Siemens, M. Hamer, W. Amelung Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in surface waters, sediments, soils and wastewater – A review on concentrations and distribution coefficients Archived 2016-03-04 at the Wayback Machine Chemosphere 91 (2013) 725–732. Review
- ^ PMID 17519394.
- ^ Lietz & Meyer 2006, pp. 7–8.
- ^ Ahrens 2011.
Sources
- Ahrens, Lutz; Shoeib, Mahiba; Harner, Tom; Lee, Sum Chi; Guo, Rui; Reiner, Eric J. (1 October 2011). "Wastewater Treatment Plant and Landfills as Sources of Polyfluoroalkyl Compounds to the Atmosphere". Environmental Science & Technology. 45 (19): 8098–8105. PMID 21466185.
- Augenstein, W. L.; et al. (1991). "Fluoride Ingestion in Children: A Review of 87 cases". Pediatrics. 88 (5): 907–912. S2CID 22106466.
- Baez, Ramon J.; Baez, Martha X.; Marthaler, Thomas M. (2000). "Urinary Fluoride Excretion by Children 4–6 Years Old in a South Texas Community". Revista Panamericana de Salud Pública. 7 (4): 242–248. PMID 10846927.
- Blodgett, D. W.; Suruda, A. J.; Crouch, B. I. (2001). "Fatal Unintentional Occupational Poisonings by Hydrofluoric Acid in the U.S" (PDF). American Journal of Industrial Medicine. 40 (2): 215–220. PMID 11494350. Archived from the original(PDF) on 17 July 2012.
- El Saadi, M. S.; Hall, A. H.; Hall, P. K.; Riggs, B. S.; Augenstein, W. L.; Rumack, B. H. (1989). "Hydrofluoric Acid Dermal Exposure". Veterinary and Human Toxicology. 31 (3): 243–247. PMID 2741315.
- Fischman, Michael L. (2001). "Semiconductor Manufacturing Hazards". In Sullivan, John B.; Krieger, Gary R. (eds.). Clinical Environmental Health and Toxic Exposures (2nd ed.). Philadelphia: Lippincott Williams & Wilkins. pp. 431–465. ISBN 978-0-683-08027-8.
- Gessner, B. D.; Beller, lM.; Middaugh, J. P.; Whitford, G. M. (1994). "Acute Fluoride Poisoning from a Public Water System". New England Journal of Medicine. 330 (2): 95–99. PMID 8259189.
- Hoffman, Robert; Nelson, Lewis; Howland, Mary; Lewin, Neal; Flomenbaum, Neal; Goldfrank, Lewis (2007). Goldfrank's Manual of Toxicologic Emergencies. New York: McGraw-Hill Professional. ISBN 978-0-07-144310-4.
- Honeywell (2006). Recommended medical treatment for hydrofluoric acid exposure (PDF). Morristown: Honeywell International. Archived from the original (PDF) on 8 October 2013. Retrieved 9 January 2014.
- Hultén, P.; Höjer, J.; Ludwigs, U.; Janson, A. (2004). "Hexafluorine vs. Standard Decontamination to Reduce Systemic Toxicity After Dermal Exposure to Hydrofluoric Acid". Clinical Toxicology. 42 (4): 355–361. S2CID 27090208.
- Keplinger, M. L.; Suissa, L. W. (January 1968). "Toxicity of Fluorine Short-Term Inhalation". American Industrial Hygiene Association Journal. 29 (1): 10–18. PMID 5667185.
- Lietz, A.C.; Meyer, Michael T. (2006). Evaluation of emerging contaminants of concern at the South District Wastewater Treatment Plant based on seasonal sampling events, Miami-Dade County, Florida, 2004 (PDF) (Report). U.S. Geological Survey.
- Liteplo, R.; Gomes, R.; Howe, P.; Malcolm, H. (2002). Environmental Health Criteria 227 (Fluoride). Geneva: United Nations Environment Programme; International Labour Organization; World Health Organization. ISBN 92-4-157227-2. Retrieved 14 October 2013.
- Reddy, D. (2009). "Neurology of Endemic Skeletal Fluorosis". Neurology India. 57 (1): 7–12. PMID 19305069.
- Roblin, I.; Urban, M.; Flicoteau, D.; Martin, C.; Pradeau, D. (2006). "Topical Treatment of Experimental Hydrofluoric Acid Skin Burns by 2.5% Calcium Gluconate". Journal of Burn Care & Research. 27 (6): 889–894. S2CID 3691306.
- Shin, Richard D.; Silverberg, Mark A. (2013). "Fluoride Toxicity". Medscape. Retrieved 15 October 2013.
- Zorich, Robert (1991). Handbook of Quality Integrated Circuit Manufacturing. San Diego: Academic Press. ISBN 978-0-323-14055-3.
External links
- A strong acid it is not, but deadly it is... (Podcast on fluorine, note the first few minutes discussion of a fatal HF burn.)

