Corannulene

| |

| |
| Names | |
|---|---|
| IUPAC name
Dibenzo[ghi,mno]fluoranthene[1]
| |
| Other names
[5]circulene
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H10 | |
| Molar mass | 250.29 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Corannulene is a
Synthesis
Several synthetic routes exist to corannulene.
The bromine substituents are removed with an excess of n-butyllithium.
A kilogram scale synthesis of corannulene has been achieved.[8]
Much effort is directed at functionalization of the corannulene ring with novel functional groups such as ethynyl groups,[3][9][10] ether groups,[11] thioether groups,[12] platinum functional groups,[13] aryl groups,[14] phenalenyl fused [15] and indeno extensions.[16] and ferrocene groups.[17]
Aromaticity
The observed
However, later theoretical calculations have disputed the validity of this approximation.[18][19]
Reactions
Reduction
Corannulene can be reduced up to a tetraanion in a series of
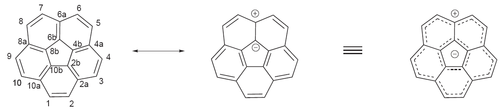
In one cyclopenta[bc]corannulene a concave - concave aggregate is observed by
Metals tend to bind to the convex face of the annulene. Concave binding has been reported for a cesium / crown ether system [24]
Oxidation
UV 193-nm photoionization effectively removes a π-electron from the twofold degenerate E1-HOMO located in the aromatic network of electrons yielding a corannulene radical cation.[25] Owing to the degeneracy in the HOMO orbital, the corannulene radical cation is unstable in its original C5v molecular arrangement, and therefore, subject to Jahn-Teller (JT) vibronic distortion.
Using electrospray ionization, a protonated corannulene cation has been produced in which the protonation site was observed to be on a peripheral sp2-carbon atom.[25]
Reaction with electrophiles
Corannulene can react with
Bicorannulenyl
Bicorannulenyl is the product of dehydrogenative coupling of corannulene. With the formula C20H9-C20H9, it consists of two corannulene units connected through a single C-C bond. The molecule's stereochemistry consists of two chiral elements: the asymmetry of a singly substituted corannulenyl, and the helical twist about the central bond. In the neutral state, bicorannulenyl exists as 12 conformers, which interconvert through multiple bowl-inversions and bond-rotations.[27] When bicorannulenyl is reduced to a dianion with potassium metal, the central bond assumes significant double-bond character. This change is attributed to the orbital structure, which has a LUMO orbital localized on the central bond.[28] When bicorannulenyl is reduced to an octaanion with lithium metal, it self-assembles into supramolecular oligomers.[29] This motif illustrates "charged polyarene stacking".
Research
Alkyl-substituted corannulenes form a thermotropic hexagonal columnar liquid crystalline mesophase.[33] Corannulene has also been used as the core group in a dendrimer.[14] Like other PAHs, corannulene ligates metals.[34][35][36][37][38][39][40] Corannulenes with ethynyl groups are investigated for their potential use as blue emitters.[10] The structure was analyzed by infrared spectroscopy, Raman spectroscopy, and X-ray photoelectron spectroscopy.[41]
See also
References
- ^ Fluoranthene is so named for its fluorescent property. It is not a fluorine compound.
- S2CID 37901191.
- ^ .
- ^ .
- S2CID 94872875.
- .
- .
- .
- PMID 18642812.
- ^ PMID 17637965.
- PMID 19905024.
- PMID 20024131.
- .
- ^ PMID 19193048.
- PMID 18937470.
- PMID 19722628.
- PMID 22595996.
- .
- PMID 18693706.
- S2CID 4979579.
- PMID 15984885.
- S2CID 1125747.
- PMID 16388648.
- PMID 21748832.
- ^ PMID 21303123.
- PMID 21404379.
- PMID 18505292.
- PMID 20814993.
- PMID 21057679.
- S2CID 25154754.
- S2CID 18247078.
- .
- PMID 19128171.
- .
- PMID 16910635.
- PMID 18214869.
- .
- .
- PMID 16910635.
- PMID 21246679.
- S2CID 224784081.


![Cyclopenta[bc]corannulene](http://upload.wikimedia.org/wikipedia/commons/thumb/2/21/Cyclopenta-bc-corannulene.png/100px-Cyclopenta-bc-corannulene.png)