Islet cell transplantation
| Islet cell transplantation | |
|---|---|
 Microscopic image of an islet of Langerhans (lighter area) surrounded by exocrine pancreas tissue (darker staining). | |
| MeSH | D016381 |
Islet transplantation is the
Islets are usually infused into the person's
In the period from 1999 to 2004, 471 people with type 1 diabetes received islet transplants at 43 institutions worldwide.[5]
Donislecel (Lantidra) allogeneic (donor) pancreatic islet cellular therapy was approved for medical use in the United States in June 2023.[6]
History
The concept of islet transplantation is not new.
Goals
The goal of islet transplantation is to infuse enough islets to control the
Newer studies have focused their attention towards reducing severe hypoglycemic events, a life-threatening state in type 1 diabetes, rather than focus on removing the need for insulin injections entirely.[8][9]
Procedure

Researchers use a mixture of highly purified enzymes (
During the transplant, a
It takes time for the islets to attach to new blood vessels and begin releasing insulin. The doctor will order many tests to check blood glucose levels after the transplant, and insulin may be needed until control is achieved.
-
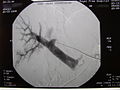
Radiographic image of the portal vein and its branches in the transplant recipient before infusion of isolated islets.
-
Post-transplant radiographic image of the recipient's portal tree.
Immunosuppression
The Edmonton protocol uses a combination of immunosuppressive drugs, including daclizumab (Zenapax), sirolimus (Rapamune) and tacrolimus (Prograf). Daclizumab is given intravenously right after the transplant and then discontinued. Sirolimus and tacrolimus, the two main drugs that keep the immune system from destroying the transplanted islets, must be taken for life.[citation needed]
Limitations
While significant progress has been made in the islet transplantation field,
Another limitation to the islet transplantation process is the inflammatory response of the liver. Dr. Melena Bellin is an associate professor of pediatric endocrinology and surgery and director of research for the islet autotransplant program at the University of Minnesota Medical Center and Masonic Children's Hospital. Her research centers on making islet transplants safer and more effective for type one diabetics. The process of infusing islet cells into the liver can trigger an inflammatory response in the body. This reaction leads to a large amount of the newly transplanted islets being destroyed. Losing islet cells decreases the probability of successful insulin production and increases the likelihood of type one diabetes developing again in the patient. Dr. Bellin is currently testing two anti-inflammatory drugs that are already on the market to see if they may be useful in preventing inflammation that destroys islet cells.[13]
References
- PMID 26644812.
- PMID 12928769.
- PMID 15848704.
- S2CID 28784928.
- PMID 15912095.
- ^ "FDA Approves First Cellular Therapy to Treat Patients with Type 1 Diabetes". U.S. Food and Drug Administration (FDA) (Press release). 28 June 2023. Retrieved 28 June 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- PMID 30505878.
- PMID 27208344.
- S2CID 22996728.
- S2CID 1307522.
- S2CID 12270800.
- ^ Brody, Barbara. "Making Islet Cell Transplants Safer". Diabetes Forecast.