N-Bromosuccinimide
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Bromopyrrolidine-2,5-dione | |||
| Other names
N-bromosuccinimide; NBS
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 113916 | |||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.004.435 | ||
| EC Number |
| ||
| 26634 | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H4BrNO2 | |||
| Molar mass | 177.985 g·mol−1 | ||
| Appearance | White solid | ||
| Density | 2.098 g/cm3 (solid) | ||
| Melting point | 175 to 178 °C (347 to 352 °F; 448 to 451 K) | ||
| Boiling point | 339 °C (642 °F; 612 K) | ||
| 14.7 g/L (25 °C) | |||
| Solubility in CCl4 | Insoluble (25 °C) | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| Safety data sheet (SDS) | [1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
N-Bromosuccinimide or NBS is a
Preparation
NBS is commercially available. It can also be synthesized in the laboratory. To do so,
Crude NBS gives better yield in the
Reactions
Addition to alkenes
NBS will react with alkenes 1 in aqueous solvents to give
Side reactions include the formation of α-bromoketones and dibromo compounds. These can be minimized by the use of freshly recrystallized NBS.
With the addition of
Allylic and benzylic bromination
Standard conditions for using NBS in allylic and/or benzylic
The
In the above reaction, while a mixture of isomeric allylic bromide products are possible, only one is created due to the greater stability of the 4-position radical over the methyl-centered radical.
Bromination of carbonyl derivatives
NBS can α-brominate carbonyl derivatives via either a radical pathway (as above) or via acid-catalysis. For example, hexanoyl chloride 1 can be brominated in the alpha-position by NBS using acid catalysis.[11]
The reaction of enolates, enol ethers, or enol acetates with NBS is the preferred method of α-bromination as it is high-yielding with few side-products.[12][13]
Bromination of aromatic derivatives
Electron-rich
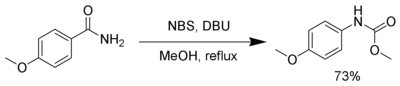
Hofmann rearrangement
NBS, in the presence of a strong base, such as
Selective oxidation of alcohols
It is uncommon, but possible for NBS to oxidize alcohols. E. J. Corey et al. found that one can selectively oxidize secondary alcohols in the presence of primary alcohols using NBS in aqueous dimethoxyethane (DME).[19]
Oxidative decarboxylation of α-amino acids
NBS electrophilically brominates the amine, which is followed by decarboxylation and release of an imine. Further hydrolysis will yield an aldehyde and ammonia.[20][21] (cf. non-oxidative PLP dependent decarboxylation)
Precautions
Although NBS is easier and safer to handle than bromine, precautions should be taken to avoid inhalation. NBS should be stored in a refrigerator. NBS will decompose over time giving off bromine. Pure NBS is white, but it is often found to be off-white or brown colored by bromine.
In general, reactions involving NBS are exothermic. Therefore, extra precautions should be taken when using on a large scale.
See also
- Halohydrin formation
- N-Chlorosuccinimide
- N-Iodosuccinimide
References
- .
- .
- ^ Hanzlik, R. P. "Selective epoxidation of terminal double bonds". Organic Syntheses; Collected Volumes, vol. 6, p. 560.
- .
- ^ Haufe, G.; Alvernhe, G.; Laurent, A.; Ernet, T.; Goj, O.; Kröger, S.; Sattler, A. (2004). "Bromofluorination of alkenes". Organic Syntheses; Collected Volumes, vol. 10, p. 128.
- PMID 18887958.
- .
- .
- .
- .
- ^ Harpp, D. N.; Bao, L. Q.; Coyle, C.; Gleason, J. G.; Horovitch, S. (1988). "2-Bromohexanoyl chloride". Organic Syntheses; Collected Volumes, vol. 6, p. 190.
- .
- .
- ^ Amat, M.; Hadida, S.; Sathyanarayana, S.; Bosc, J. (1998). "Regioselective synthesis of 3-substituted indoles". Organic Syntheses; Collected Volumes, vol. 9, p. 417.
- .
- ^ Brown, W. D.; Gouliaev, A. H. (2005). "Synthesis of 5-bromoisoquinoline and 5-bromo-8-nitroisoquinoline". Organic Syntheses. 81: 98.
- .
- ^ Keillor, J. W.; Huang, X. (2004). "Methyl carbamate formation via modified Hofmann rearrangement reactions". Organic Syntheses; Collected Volumes, vol. 10, p. 549.
- .
- .
- PMID 25222505.