Sirolimus
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Rapamune, Fyarro, Hyftor |
| Other names | Rapamycin, ABI-009 |
| License data | |
| Pregnancy category |
|
topical | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 14% (oral solution), lower with high-fat meals; 18% (tablet), higher with high-fat meals[7] |
| Protein binding | 92% |
| Metabolism | Liver |
| Elimination half-life | 57–63 hours[8] |
| Excretion | Mostly fecal |
| Identifiers | |
| |
JSmol) | |
| Solubility in water | 0.0026 [9] |
| |
| |
| (verify) | |
Sirolimus, also known as rapamycin and sold under the brand name Rapamune among others, is a
This compound also has a use in cardiovascular drug-eluting stent technologies to inhibit restenosis.
It is produced by the
Medical uses
Sirolimus is
Sirolimus (Fyarro), as protein-bound particles, is indicated for the treatment of adults with locally advanced unresectable or metastatic
In the EU, sirolimus, as Rapamune, is indicated for the prophylaxis of organ rejection in adults at low to moderate immunological risk receiving a renal transplant[5] and, as Hyftor, is indicated for the treatment of facial angiofibroma associated with tuberous sclerosis complex.[6]
Prevention of transplant rejection
The chief advantage sirolimus has over
Sirolimus can also be used alone, or in conjunction with a
Lymphangioleiomyomatosis
In May 2015, the FDA approved sirolimus to treat
The safety and efficacy of sirolimus treatment of LAM were investigated in
While sirolimus was considered for treatment of LAM, it received orphan drug designation status because LAM is a rare condition.[22]
The safety of LAM treatment by sirolimus in people younger than 18 years old has not been tested.[2]
Coronary stent coating
The antiproliferative effect of sirolimus has also been used in conjunction with coronary stents to prevent restenosis in coronary arteries following balloon angioplasty. The sirolimus is formulated in a polymer coating that affords controlled release through the healing period following coronary intervention. Several large clinical studies have demonstrated lower restenosis rates in patients treated with sirolimus-eluting stents when compared to bare-metal stents, resulting in fewer repeat procedures. A sirolimus-eluting coronary stent was marketed by Cordis, a division of Johnson & Johnson, under the tradename Cypher.[11] However, this kind of stent may also increase the risk of vascular thrombosis.[23]
Vascular malformations
Sirolimus is used to treat vascular malformations. Treatment with sirolimus can decrease pain and the fullness of vascular malformations, improve coagulation levels, and slow the growth of abnormal lymphatic vessels.[24] Sirolimus is a relatively new medical therapy for the treatment of vascular malformations[25] in recent years, sirolimus has emerged as a new medical treatment option for both vascular tumors and vascular malformations, as a mammalian target of rapamycin (mTOR), capable of integrating signals from the PI3K/AKT pathway to coordinate proper cell growth and proliferation. Hence, sirolimus is ideal for "proliferative" vascular tumors through the control of tissue overgrowth disorders caused by inappropriate activation of the PI3K/AKT/mTOR pathway as an antiproliferative agent.[26][27]
Angiofibromas
Sirolimus has been used as a topical treatment of angiofibromas with tuberous sclerosis complex (TSC). Facial angiofibromas occur in 80% of patients with TSC, and the condition is very disfiguring. A retrospective review of English-language medical publications reporting on topical sirolimus treatment of facial angiofibromas found sixteen separate studies with positive patient outcomes after using the drug. The reports involved a total of 84 patients, and improvement was observed in 94% of subjects, especially if treatment began during the early stages of the disease. Sirolimus treatment was applied in several different formulations (ointment, gel, solution, and cream), ranging from 0.003 to 1% concentrations. Reported adverse effects included one case of perioral dermatitis, one case of cephalea, and four cases of irritation.[28]
In April 2022, sirolimus was approved by the FDA for treating angiofibromas.[29]
Adverse effects
The most common adverse reactions (≥30% occurrence, leading to a 5% treatment discontinuation rate) observed with sirolimus in clinical studies of organ rejection prophylaxis in individuals with kidney transplants include: peripheral edema, hypercholesterolemia, abdominal pain, headache, nausea, diarrhea, pain, constipation, hypertriglyceridemia, hypertension, increased creatinine, fever, urinary tract infection, anemia, arthralgia, and thrombocytopenia.[2]
The most common adverse reactions (≥20% occurrence, leading to an 11% treatment discontinuation rate) observed with sirolimus in clinical studies for the treatment of lymphangioleiomyomatosis are: peripheral edema, hypercholesterolemia, abdominal pain, headache, nausea, diarrhea, chest pain,
The following adverse effects occurred in 3–20% of individuals taking sirolimus for organ rejection prophylaxis following a kidney transplant:[2]
| System | Adverse effects |
|---|---|
| Body as a whole | herpes simplex infection
|
Cardiovascular |
deep venous thrombosis), rapid heart rate
|
Digestive |
Stomatitis |
lymphatic |
hemolytic uremic syndrome (TTP/HUS), leukopenia
|
| Metabolic | Abnormal healing, increased lactic dehydrogenase (LDH), hypokalemia, diabetes
|
Musculoskeletal |
Bone necrosis |
Respiratory |
epistaxis
|
| Skin | basal cell carcinoma
|
Urogenital |
menorrhagia )
|
Diabetes-like symptoms
While sirolimus inhibition of mTORC1 appears to mediate the drug's benefits, it also inhibits mTORC2, which results in diabetes-like symptoms.[30] This includes decreased glucose tolerance and insensitivity to insulin.[30] Sirolimus treatment may additionally increase the risk of type 2 diabetes.[31] In mouse studies, these symptoms can be avoided through the use of alternate dosing regimens or analogs such as everolimus or temsirolimus.[32]
Lung toxicity
Lung toxicity is a serious complication associated with sirolimus therapy,
Lowered effectiveness of immune system
There have been warnings about the use of sirolimus in transplants, where it may increase mortality due to an increased risk of infections.[2]
Cancer risk
Sirolimus may increase an individual's risk for contracting
Impaired wound healing
Individuals taking sirolimus are at increased risk of experiencing impaired or delayed wound healing, particularly if they have a body mass index in excess of 30 kg/m2 (classified as obese).[2]
Interactions
Sirolimus is metabolized by the CYP3A4 enzyme and is a substrate of the P-glycoprotein (P-gp) efflux pump; hence, inhibitors of either protein may increase sirolimus concentrations in blood plasma, whereas inducers of CYP3A4 and P-gp may decrease sirolimus concentrations in blood plasma.[2]
Pharmacology
Pharmacodynamics
Unlike the similarly named
The mode of action of sirolimus is to bind the
mTOR has also been called FRAP (FKBP-rapamycin-associated protein), RAFT (rapamycin and FKBP target), RAPT1, or SEP. The earlier names FRAP and RAFT were coined to reflect the fact that sirolimus must bind FKBP12 first, and only the FKBP12-sirolimus complex can bind mTOR. However, mTOR is now the widely accepted name, since Tor was first discovered via genetic and molecular studies of sirolimus-resistant mutants of Saccharomyces cerevisiae that identified FKBP12, Tor1, and Tor2 as the targets of sirolimus and provided robust support that the FKBP12-sirolimus complex binds to and inhibits Tor1 and Tor2.[46][12]
Pharmacokinetics
Sirolimus is metabolized by the
The absorption of sirolimus into the blood stream from the intestine varies widely between patients, with some patients having up to eight times more exposure than others for the same dose. Drug levels are, therefore, taken to make sure patients get the right dose for their condition.
Chemistry
This section needs expansion with: content on this topic from [8]. You can help by adding to it. (August 2016) |
Sirolimus is a
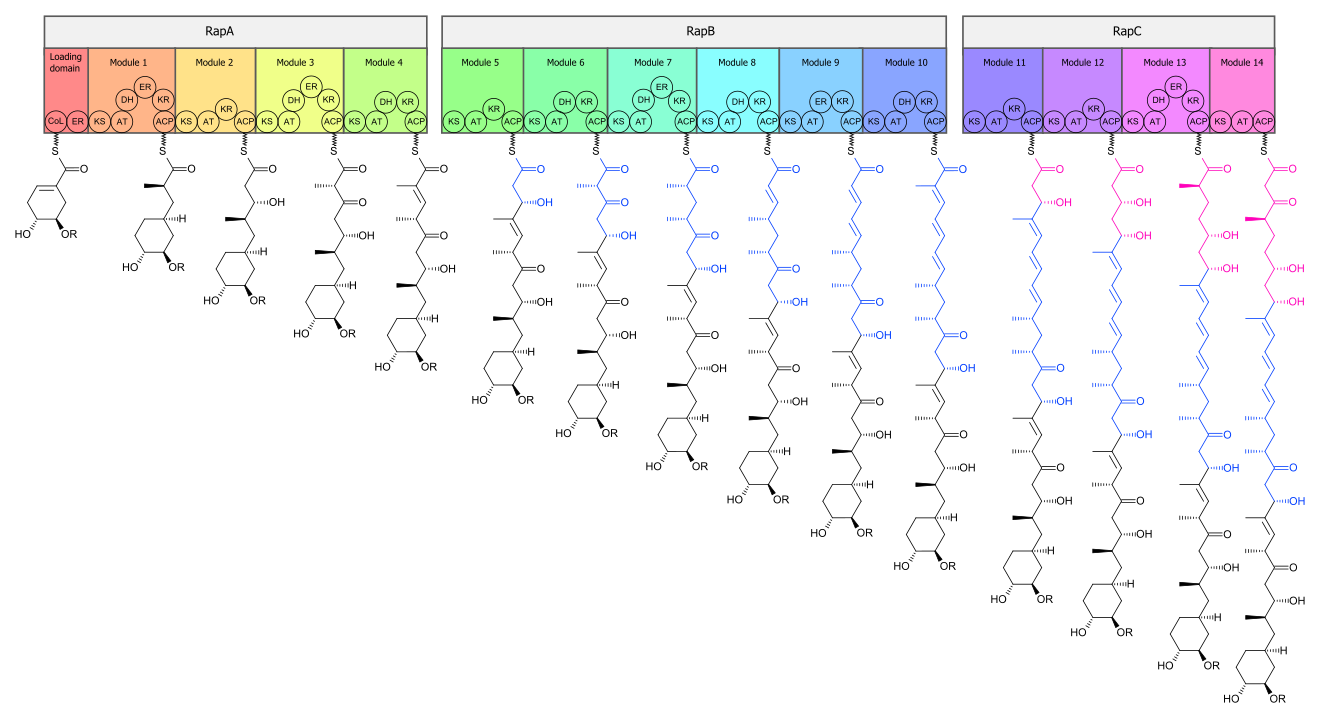
Biosynthesis
The

The core
The biosynthetic
Research

Cancer
The antiproliferative effects of sirolimus may have a role in treating cancer. When dosed appropriately, sirolimus can enhance the immune response to tumor targeting[54] or otherwise promote tumor regression in clinical trials.[55] Sirolimus seems to lower the cancer risk in some transplant patients.[56]
Sirolimus was shown to inhibit the progression of dermal
A
Tuberous sclerosis complex
Sirolimus also shows promise in treating
Effects on longevity
Sirolimus has potential for widespread use as a longevity-promoting drug, with evidence pointing to its ability to prevent age-associated decline of cognitive and physical health. In 2014, researchers at Novartis showed a related compound, everolimus, increased elderly patients' immune response on an intermittent dose.[69] This led to many in the anti-aging community self-experimenting with the compound.[70] However, because of the different biochemical properties of Sirolimus the dosing is potentially very different than everolimus. Ultimately, due to known side effects of sirolimus, as well as inadequate evidence for optimal dosing, more research is required before sirolimus could be widely prescribed for this purpose.[66][71]
Sirolimus has complex effects on the immune system—while IL-12 goes up and IL-10 decreases, which suggests an immunostimulatory response, TNF and IL-6 are decreased, which suggests an immunosuppressive response. The duration of the inhibition and the exact extent to which mTORC1 and mTORC2 are inhibited play a role, but are not yet well understood.[72]
Topical administration
When applied as a topical preparation, researchers showed that rapamycin can regenerate collagen and reverse clinical signs of aging in elderly patients.[73] The concentrations are far lower than those used to treat angiofibromas.
SARS-CoV-2
Rapamycin has been proposed as a treatment for
Atherosclerosis
Rapamycin can accelerate degradation of oxidized LDL cholesterol in endothelial cells, thereby lowering the risk of atherosclerosis.[75] Oxidized LDL cholesterol is a major contributor to atherosclerosis.[76]
Lupus
As of 2016, studies in cells, animals, and humans have suggested that mTOR activation as process underlying
Lymphatic malformation
Lymphatic malformation, or cystic hygroma, is an abnormal growth of lymphatic vessels that usually affects children around the head and neck. Treatment often consists of to removal of the tissue, but the rate of recurrence is high. Sirolimus has shown evidence of being helpful in alleviating symptoms and reducing the size of the malformation.[78]
Graft-versus-host disease
Due to its immunosuppressant activity, Rapamycin has been assessed as prophylaxis or treatment agent of Graft-versus-host disease (GVHD), a complication of hematopoietic stem cell transplantation. While contrasted results were obtained in clinical trials,[79] pre-clinical studies have shown that Rapamycin can mitigate GVHD by increasing the proliferation of regulatory T cells, inhibiting cytotoxic T cells and lowering the differentiation of effector T cells.[80][81]
Applications in biology research
Rapamycin is used in biology research as an agent for chemically induced dimerization.[82] In this application, rapamycin is added to cells expressing two fusion constructs, one of which contains the rapamycin-binding FRB domain from mTOR and the other of which contains an FKBP domain. Each fusion protein also contains additional domains that are brought into proximity when rapamycin induces binding of FRB and FKBP. In this way, rapamycin can be used to control and study protein localization and interactions.
Veterinary uses
A number of
References
- FDA. Retrieved 22 October 2023.
- ^ a b c d e f g h i j k l m n "Rapamune- sirolimus solution Rapamune- sirolimus tablet, sugar coated". DailyMed. Archived from the original on 27 November 2021. Retrieved 26 November 2021.
- ^ a b c d "Fyarro- sirolimus injection, powder, lyophilized, for suspension". DailyMed. Archived from the original on 19 December 2021. Retrieved 19 December 2021.
- ^ "Hyftor- sirolimus gel". DailyMed. 28 January 2021. Archived from the original on 24 March 2022. Retrieved 23 March 2022.
- ^ a b "Rapamune EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 13 August 2021. Retrieved 26 November 2021.
- ^ a b c "Hyftor EPAR". European Medicines Agency (EMA). 9 June 2023. Retrieved 12 June 2023. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Buck ML (2006). "Immunosuppression With Sirolimus After Solid Organ Transplantation in Children". Pediatric Pharmacotherapy. 12 (2). Archived from the original on 18 April 2020. Retrieved 4 April 2022.
- ^ a b c d "Rapamycin". PubChem Compound. National Center for Biotechnology Information. Archived from the original on 16 August 2016. Retrieved 1 August 2016.
- PMID 11165091.
- ^ PMID 1102508.
- ^ Cypher Stent. Archived from the originalon 27 April 2003. Retrieved 1 April 2008.
- ^ PMID 20130772.
- ^ Qari S, Walters P, Lechtenberg S (21 May 2021). "The Dirty Drug and the Ice Cream Tub". Radiolab. Archived from the original on 25 July 2021. Retrieved 25 July 2021.
- PMID 23369283.
- PMID 15896681.
- ^ "Drug Approval Package: Rapamune (Sirolimus) NDA# 021083". U.S. Food and Drug Administration (FDA). Archived from the original on 1 February 2022. Retrieved 1 February 2022.
- ^ "Aadi Bioscience Announces FDA Approval of its First Product Fyarro for Patients with Locally Advanced Unresectable or Metastatic Malignant Perivascular Epithelioid Cell Tumor (PEComa)". Aadi Bioscience, Inc. (Press release). 23 November 2021. Archived from the original on 27 November 2021. Retrieved 26 November 2021.
- ISBN 978-1-118-35446-9. Archivedfrom the original on 19 August 2020. Retrieved 1 August 2016.
- ISBN 978-1-60650-250-1. Archivedfrom the original on 30 September 2020. Retrieved 1 August 2016.
- ^ "Sirolimus (marketed as Rapamune) Safety". U.S. Food and Drug Administration (FDA). 11 June 2009. Archived from the original on 16 September 2016. Retrieved 1 August 2016.
- PMID 31461151.
- ^ a b c Pahon E (28 May 2015). "FDA approves Rapamune to treat LAM, a very rare lung disease". U.S. Food and Drug Administration (FDA). Archived from the original on 14 August 2016. Retrieved 1 August 2016.
- PMID 17093244.
- ^ "Venous Malformation: Treatments". Boston Children's Hospital. Archived from the original on 17 January 2021. Retrieved 22 April 2020.
- PMID 32200879.
- PMID 31864650.
- PMID 27723921.
- S2CID 9967001.
- ^ "FDA Approves Nobelpharma's Topical Treatment for Facial Angiofibroma". FDAnews. 7 April 2022. Archived from the original on 1 June 2022. Retrieved 24 May 2022.
- ^ PMID 22461615.
- PMID 18385422.
- PMID 26463117.
- ^ S2CID 24408680.
- S2CID 12116798.
- from the original on 7 August 2020. Retrieved 24 September 2019.
- S2CID 24496443.
- (PDF) from the original on 5 June 2022. Retrieved 4 April 2022.
- PMID 17346635.
- PMID 16962483.
- PMID 12581772.
- S2CID 39914334.
- PMID 22240222.
- PMID 21444868.
- PMID 20970608.
- PMID 22830463.
- S2CID 9937225.
- S2CID 34950948.
- ^ PMID 7644502.
- PMID 15127450.
- PMID 16990929.
- PMID 19387497.
- PMID 16536560.
- PMID 8635756.
- PMID 22379028.
- S2CID 19250234.
- PMID 16039868.
- S2CID 198175394.
- ^ Bankhead C (17 February 2013). "Fatal AEs Higher with mTOR Drugs in Cancer". Med Page Today. Archived from the original on 28 February 2021. Retrieved 19 February 2013.
- PMID 16103051.
- PMID 15365568.
- S2CID 4426215.
- ^ "Combination therapy drives cancer into remission". Cold Spring Harbor Laboratory. 17 March 2004. Archived from the original on 1 June 2022. Retrieved 23 March 2022.
- S2CID 45906785.
- PMID 30760308.
- ^ Lawton G. "What is rapamycin?". New Scientist. Archived from the original on 25 July 2021. Retrieved 25 July 2021.
- ^ PMID 27208895.
A diverse and severe set of negative side effects likely preclude the wide-scale use of rapamycin and its analogs as a prolongevity agent.
- PMID 33145977.
- ^ "Late-Life Rapamycin Regimens Extend Mouse Lifespan in a Sex-Specific Manner". Nicotinamide Mononucleotide (NMN). 11 November 2020. Archived from the original on 28 February 2021. Retrieved 23 March 2022.
- S2CID 206685475.
- ^ Janin A (May 2023). "Can a Kidney Transplant Drug Keep You From Aging?". Wall Street Journal. Archived from the original on 8 May 2023. Retrieved 9 May 2023.
- PMID 23325216.
- PMID 26403194.
- PMID 31761958.
- ^ PMID 33031791.
- PMID 30666207.
- PMID 15383655.
- ^ PMID 27812954.
- PMID 35526176.
- PMID 27245261.
- S2CID 31313976.
- S2CID 231766741.
- S2CID 30469863.
- ^
- "Study to assess healthy aging in dogs: the Dog Aging Project and Test of Rapamycin in Aging Dogs (TRIAD study)". University of Georgia College of Veterinary Medicine. 29 July 2022. Archived from the original on 23 February 2023. Retrieved 23 February 2023.
- "Dog Aging Project TRIAD Study". Washington State University Veterinary Teaching Hospital. 28 March 2022. Archived from the original on 23 February 2023. Retrieved 23 February 2023.
- "Dog aging project - TRIAD (Test of Rapamycin in Aging Dogs)". Iowa State University College of Veterinary Medicine. Archived from the original on 23 February 2023. Retrieved 23 February 2023.
Further reading
- Benjamin D, Colombi M, Moroni C, Hall MN (October 2011). "Rapamycin passes the torch: a new generation of mTOR inhibitors". Nature Reviews. Drug Discovery. 10 (11): 868–880. S2CID 1227277.
- Freixo C, Ferreira V, Martins J, Almeida R, Caldeira D, Rosa M, et al. (January 2020). "Efficacy and safety of sirolimus in the treatment of vascular anomalies: A systematic review". Journal of Vascular Surgery. 71 (1): 318–327. S2CID 207831199.
- Geeurickx M, Labarque V (September 2021). "A narrative review of the role of sirolimus in the treatment of congenital vascular malformations". Journal of Vascular Surgery. Venous and Lymphatic Disorders. 9 (5): 1321–1333. PMID 33737259.
External links
- Clinical trial number NCT02494570 for "A Phase 2 Study of ABI-009 in Patients With Advanced Malignant PEComa (AMPECT)" at ClinicalTrials.gov