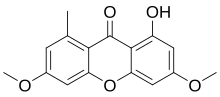
Lichexanthone
| |
| Names | |
|---|---|
| IUPAC name
1-Hydroxy-3,6-dimethoxy-8-methyl-9H-xanthen-9-one
| |
| Other names
Lichenxanthone,
1-hydroxy-3,6-dimethoxy-8-methylxanthen-9-one | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H14O5 | |
| Molar mass | 286.283 g·mol−1 |
| Appearance | long yellow prismatic crystals |
| Density | 1.323 g/cm3 |
| Melting point | 189–190 °C (372–374 °F; 462–463 K) |
| Boiling point | 494 °C (921 °F) |
| Structure[1] | |
Monoclinic
| |
| P21/c (No. 14) | |
a = 11.6405 Å, b = 7.5444 Å, c = 15.2341 Å
| |
Lattice volume (V)
|
1307.26 Å3 |
Formula units (Z)
|
4 |
| Hazards | |
| Flash point | 186.9 °C (368.4 °F) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lichexanthone is an
In lichens, the
History
Lichexanthone was first reported by Japanese chemists Yasuhiko Asahina and Hisasi Nogami in 1942. They isolated the lichen product from Parmelia formosana[2] (known today as Hypotrachyna osseoalba), a lichen that is widespread in Asia.[3] Another early publication described its isolation from Parmelia quercina (now Parmelina quercina[4]).[5] Lichexanthone was the first xanthone to be reported from lichens,[6] and it was given its name by Asahina and Nogami for this reason.[2]
Asahina and Nogami used a chemical method called potash fusion (
Properties
Lichexanthone is a member of the class of chemical compounds called
The
Biological activities
Various
In laboratory tests, the presence of lichexanthone enhances the
Biosynthesis
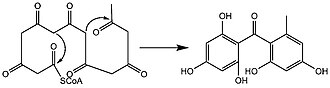
In lichens, biosynthesis of lichexanthone occurs through the acetate-malonate
A standardized
Occurrence

Although first isolated from foliose (leafy) Parmelia species, lichexanthone has since been found in a wide variety of lichens. For example, in the foliose genus Hypotrachyna, it is found in about a dozen species; when present, it usually completely replaces other cortical substances common in that genus, like atranorin and usnic acid.[12] The presence or absence of lichexanthone is a character used in classifying species of the predominantly tropical genus Pyxine; of about 70 species in the genus, 20 contain lichexanthone. This represents the largest group of foliose lichens with the compound, as it is generally restricted to some groups of tropical crustose lichens, chiefly pyrenocarps and Graphidaceae.[28] The large genus Pertusaria relies heavily on thallus chemistry to distinguish and classify species, some of which differ only in the presence or absence of a single secondary chemical. Lichexanthone, norlichexanthone, and their chlorinated derivatives are common in this genus.[29]
Although normally considered a secondary metabolite of lichens, lichexanthone has also been isolated from several plants, listed here organized by family:
- Clusiaceae: Garcinia forbesii[33]
- Euphorbiaceae: Croton cuneatus[34]
- Gentianaceae: Anthocleista djalonensis[35][36]
- Hypericaceae: Vismia baccifera var. dealbata[1]
- Meliaceae: Trichilia rubescens[37]
- Melastomataceae: Henriettella fascicularis[38]
- Minquartia guianensis[39]
- Polygonaceae: Ruprechtia tangarana[40]
- Feroniella lucida,[42] Zanthoxylum microcarpum, Z. valens,[43] Z. setulosum,[44] Z. tetraspermum[45]
- Sapindaceae: Cupania cinerea[22]
Lichexanthone has also been reported to occur in the bark of
Xanthones are known to have strong UV-absorbing properties.
Related compounds

Me = methyl (–CH3)
Norlichexanthone (1,3,6-trihydroxy-8-methylxanthone) differs from lichexanthone in having hydroxy rather than methoxy groups at positions 3 and 6.[11] In griseoxanthone C (1,6-dihydroxy-3-methoxy-8-methylxanthen-9-one), the methoxy at position 6 of lichexanthone is replaced with a hydroxy.[20] Dozens of chlorinated lichexanthone derivatives have been reported, some isolated from a variety of lichen species, and some produced synthetically. These derivatives are variously mono-, bi-, or trichlorinated with the chlorines at positions 2, 4, 5, and 7.[6] As of 2016, 62 molecules with the lichexanthone scaffold had been described, and another eight additional lichexanthone derivatives were considered "putative"–thought to exist in nature, but not yet discovered in lichens.[20]
The effects of chlorine
Eponyms
Some authors have explicitly named lichexanthone in the specific epithets of their published lichen species, thereby acknowledging the presence of this compound as an important taxonomic characteristic. These eponyms are listed here, followed by their author citation and year of publication. All of these species occur in Brazil:
- Parmotrema lichexanthonicum Eliasaro & Adler (1997)[53]
- Lecanora lichexanthona Guderley (2000)[54]
- Crypthonia lichexanthonica A.A.Menezes, M.Cáceres & Aptroot (2013)[55]
- Cryptothecia lichexanthonica E.L.Lima, Aptroot & M.Cáceres (2013)[56]
- Buellia lichexanthonica Aptroot & M.Cáceres (2017)[57]
- Chiodecton lichexanthonicum M.Cáceres & Aptroot (2017)[58]
- Enterographa lichexanthonica M.Cáceres & Aptroot (2017)[58]
- Cladonia lichexanthonica Aptroot & Cáceres (2018)[59]
- Pertusaria lichexanthofarinosa Aptroot & Cáceres (2018)[59]
- Pertusaria lichexanthoimmersa Aptroot & Cáceres (2018)[59]
- Pertusaria lichexanthoverrucosa Aptroot & Cáceres (2018)[59]
- Diorygma isidiolichexanthonicum Aptroot (2020)[60]
- Caprettia lichexanthotricha Aptroot & M.F.Souza (2021)[61]
- Lecanora lichexanthoxylina Aptroot & M.F.Souza (2021)[62]
- Lepra lichexanthonorstictica Aptroot (2021) – named for both lichexanthone and norstictic acid[63]
- Aggregatorygma lichexanthonicum Aptroot (2022)[64]
- Allographa lichexanthonica Aptroot (2023)[65]
- Ocellularia fuscolichexanthonica Aptroot (2023)[65]
- Ocellularia lichexanthocavata Aptroot (2023)[65]
In the case of
References
- ^ a b c d Buitrago Díaz, Alexis; Rojas Vera, Janne; Cote, Valentina; Bruno-Colmenárez, Julia; Díaz de Delgado, Graciela (2010). "NMR elucidation and crystal structure analysis of 1-hydroxy-3,6-dimethoxy-8-methyl-9h-xanthen-9-one (lichexanthone) isolated from Vismia baccifera (Guttiferae)" (PDF). Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas. 9 (6): 470–474.
- ^ .
"Da dasselbe ein in der Flechte zum ersten mal entdeckte Xanthone-Derivat ist, so nennen wir es 'Lichexanthone'." [Since it is a xanthone derivative first discovered in lichen, we call it lichexanthone.]
- JSTOR 1220900.
- ^ "Record Details: Parmelia quercina (Willd.) Vain., Term. Füz. 22: 279 (1899)". Index Fungorum. Retrieved 4 February 2022.
- ^ Aghoramurthy, K.; Seshadri, T.R. (1953). "An improved synthesis of lichexanthone". Journal of Scientific and Industrial Research (India). 12B: 350–352.
- ^ PMID 22617028.
- ^ .
- ISBN 978-0-08-045933-2.
- ^ Grover, P.K.; Shah, G.D.; Shah, R.C. (1956). "Xanthones: part V. A new synthesis of lichexanthone". Journal of Scientific and Industrial Research (India). 15B: 629–630.
- .
- ^ OCLC 851387266.
- ^ .
- .
- .
- ^ Manojlovic, Nedeljko T.; Vasiljevic, Perica J.; Marković, Zoran S. (2010). "Antimicrobial activity of extracts and various fractions of chloroform extract from the lichen Laurera benguelensis". Journal of Biological Research-Thessaloniki. 13: 27–34.
- ^ ISBN 978-3-030-16813-1.
- PMID 19683421.
- PMID 9795033.
- ^ PMID 23106287.
- ^ PMID 26950106.
- PMID 22061662.
- ^ PMID 21438586.
- PMID 23207680.
- .
- ISBN 978-0-521-69216-8.
- .
- .
- .
- ISBN 978-3-443-58048-3.
- PMID 28930201.
- .
- .
- PMID 34680496.
- PMID 16314057.
- .
- PMID 26432351.
- PMID 29502449.
- PMID 12502307.
- .
- PMID 12932125.
- .
- .
- .
- PMID 22312711.
- PMID 11324918.
- .
- PMID 15280651.
- ^ Frisvad, Jens C.; Smedsgaard, Jørn; Larsen, Thomas O.; Samson, Robert A. (2004). "Mycotoxins, drugs and other extrolites produced by species in Penicillium subgenus Penicillium" (PDF). Studies in Mycology. 49: 201–241.
- hdl:1885/57129.
- ISBN 978-3-443-58032-2.
- .
- .
- ^ Eliasaro, Sionara; Adler, Monica T. (1997). "Two new species and new reports in the Parmeliaceae sensu stricto (lichenized Ascomycotina) from Brazil". Mycotaxon. 63: 49–56.
- JSTOR 3244290.
- ^ .
- .
- .
- ^ .
- ^ .
- ^ Aptroot, André; Feuerstein, Shirley (2020). "New Graphidaceae from South and Central Brazil" (PDF). Archive for Lichenology. 16: 1–10.
- ^ .
- .
- ^ Aptroot, A.; Spielmann, A.A.; Gumboski, E.L. (2021). "New lichen species and records from Santa Catarina and Rio Grande do Sul, Brazil" (PDF). Archive for Lichenology. 23: 1–18.
- .
- ^ .
