MLH1
DNA mismatch repair protein Mlh1 or MutL protein homolog 1 is a
Function
Variants in this gene can cause
Role in DNA mismatch repair
MLH1 protein is one component of a system of seven
DNA mismatches occur where one base is improperly paired with another base, or where there is a short addition or deletion in one strand of DNA that is not matched in the other strand. Mismatches commonly occur as a result of DNA replication errors or during genetic recombination. Recognizing those mismatches and repairing them is important for cells because failure to do so results in microsatellite instability] and an elevated spontaneous mutation rate (mutator phenotype). Among 20 cancers evaluated, microsatellite instable colon cancer (mismatch repair deficient) had the second highest frequency of mutations (after melanoma).
A heterodimer between MSH2 and MSH6 first recognizes the mismatch, although a heterodimer between MSH2 and MSH3 also can start the process. The formation of the MSH2-MSH6 heterodimer accommodates a second heterodimer of MLH1 and PMS2, although a heterodimer between MLH1 and either PMS3 or MLH3 can substitute for PMS2. This protein complex formed between the 2 sets of heterodimers enables initiation of repair of the mismatch defect.[5]
Other gene products involved in mismatch repair (subsequent to initiation by DNA mismatch repair genes) include
Deficient expression in cancer
| Cancer type | Frequency of deficiency in cancer | Frequency of deficiency in adjacent field defect |
|---|---|---|
| Stomach | 32%[10][11] | 24–28% |
| Stomach (foveolar type tumors) | 74%[12] | 71% |
| Stomach in high-incidence Kashmir Valley | 73%[13] | 20% |
| Esophageal | 73%[14] | 27% |
| Head and neck squamous cell carcinoma (HNSCC) | 31–33%[15][16] | 20–25% |
| Non-small cell lung cancer (NSCLC) | 69%[17] | 72% |
| Colorectal | 10%[6] |
Epigenetic repression
Only a minority of sporadic cancers with a DNA repair deficiency have a mutation in a DNA repair gene. However, a majority of sporadic cancers with a DNA repair deficiency do have one or more epigenetic alterations that reduce or silence DNA repair gene expression.[18] In the table above, the majority of deficiencies of MLH1 were due to methylation of the promoter region of the MLH1 gene. Another epigenetic mechanism reducing MLH1 expression is over-expression of miR-155.[19] MiR-155 targets MLH1 and MSH2 and an inverse correlation between the expression of miR-155 and the expression of MLH1 or MSH2 proteins was found in human colorectal cancer.[19]
Deficiency in field defects
A field defect is an area or "field" of epithelium that has been preconditioned by epigenetic changes and/or mutations so as to predispose it towards development of cancer. As pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors in vivo, or on discrete neoplastic foci in vitro.[20] Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion."[21] Similarly, Vogelstein et al.[22] point out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells.
In the Table above, MLH1 deficiencies were noted in the field defects (histologically normal tissues) surrounding most of the cancers. If MLH1 is epigenetically reduced or silenced, it would not likely confer a selective advantage upon a stem cell. However, reduced or absent expression of MLH1 would cause increased rates of mutation, and one or more of the mutated genes may provide the cell with a selective advantage. The expression-deficient MLH1 gene could then be carried along as a selectively neutral or only slightly deleterious passenger (hitch-hiker) gene when the mutated stem cell generates an expanded clone. The continued presence of a clone with an epigenetically repressed MLH1 would continue to generate further mutations, some of which could produce a tumor.
Repression in coordination with other DNA repair genes
In a cancer, multiple DNA repair genes are often found to be simultaneously repressed.
Deficient expression of multiple DNA repair genes are often found in cancers,[18] and may contribute to the thousands of mutations usually found in cancers (see Mutation frequencies in cancers).
Meiosis
In addition to its role in DNA mismatch repair, MLH1 protein is also involved in meiotic crossing over.[25] MLH1 forms a heterodimer with MLH3 that appears to be necessary for oocytes to progress through metaphase II of meiosis.[26] Female and male MLH1(-/-) mutant mice are infertile, and sterility is associated with a reduced level of chiasmata.[25][27] During spermatogenesis in MLH1(-/-) mutant mice chromosomes often separate prematurely and there is frequent arrest in the first division of meiosis.[25] In humans, a common variant of the MLH1 gene is associated with increased risk of sperm damage and male infertility.[28]
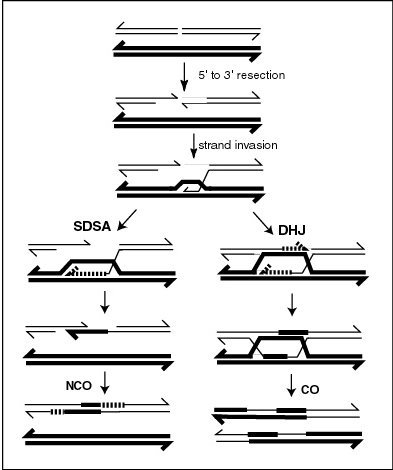
MLH1 protein appears to localize to sites of crossing over in meiotic chromosomes.[25] Recombination during meiosis is often initiated by a DNA double-strand break (DSB) as illustrated in the accompanying diagram. During recombination, sections of DNA at the 5' ends of the break are cut away in a process called resection. In the strand invasion step that follows, an overhanging 3' end of the broken DNA molecule then "invades" the DNA of an homologous chromosome that is not broken forming a displacement loop (D-loop). After strand invasion, the further sequence of events may follow either of two main pathways leading to a crossover (CO) or a non-crossover (NCO) recombinant (see Genetic recombination). The pathway leading to a CO involves a double Holliday junction (DHJ) intermediate. Holliday junctions need to be resolved for CO recombination to be completed.
In the budding yeast
Clinical significance
It can also be associated with
Interactions
MLH1 has been shown to
See also
- Mismatch repair#MutH: an endonuclease present in E. coli and Salmonella
References
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000032498 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Entrez Gene: MLH1 mutL homolog 1, colon cancer, nonpolyposis type 2 (E. coli)".
- ^ PMID 18543306.
- ^ PMID 15887099.
- PMID 25956862.
- PMID 18157157.
- PMID 24767944.
- PMID 24509146.
- PMID 12163364.
- PMID 10980110.
- PMID 23098428.
- PMID 25436004.
- PMID 21353335.
- S2CID 8357370.
- PMID 15958624.
- ^ PMID 25987950.
- ^ PMID 20351277.
- S2CID 44981539.
- PMID 10655514.
- PMID 23539594.
- S2CID 54278905.
- PMID 12861399.
- ^ S2CID 37096830.
- PMID 18057311.
- PMID 12114115.
- PMID 22594646.
- ^ PMID 24443562.
- PMID 24403070.
- PMID 22500800.
- S2CID 21591979.
- PMID 10783165.
- PMID 11325959.
- PMID 11470874.
- PMID 11691925.
- PMID 11427529.
- PMID 10097147.
- PMID 10928988.
- ^ PMID 12584560.
- PMID 11292842.
- PMID 10037723.
Further reading
- Paraf F, Sasseville D, Watters AK, Narod S, Ginsburg O, Shibata H, Jothy S (1995). "Clinicopathological relevance of the association between gastrointestinal and sebaceous neoplasms: the Muir-Torre syndrome". Hum. Pathol. 26 (4): 422–7. PMID 7705822.
- Kolodner RD (1996). "Mismatch repair: mechanisms and relationship to cancer susceptibility". Trends Biochem. Sci. 20 (10): 397–401. PMID 8533151.
- Peltomäki P, de la Chapelle A (1997). Mutations predisposing to hereditary nonpolyposis colorectal cancer. Advances in Cancer Research. Vol. 71. pp. 93–119. )
- Papadopoulos N, Lindblom A (1997). "Molecular basis of HNPCC: mutations of MMR genes". Hum. Mutat. 10 (2): 89–99. S2CID 6799575.
- Kauh J, Umbreit J (2004). "Colorectal cancer prevention". Current Problems in Cancer. 28 (5): 240–64. PMID 15375803.
- Warusavitarne J, Schnitzler M (2007). "The role of chemotherapy in microsatellite unstable (MSI-H) colorectal cancer". International Journal of Colorectal Disease. 22 (7): 739–48. S2CID 6460105.
- Niv Y (2007). "Microsatellite instability and MLH1 promoter hypermethylation in colorectal cancer". World J. Gastroenterol. 13 (12): 1767–9. PMID 17465465.
External links
- Information on HNPCC from the National Institute of Health
- GeneReviews/NCBI/NIH/UW entry on Lynch syndrome
- MLH1+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P40692 (DNA mismatch repair protein Mlh1) at the PDBe-KB.
