Chlorothalonil
| |
| Names | |
|---|---|
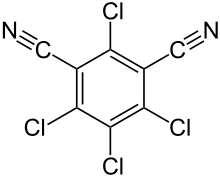
| Preferred IUPAC name
2,4,5,6-Tetrachlorobenzene-1,3-dicarbonitrile | |
| Other names
2,4,5,6-Tetrachloroisophthalonitrile
Bravo Daconil Tetrachloroisophthalonitrile Celeste Bronco Agronil Aminil | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.015.990 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3276, 2588 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8Cl4N2 | |
| Molar mass | 265.90 g·mol−1 |
| Appearance | white crystalline solid |
| Density | 1.8 g cm−3, solid |
| Melting point | 250 °C (482 °F; 523 K) |
| Boiling point | 350 °C (662 °F; 623 K) (760 mmHg) |
| 10 mg/100 mL[1] | |
| log P | 2.88–3.86 |
| Hazards | |
| GHS labelling: | |
    
| |
| Danger | |
| H317, H318, H330, H335, H351, H410 | |
| P201, P202, P260, P261, P271, P272, P273, P280, P281, P284, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P312, P320, P321, P333+P313, P363, P391, P403+P233, P405, P501 | |
| Related compounds | |
Related
organochlorides |
benzonitrile; hexachlorobenzene, dichlorobenzene, chlorobenzene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chlorothalonil (2,4,5,6-tetrachloroisophthalonitrile) is an
Uses
In the US, chlorothalonil is used predominantly on peanuts (about 34% of usage), potatoes (about 12%), and tomatoes (about 7%), although the EPA recognizes its use on many other crops.[2] It is also used on golf courses and lawns (about 10%) and as a preservative additive in some paints (about 13%), resins, emulsions, and coatings.[2]
Chlorothalonil is commercially available in many different formulations and delivery methods. It is applied as a dust, dry or water-soluble grains, a wettable powder, a liquid spray, a fog, and a dip. It may be applied by hand, by ground sprayer, or by aircraft.[2]
Mechanism of action
Chlorothalonil
Toxicity
Acute
According to the EPS, chlorothalonil is a toxicity category I eye irritant, producing severe eye irritation. It is in toxicity category II, "moderately toxic", if inhaled (inhaled LD50 0.094 mg/L in rats.) For skin contact and ingestion, chlorothalonil is rated toxicity category IV, "practically nontoxic", meaning the oral and dermal LD50 is greater than 10,000 mg/kg.[2]
Chronic
Long-term exposure to chlorothalonil resulted in kidney damage and tumors in animal tests.[2]
Carcinogenic
Chlorothalonil is a Group 2B "possible human
Environmental
Chlorothalonil was found to be an important factor in the decline of the honey bee population, by making the bees more vulnerable to the gut parasite Nosema ceranae.[6]
Chlorothalonil is highly toxic to fish and aquatic invertebrates, but not toxic to birds.[7]
At a concentration of 164 μg/L, chlorothalonil was found to kill a species of frog within a 24-hour exposure.[8]
Contaminants
Common chlorothalonil synthesis procedures frequently result in contamination of it with small amounts of hexachlorobenzene (HCB), which is toxic. US regulations limit HCB in commercial production to 0.05% of chlorothalonil. According to the EPA report, "post-application exposure to HCB from chlorothalonil is not expected to be a concern based on the low level of HCB in chlorothalonil. 2,3,7,8-Tetrachlorodibenzodioxin being one of the most potent carcinogens known is also a known contaminant".[2]
Environmental contamination
Chlorothalonil has been detected in ambient air in
The main breakdown product of chlorothalonil is hydroxy-2,5,6-trichloro-1,3-dicyanobenzene (SDS-3701). It has been shown to be 30 times more acutely toxic than chlorothalonil and more persistent in the environment.[12][13] Laboratory experiments have shown it can thin the eggshells of birds, but no evidence supports this happening in the environment.[2]
In 2019, a review of the evidence found that "a high risk to amphibians and fish was identified for all representative uses", and that chlorothalonil breakdown products may cause DNA damage.[14][15] Around the same time, research indicated chlorothalonil and other fungicides to be the strongest factor in bumblebee population decline.[14]
- Important metabolites of chlorothalonil
-
2,4,5-trichloro-6-hydroxyisophthalonitrile (SDS-3701)
-
Chlorothalonil sulfonic acid (R417888)
-
R471811
Gag order
Syngenta sued to stop communication by Swiss health authorities with the Swiss public regarding the "relevance" of specific metabolites of chlorothalonil that Swiss authorities detected in high concentrations in the groundwater from which hundreds of thousands of Swiss people obtain drinking water. The court banned Swiss health authorities from communicating with the public about the dangers posed by some of the metabolites.[16]
Bans
In March 2019, as a result of the previously mentioned research, the European Union banned the use of chlorothalonil[14] dated to take effect May 20, 2020.[17] Switzerland followed in December 2019.[18]
Removal
The removal of chlorothalonil and its metabolites is usually achieved by very expensive, technically complex and unecological methods such as reverse osmosis, nano- or microfiltration. In addition, dilution is achieved by mixing with uncontaminated water. However, this only reduces the concentration, the substances still remain in the water. In technologies based on membrane technology, the membranes must be cleaned regularly with chemicals, which is expensive and not ecological.
Recent research results from a Swiss company show very good results for the removal of chlorothalonil and its metabolites. In contrast to the conclusion described in the Eawag fact-sheet, it was also possible to remove the metabolites R471811 and chlorothalonil sulfonic acid from water using a filter material based on activated carbon. The first measurements did not even reach the detection limit. Thus, it was shown that it is possible to completely remove the pesticide residues.[19]
Production
Chlorothalonil can be produced by the direct chlorination of
References
- ^ "Chlorothalonil". Pubchem.
- ^ a b c d e f g h i j k l m Reregistration Eligibility Decision for chlorothalonil, US EPA, 1999.
- ^ PESTICIDE USE IN U.S. CROP PRODUCTION: 1997 Archived 10 December 2006 at the Wayback Machine National Center for Food and Agricultural Policy, 1997.
- PMID 5474238.
- ^ Jeffery S. Pettis, Elinor M. Lichtenberg, Michael Andree, Jennie Stitzinger, Robyn Rose, Dennis vanEngelsdorp "Crop Pollination Exposes Honey Bees to Pesticides Which Alters Their Susceptibility to the Gut Pathogen Nosema ceranae" PLOS One, 24 July 2013, Online: 9 April 2014. [1]
- ^ a b Environmental Health Criteria 183, World Health Organization, 1996.
- ^ Pesticides and Air Pollution in Minnesota: The Frequency of Detection of Chlorothalonil, a Fungicide Used on Potatoes.[permanent dead link] Pesticide Action Network North America, Oct 2007.
- PMID 16358323.
- ^ "Island Fish Kills from 1962 to 2016" (PDF). Government of PEI. Retrieved 12 December 2016.
- PMID 32625673.
- ^ Cox, Caroline (1997), "Fungicide Factsheet: Chlorothalonil", Journal of Pesticide Reform, 17 (4): 14–20, archived from the original on 10 November 2010
- ^ a b c Carrington, Damian (29 March 2019). "EU bans UK's most-used pesticide over health and environment fears". The Guardian. Retrieved 29 March 2019.
- ^ "European Commission votes to ban fungicide Chlorothalonil".
- ^ Le Monde, 27 Sept. 2022 "By-Products of Pesticide Banned in 2019 Found in Swiss Drinking Water, Syngenta, The Producer of The Fungicide Chlorothalonil, Has Sued Swiss Health Authorities for Making Public The Danger Posed by Certain Derivatives of The Pesticide,"
- ^ National Farmers' Union of England and Wales. "Chlorothalonil use-by date approaching". NFU Online. Retrieved 16 March 2021.
- swissinfo. Retrieved 12 December 2019.
- ^ "BioGreen P". United Waters Int. Retrieved 23 March 2021.
- ISBN 978-3527306732.
External links
- Extension Toxicology Network: Chlorothalonil Pesticide Information Profile
- Chlorothalonil in the Pesticide Properties DataBase (PPDB)
- "2017 Pesticide Use Maps - Low". Water Resources. United States Geological Survey. Retrieved 16 March 2021.
- "2017 Pesticide Use Maps - High". Water Resources. United States Geological Survey. Retrieved 16 March 2021.



